 Back
BackBiofilms: Structure, Life Cycle, and Clinical Significance
Study Guide - Smart Notes

Biofilms
Definition and Overview
A biofilm is a structured community of microorganisms, such as bacteria or fungi, that adhere to a surface and are embedded within a self-produced matrix of extracellular polymeric substances (EPS). This slimy protective layer enhances their survival under adverse conditions, increases resistance to antibiotics (up to 1,000 times more than planktonic cells), and helps them evade the immune system.
Key Terms: Extracellular Polymeric Substances (EPS) – a mixture of polysaccharides, proteins, and DNA that forms the biofilm matrix.
Antibiotic Resistance: Biofilm-embedded bacteria are significantly more resistant to antimicrobial agents compared to free-floating (planktonic) cells.
Where Biofilms Are Found
Biofilms are ubiquitous and can develop on almost any surface exposed to water, including natural, industrial, and medical environments.
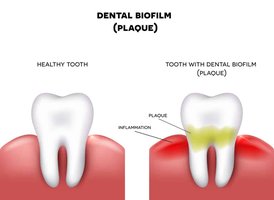
Human Body: Biofilms are present on teeth (dental plaque), mucosal surfaces (respiratory tract, gut, vagina), and medical implants (catheters, heart valves, pacemakers, artificial joints).
Beneficial Biofilms: Lactobacilli in the vagina inhibit the growth of harmful microbes.
Harmful Biofilms: In cystic fibrosis patients, lung biofilms cause persistent infections that are difficult to treat.
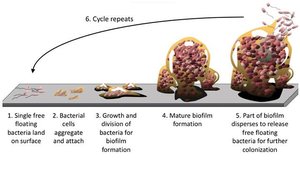
Biofilm Life Cycle
Stages of Biofilm Development
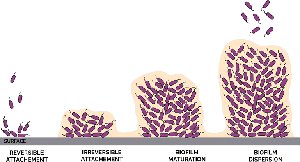
The biofilm life cycle is a highly organized process, often described as a 'microbial city' with specialized roles and communication networks. It consists of five main stages:
Initial Attachment (Reversible): Free-floating bacteria make transient contact with a surface using structures like pili or flagella. They can be removed by gentle rinsing.
Irreversible Adhesion: Bacteria firmly attach and begin producing EPS, anchoring themselves and each other to the surface.
Maturation I (Microcolony Formation): Cells proliferate and form microcolonies, initiating cell-to-cell communication (quorum sensing).
Maturation II (3D Structure): The biofilm develops complex three-dimensional architecture with water channels for nutrient and waste exchange. The EPS matrix thickens, enhancing defense.
Dispersion: Some cells actively break free from the biofilm to colonize new surfaces, restarting the cycle.
Biofilm Differentiation and Communication
Within a biofilm, cells differentiate into specialized roles, despite being genetically identical. This division of labor is coordinated by chemical and physical signals.
Fast-growing cells: Located on the periphery, focus on rapid expansion.
Matrix producers: Synthesize the EPS matrix for structural integrity and protection.
Persister cells: Dormant cells deep within the biofilm, highly resistant to antibiotics and environmental stress.
Disperser cells: Facilitate the spread of the biofilm to new locations when conditions change.
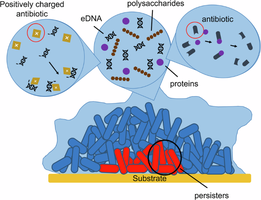
Quorum Sensing: Bacterial Communication in Biofilms
Mechanism and Importance
Quorum sensing (QS) is a cell-to-cell communication process that enables bacteria to sense their population density through the release and detection of signaling molecules called autoinducers. When a threshold concentration is reached, coordinated gene expression occurs, regulating biofilm formation, virulence, and other group behaviors.
Positive Feedback Loop: Signal molecules accumulate as the population grows, triggering synchronized responses once a critical threshold is reached.
Types of Quorum Sensing Signals
Acyl-homoserine lactones (AHLs): Used by Gram-negative bacteria. Synthesized by LuxI enzymes and detected by LuxR receptors. Structure: homoserine lactone ring with variable side chain.
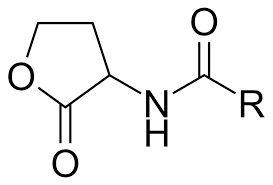
Autoinducing peptides (AIPs): Used by Gram-positive bacteria. Processed into cyclic forms and detected by membrane-bound receptors.
Autoinducer-2 (AI-2): A universal signal for interspecies communication in mixed biofilm communities.
