 Back
BackChapter 2: Chemical Principles – Atoms, Molecules, and the Chemistry of Life
Study Guide - Smart Notes

Chapter 2: Chemical Principles – Atoms, Molecules, and the Chemistry of Life
Matter and Atoms
All living and non-living things are composed of matter, which consists of atoms. Atoms are the fundamental building blocks of matter and are incredibly small, about 10 million times smaller than a typical object visible to the naked eye.
Matter: Anything that has mass and occupies space (solids, liquids, gases).
Atom: The smallest unit of an element that retains the properties of that element.
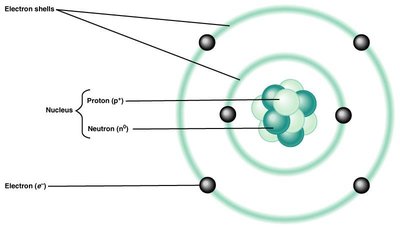
Structure of an Atom: Atoms consist of a central nucleus (containing protons and neutrons) surrounded by electrons in defined shells.
Protons (p+): Positively charged particles in the nucleus; determine the element's identity.
Neutrons (n0): Neutral particles in the nucleus.
Electrons (e-): Negatively charged particles orbiting the nucleus in shells; involved in chemical bonding.
Charge Balance: Atoms typically have equal numbers of protons and electrons, resulting in no net charge.
Elements
Elements are pure substances consisting of only one type of atom, defined by the number of protons in their nuclei.
Element: A substance made entirely of one type of atom (e.g., all atoms with 6 protons are carbon).
There are over 100 known elements, each with a unique one- or two-letter symbol.
The number of protons distinguishes one element from another.
Molecules and Chemical Bonds
Atoms rarely exist alone; they combine to form molecules through chemical bonds. Molecules can consist of atoms of the same element or different elements (compounds).
Molecule: Two or more atoms bonded together.
Compound: A molecule composed of atoms from different elements (e.g., H2O, C6H12O6).
Bonds: Forces that hold atoms together in molecules.
Ionic Bonds
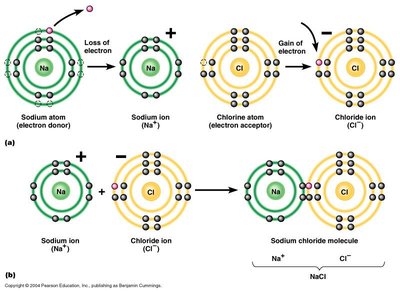
Ionic bonds form when atoms transfer electrons, resulting in oppositely charged ions that attract each other. Compounds formed by ionic bonds are often called salts.
Ion: An atom or molecule with a net electric charge due to the loss or gain of electrons.
Ionic Bond: The electrostatic attraction between oppositely charged ions.
Covalent Bonds
Covalent bonds involve the sharing of electrons between atoms. These bonds are stronger than ionic bonds and do not result in charged atoms.
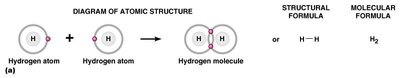
Single Bond: Two electrons shared (e.g., H2).
Double Bond: Four electrons shared (e.g., O2).
Triple Bond: Six electrons shared.
Chemical Notation and Structural Formulas
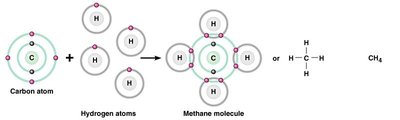
Chemical formulas use element symbols and subscripts to indicate the types and numbers of atoms in a molecule. Structural formulas show the arrangement of atoms and bonds.
Chemical Formula: Indicates the elements and their ratios (e.g., H2O, CH4).
Structural Formula: Shows how atoms are connected (e.g., H—O—H for water).
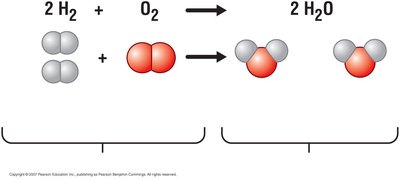
Chemical Reactions
Chemical reactions involve breaking and forming bonds to convert reactants into products. These reactions are essential for life, enabling cells to build and break down molecules as needed.
