 Back
BackChapter 2: Chemical Principles – Microbiology Study Guide
Study Guide - Smart Notes

Q1. Describe the structure of an atom and its relation to the physical properties of elements.
Background
Topic: Atomic Structure and Chemical Properties
This question tests your understanding of atomic structure (protons, neutrons, electrons), atomic number, atomic mass, and how these relate to the properties of elements on the periodic table. It also covers how electronic configurations influence chemical behavior and molecule formation.
Key Terms and Concepts:
Atom: Smallest unit of an element, composed of protons, neutrons, and electrons.
Atomic Number: Number of protons in the nucleus.
Atomic Mass: Sum of protons and neutrons.
Electronic Configuration: Arrangement of electrons in shells around the nucleus.
Periodic Table: Organizes elements by atomic number and properties.
Step-by-Step Guidance
Recall that the atomic number determines the identity of an element and its position on the periodic table.
Understand that the atomic mass is the sum of protons and neutrons, which affects the isotope and stability of the atom.
Review how electrons are arranged in shells and how this configuration determines chemical reactivity and bonding.
Consider how atoms with incomplete outer shells tend to form bonds to achieve stability, leading to molecule formation.
Try solving on your own before revealing the answer!
Q2. Describe Endergonic vs Exergonic Reactions.
Background
Topic: Chemical Reactions and Energy Changes
This question focuses on the difference between reactions that absorb energy (endergonic) and those that release energy (exergonic), which is fundamental to metabolism in living organisms.
Key Terms and Concepts:
Endergonic Reaction: Absorbs energy from surroundings.
Exergonic Reaction: Releases energy to surroundings.
Activation Energy: Minimum energy required to start a reaction.
Step-by-Step Guidance
Define what it means for a reaction to be endergonic or exergonic in terms of energy flow.
Think about examples of each type of reaction in biological systems (e.g., photosynthesis vs. cellular respiration).
Consider the role of ATP in coupling these reactions in cells.
Try solving on your own before revealing the answer!
Q3. Define ionic bond, covalent bond (polar vs. non-polar), hydrogen bond, molecular weight and give examples. Indicate which are weak or strong bonds.
Background
Topic: Chemical Bonds and Molecular Structure
This question tests your ability to distinguish between different types of chemical bonds, their relative strengths, and their importance in biological molecules.
Key Terms and Concepts:
Ionic Bond: Transfer of electrons between atoms, forming charged ions.
Covalent Bond: Sharing of electrons between atoms (polar = unequal sharing, non-polar = equal sharing).
Hydrogen Bond: Weak attraction between a hydrogen atom and an electronegative atom (e.g., O or N).
Molecular Weight: Sum of atomic masses in a molecule.
Step-by-Step Guidance
Define each type of bond and provide a biological example (e.g., NaCl for ionic, H2O for polar covalent, O2 for non-polar covalent, DNA base pairing for hydrogen bonds).
Classify each bond as strong or weak in biological contexts.
Explain why the strength of these bonds matters for biological molecules and processes.
Try solving on your own before revealing the answer!
Q4. Define and be able to use the terms: Anabolism, Catabolism, Metabolism
Background
Topic: Metabolic Pathways
This question is about the basic vocabulary of metabolism, including the processes that build up (anabolism) and break down (catabolism) molecules in cells.
Key Terms and Concepts:
Anabolism: Biosynthetic reactions that build complex molecules from simpler ones (require energy).
Catabolism: Degradative reactions that break down complex molecules into simpler ones (release energy).
Metabolism: The sum of all chemical reactions in a cell.
Step-by-Step Guidance
Define each term clearly and concisely.
Provide an example of an anabolic and a catabolic reaction in cells.
Explain how these processes are interconnected in metabolism.
Try solving on your own before revealing the answer!
Q5. List several properties of water that are important to living systems.
Background
Topic: Properties of Water in Biology
This question focuses on the unique chemical and physical properties of water that make it essential for life, such as polarity, cohesion, adhesion, solvent abilities, and temperature regulation.
Key Terms and Concepts:
Polarity: Unequal sharing of electrons, leading to partial charges.
Cohesion: Attraction between water molecules.
Adhesion: Attraction between water and other substances.
Solvent: Ability to dissolve many substances.
High Specific Heat: Resists temperature changes.
Density: Ice is less dense than liquid water.