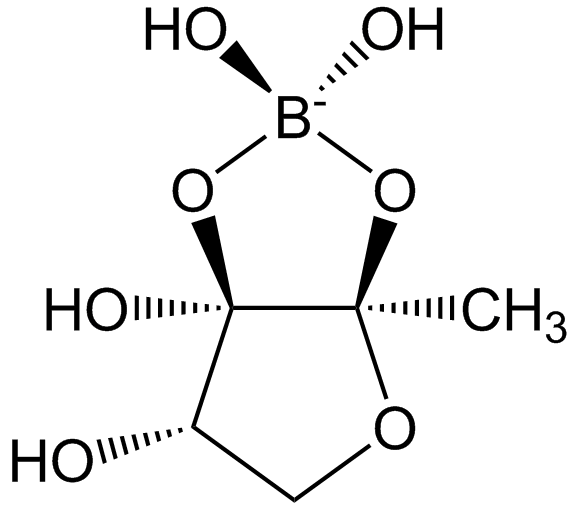
Critical Tasks Managed by Quorum Sensing
Matrix construction: Regulates EPS production.
Structural organization: Coordinates formation of water channels and architecture.
Virulence: Controls timing of toxin production to maximize infection success.
Dispersion: Triggers release of cells when conditions become unfavorable.
Quorum Quenching: Disrupting Bacterial Communication
Quorum quenching (QQ) refers to strategies that disrupt QS, thereby reducing biofilm formation and virulence without directly killing bacteria. This approach may help prevent antibiotic resistance.
Signal degradation: Enzymes break down autoinducers.
Receptor blockers: Molecules that prevent signal detection.
Natural inhibitors: Compounds from garlic, turmeric, and clove oil can interfere with QS.
Pseudomonas aeruginosa and Quorum Sensing
Multi-Layered QS Network
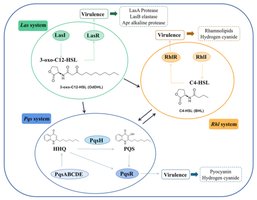
Pseudomonas aeruginosa is a model organism for studying biofilms and QS. It possesses four interconnected QS systems that regulate virulence, biofilm formation, and adaptation to stress, especially in chronic infections (e.g., cystic fibrosis lungs).
Las system: Master regulator using 3-oxo-C12-HSL and LasR.
Rhl system: Uses C4-HSL and RhlR; important for chronic infection factors.
PQS system: Links Las and Rhl systems; regulates biofilm and iron uptake.
IQS system: Activated under stress, maintains virulence in harsh conditions.
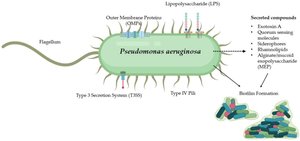
Host Interaction and Immune Evasion
Signal integration: Detects host molecules to enhance its own signaling.
Immune evasion: Some QS molecules can suppress or damage host immune cells.
Treatment Strategies for P. aeruginosa Biofilms
Physical disruption: Removal or debridement of biofilm from wounds or devices.
Enzymatic "slime breakers": Enzymes like DNase degrade the EPS matrix.
Combination antibiotics: High-dose or inhaled antibiotics, often used together for increased efficacy.
Topical treatments: Diluted acetic acid (vinegar) and medical-grade honey can disrupt biofilms.
Biofilms in Everyday Life: The Case of "Permastink"
Biofilms and Odor in Synthetic Fabrics
"Permastink" refers to persistent odors in synthetic fabrics (e.g., polyester, spandex) caused by biofilm formation. Bacteria use QS to coordinate the production of EPS, adhere to fibers, and metabolize sweat into malodorous compounds.
Coordinated defense: EPS shields bacteria from washing.
Metabolic switch: QS triggers breakdown of sweat into smelly molecules (e.g., isovaleric acid).
Re-stink effect: Surviving bacteria reactivate with heat and sweat, causing odor to return.
Why Synthetics Are Prone to Biofilm Odor
Oil traps: Synthetics repel water but retain oils, providing nutrients for bacteria.
Biofilm hiding spots: Microscopic crevices in synthetic fibers facilitate bacterial attachment and protection.
Strategies to Remove Biofilms from Laundry
Biofilm disruptors: Enzyme-based laundry additives break down EPS, allowing detergents to remove bacteria and residues.

High temperature: Washing at higher temperatures (if fabric-safe) denatures biofilm proteins.

Acidic rinses: Adding white vinegar to the rinse cycle helps dissolve oils and weaken the biofilm matrix.

Summary Table: Biofilm Life Cycle Stages
Stage | Description |
|---|---|
1. Initial Attachment | Reversible contact with surface; easily removed |
2. Irreversible Adhesion | Firm attachment and EPS production |
3. Maturation I | Microcolony formation and quorum sensing begins |
4. Maturation II | 3D structure, water channels, increased defense |
5. Dispersion | Cells released to colonize new surfaces |
Key Equations and Concepts
Quorum Sensing Threshold: The critical concentration of autoinducers required to trigger a coordinated response is given by:
Where is the autoinducer concentration, is the number of cells, is the volume of a single cell, and is the total volume of the environment.
Additional info: This equation is a simplified representation; actual QS dynamics are more complex and species-dependent.
