 Back
BackBiochemical Tests for Differentiation of Microorganisms
Study Guide - Smart Notes

Biochemical Tests in Microbiology
Introduction to Biochemical Tests
Biochemical tests are essential tools in microbiology for differentiating microorganisms based on their unique metabolic and enzymatic characteristics. These tests exploit differences in microbial metabolism, allowing for the identification and classification of bacteria that may appear similar morphologically.
Biochemical characteristics are determined by specific enzymatic reactions in metabolic pathways.
Tests are especially useful for distinguishing closely related bacteria, such as Escherichia coli, Salmonella, and Shigella.
Categories of Differential Biochemical Tests
Energy metabolism: Includes fermentation of carbohydrates and aerobic/anaerobic respiration.
Utilization of medium components: Tests for the ability to use specific nutrients.
Amino acid metabolism: Decarboxylation and deamination reactions.
Hydrolytic reactions: Require specific enzymes to break down substrates.
Combination tests: Multiple reactions in a single medium.
Miscellaneous tests: Other unique metabolic activities.
IMViC Tests
Overview of IMViC Tests
The IMViC series (Indole, Methyl Red, Voges-Proskauer, Citrate) is a set of biochemical tests used to differentiate members of the family Enterobacteriaceae and distinguish them from other Gram-negative rods.
Indole Test
Methyl Red Test
Voges-Proskauer Test
Citrate Test
Indole Test Using SIM Medium
The SIM (Sulfide, Indole, Motility) medium is a combination differential medium that allows for the detection of sulfur reduction, indole production, and motility.
Sulfur reduction: Production of H2S from cysteine or sulfate reduction forms a black precipitate (FeS).
Indole production: Tryptophan is hydrolyzed by tryptophanase to indole, pyruvate, and ammonia. Indole reacts with Kovac’s reagent to produce a red color.
Motility: Growth radiating from the stab line indicates motility.


Interpretation of SIM Results
Motility: Hazy appearance throughout the medium indicates motility.
Sulfur reduction: Black precipitate indicates positive result.
Indole production: Red color after Kovac’s reagent indicates positive result.
Methyl Red and Voges-Proskauer Tests
Methyl Red Test
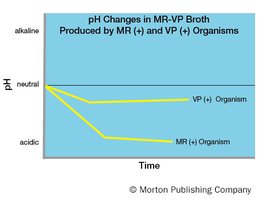
The Methyl Red (MR) test detects mixed acid fermentation, where stable acids are produced that lower the pH below 4.4. Methyl red dye is used as a pH indicator.
Positive result: Red color at pH < 4.4.
Negative result: Yellow color at pH > 6.2.
Voges-Proskauer Test
The Voges-Proskauer (VP) test detects the conversion of glucose fermentation products to acetoin and 2,3-butanediol. VP reagents react with acetoin to form a red color.
Positive result: Red color after addition of reagents.
Negative result: No color change or copper color.
MRVP Medium and Procedure
Contains peptone, glucose, and potassium phosphate buffer.
Incubate at 37°C for 48 hours.
Add methyl red for MR test; add VP reagents A and B for VP test.
Citrate Utilization Test
Principle of Citrate Test
The citrate test determines the ability of an organism to use citrate as its sole carbon source and ammonium phosphate as the sole nitrogen source. Simmons citrate agar contains bromothymol blue as a pH indicator.
Positive result: Growth and blue color (alkaline pH).
Negative result: No growth and green color (neutral pH).

Nitrate Reduction Test
Principle of Nitrate Reduction
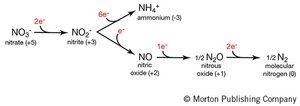
This test identifies bacteria capable of anaerobic respiration using nitrate as a terminal electron acceptor. Nitrate reductase reduces nitrate (NO3-) to nitrite (NO2-), and further reduction (denitrification) can produce nitrogen gas (N2).
Medium contains potassium nitrate, beef extract, peptone, and a Durham tube to capture gas.
Gas production in non-fermenters indicates denitrification.
Interpretation of Nitrate Test
After incubation, observe for gas in Durham tube.
If no gas, add Nitrate Reagents A and B:
Red color: Nitrate reduced to nitrite (positive).
No color: Add zinc dust.
Red after zinc: Nitrate not reduced (negative).
No color after zinc: Nitrate reduced beyond nitrite (positive for further reduction).

Urease Test
Principle of Urease Test
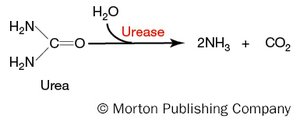
The urease test detects the ability of bacteria to hydrolyze urea to ammonia and carbon dioxide using the enzyme urease. The production of ammonia raises the pH, detected by phenol red indicator.
Positive result: Pink to magenta color (alkaline pH > 7.4).
Negative result: Yellow or orange color (acidic or neutral pH).

Summary Table: Key Biochemical Tests
Test | Principle | Positive Result | Negative Result |
|---|---|---|---|
Indole (SIM) | Tryptophan hydrolysis | Red ring after Kovac's reagent | No color change |
Methyl Red | Mixed acid fermentation | Red color | Yellow color |
Voges-Proskauer | Acetoin production | Red color | No color/copper |
Citrate | Citrate utilization | Growth, blue color | No growth, green color |
Nitrate Reduction | Nitrate to nitrite/N2 | Red after reagents or gas | Red after zinc |
Urease | Urea hydrolysis | Pink/magenta | Yellow/orange |
Conclusion
Biochemical tests are fundamental for the identification and differentiation of bacteria in clinical and research microbiology. Understanding the principles, procedures, and interpretations of these tests is essential for accurate microbial classification and diagnosis.