Reactants: Starting materials in a chemical reaction.
Products: Substances formed as a result of the reaction.
General Equation:
Polar Molecules and Hydrogen Bonds
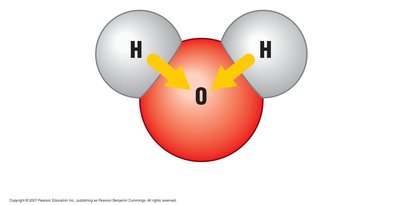
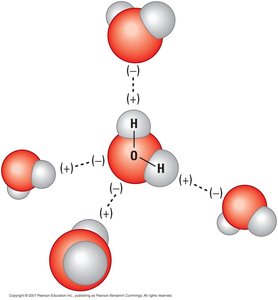
Polar molecules have regions of slight positive and negative charge due to unequal sharing of electrons. Water is a classic example, with oxygen being more electronegative than hydrogen.
Polar Molecule: A molecule with an uneven distribution of charge (e.g., H2O).
Hydrogen Bond: A weak attraction between the slightly positive hydrogen of one polar molecule and the slightly negative atom (O, N, or S) of another.
Organic and Inorganic Molecules
Organic molecules contain both carbon and hydrogen, often with other elements in functional groups. Inorganic molecules lack either carbon or hydrogen or both.
Organic Molecules: Carbohydrates, lipids, proteins, nucleic acids.
Inorganic Molecules: Water, salts, acids, bases.
Functional Groups: Specific groups of atoms that confer chemical properties to organic molecules.

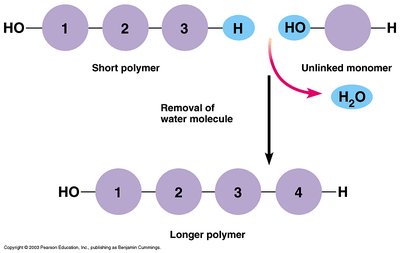
Macromolecules and Polymers
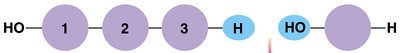
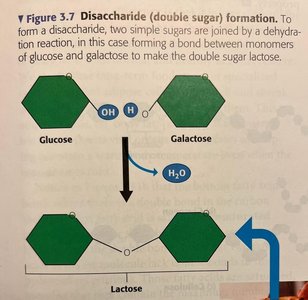
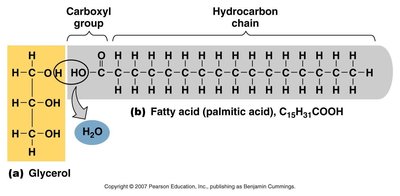
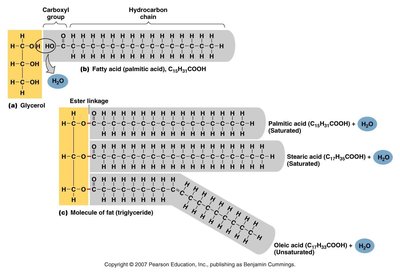
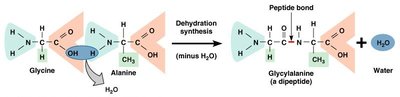
Macromolecules are large organic molecules made by joining smaller units (monomers) into chains (polymers) through dehydration synthesis. Hydrolysis breaks polymers into monomers by adding water.
Dehydration Synthesis: Joins monomers by removing water.
Hydrolysis: Breaks polymers into monomers by adding water.
Carbohydrates
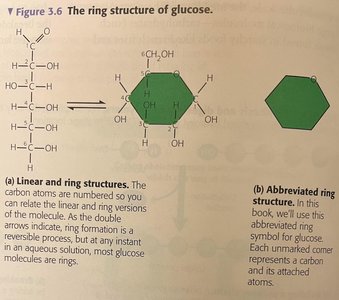
Carbohydrates are organic molecules consisting of carbon, hydrogen, and oxygen, typically with the formula (CH2O)n. They serve as energy sources and structural materials.
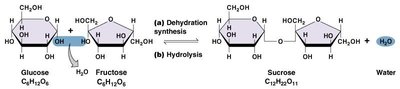
Monosaccharides: Simple sugars (e.g., glucose, fructose); monomers of carbohydrates.
Disaccharides: Two monosaccharides joined by dehydration synthesis (e.g., sucrose, lactose).
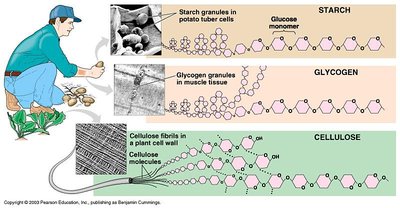
Polysaccharides: Long chains of monosaccharides (e.g., starch, glycogen, cellulose, peptidoglycan).
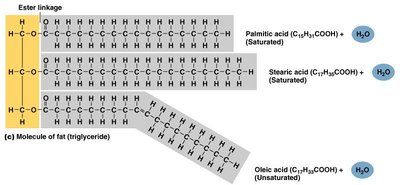
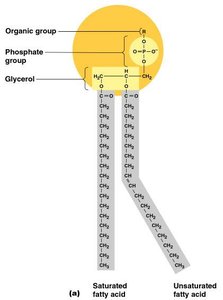
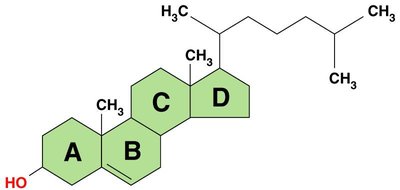
Lipids
Lipids are nonpolar molecules that do not mix with water. They are important for energy storage and as structural components of cell membranes.
Triglycerides: Composed of glycerol and three fatty acids; main form of stored energy.
Phospholipids: Glycerol, two fatty acids, and a phosphate group; major component of cell membranes.
Steroids: Four fused carbon rings; include cholesterol and hormones.
Proteins
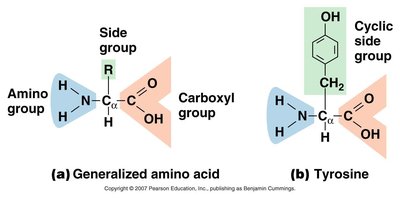
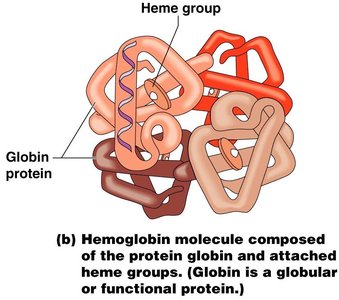
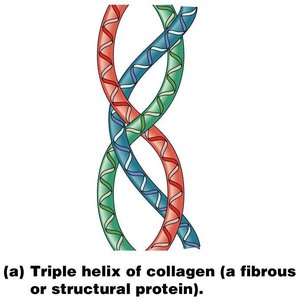
Proteins are polymers of amino acids and are essential for cell structure and function. They serve as enzymes, structural components, transporters, and more.
Amino Acid: Building block of proteins; contains an amino group, carboxyl group, hydrogen, and a unique side chain (R group).
Peptide Bond: Covalent bond joining amino acids in a polypeptide chain.
Polypeptide: Chain of amino acids; one or more polypeptides make a protein.
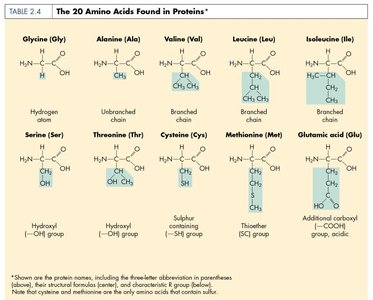
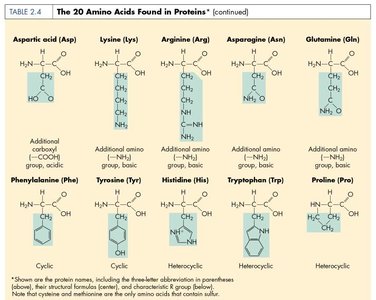
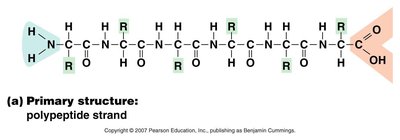
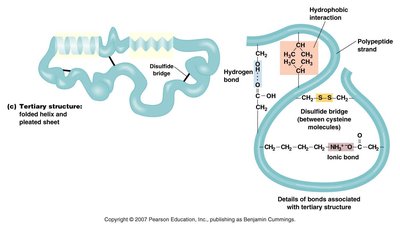
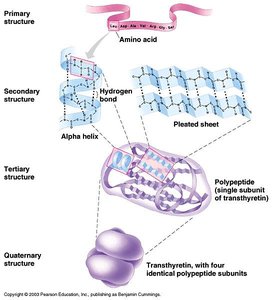
Levels of Protein Structure
Primary Structure: Sequence of amino acids in a polypeptide.
Secondary Structure: Local folding (alpha helix, beta sheet) stabilized by hydrogen bonds.
Tertiary Structure: Overall 3D shape of a single polypeptide, maintained by various interactions (hydrogen bonds, ionic bonds, disulfide bridges, hydrophobic interactions).
Quaternary Structure: Association of multiple polypeptides to form a functional protein (e.g., hemoglobin).
Nucleic Acids
Nucleic acids are polymers of nucleotides and store genetic information (DNA) or are involved in protein synthesis (RNA).
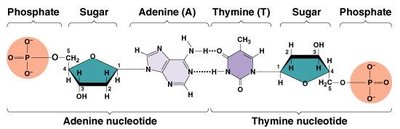
Nucleotide: Composed of a sugar (ribose or deoxyribose), a phosphate group, and a nitrogenous base.
DNA: Double-stranded helix; stores genetic information; bases pair A-T and C-G via hydrogen bonds.
RNA: Single-stranded; involved in protein synthesis; bases pair A-U and C-G.
ATP: The Energy Currency of the Cell
ATP (adenosine triphosphate) is a modified nucleotide that stores and releases energy for cellular processes.
ATP Structure: Adenine base, ribose sugar, and three phosphate groups.
ATP Hydrolysis: Releases energy by converting ATP to ADP and inorganic phosphate.
ATP Synthesis: Formed from ADP and phosphate using energy from food or sunlight.
Water: The Liquid of Life
Water is essential for life due to its unique properties as a polar molecule. It is the medium for chemical reactions, helps maintain temperature, and acts as a solvent for many substances.
High Heat Capacity: Maintains stable temperatures.
Excellent Solvent: Dissolves polar and ionic substances, enabling chemical reactions.
Liquid State: Supports movement and mixing of molecules.
Acids, Bases, and pH
Acids and bases affect the concentration of hydrogen ions (H+) in solution, influencing pH. Cells require a stable pH for optimal function.
Acid: Releases H+ ions in solution; increases acidity (lowers pH).
Base: Releases OH- ions or absorbs H+; decreases acidity (raises pH).
Salt: Compound formed from the neutralization of an acid and a base.
pH Scale: Ranges from 0 (most acidic) to 14 (most basic); 7 is neutral.
Buffers: Compounds that stabilize pH by absorbing or releasing H+ ions.
Summary Table: Types of Biological Molecules
Type | Monomer | Polymer | Function |
|---|---|---|---|
Carbohydrate | Monosaccharide | Polysaccharide | Energy, structure |
Lipid | Fatty acid, glycerol | Triglyceride, phospholipid | Energy storage, membranes |
Protein | Amino acid | Polypeptide | Structure, enzymes, transport |
Nucleic Acid | Nucleotide | DNA, RNA | Genetic information, protein synthesis |