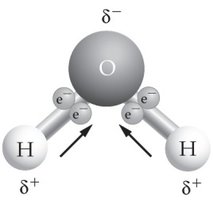
Step-by-Step Guidance
List at least three properties of water that are biologically important (e.g., polarity, solvent properties, cohesion/adhesion).
Explain how each property supports life (e.g., polarity allows water to dissolve ions and molecules, cohesion enables transport in plants).
Relate these properties to the structure of the water molecule (see image above for polarity and hydrogen bonding).
Try solving on your own before revealing the answer!
Q6. Define acid, base, salt, and pH. Describe the buffering effect of water.
Background
Topic: Acids, Bases, Salts, and pH in Biological Systems
This question tests your understanding of basic chemical definitions and the importance of pH and buffering in maintaining homeostasis in cells.
Key Terms and Concepts:
Acid: Substance that donates H+ ions.
Base: Substance that accepts H+ ions or donates OH-.
Salt: Compound formed from the reaction of an acid and a base.
pH: Measure of hydrogen ion concentration.
Buffer: Substance that minimizes changes in pH.
Step-by-Step Guidance
Define each term and provide a biological example (e.g., HCl as an acid, NaOH as a base, NaCl as a salt).
Explain how pH is calculated and why it is important for cells.
Describe how water acts as a buffer to resist changes in pH.
Try solving on your own before revealing the answer!
Q7. Distinguish organic and inorganic compounds. What are biological molecules?
Background
Topic: Organic vs. Inorganic Chemistry in Biology
This question asks you to differentiate between organic and inorganic compounds and to define what makes a molecule 'biological'.
Key Terms and Concepts:
Organic Compounds: Contain carbon-hydrogen bonds (e.g., glucose, proteins).
Inorganic Compounds: Do not contain C-H bonds (e.g., water, salts).
Biological Molecules: Molecules essential for life, usually organic (carbohydrates, lipids, proteins, nucleic acids).
Step-by-Step Guidance
Define organic and inorganic compounds and give examples of each.
Explain what makes a molecule 'biological' and list the four major classes of biological molecules.
Try solving on your own before revealing the answer!
Q8. Define and differentiate functional groups and R-groups.
Background
Topic: Functional Groups in Organic Molecules
This question focuses on the chemical groups that give molecules their specific properties and reactivity, especially in macromolecules.
Key Terms and Concepts:
Functional Group: Specific group of atoms responsible for characteristic reactions of a molecule (e.g., hydroxyl, carboxyl).
R-group: Side chain in amino acids that determines the properties of the amino acid.
Step-by-Step Guidance
Define what a functional group is and list common examples.
Explain what an R-group is in the context of amino acids.
Describe how functional groups and R-groups affect molecular function and diversity.
Try solving on your own before revealing the answer!
Q9. Identify the building blocks of carbohydrates and give examples of molecules.
Background
Topic: Carbohydrate Structure and Function
This question tests your knowledge of the monomers (building blocks) of carbohydrates and examples of simple and complex carbohydrates.
Key Terms and Concepts:
Monosaccharide: Simple sugar (e.g., glucose, fructose).
Disaccharide: Two monosaccharides joined (e.g., sucrose).
Polysaccharide: Many monosaccharides (e.g., starch, cellulose).
Step-by-Step Guidance
Identify the basic building block (monomer) of carbohydrates.
List examples of monosaccharides, disaccharides, and polysaccharides.
Explain the biological roles of these molecules.
Try solving on your own before revealing the answer!
Q10. Identify the building blocks of lipids and give examples of molecules. Differentiate simple lipids, complex lipids, and steroids.
Background
Topic: Lipid Structure and Function
This question covers the types of lipids, their building blocks, and examples of each type.
Key Terms and Concepts:
Fatty Acid: Building block of many lipids.
Glycerol: Combines with fatty acids to form triglycerides.
Simple Lipid: Fats and oils (triglycerides).
Complex Lipid: Contains additional elements (e.g., phospholipids).
Steroid: Four fused carbon rings (e.g., cholesterol).
Step-by-Step Guidance
Identify the building blocks of simple lipids (fatty acids and glycerol).
List examples of simple lipids, complex lipids, and steroids.
Describe the structural differences between these types of lipids.
Try solving on your own before revealing the answer!
Q11. Identify the building blocks and structure of proteins and give examples of molecules.
Background
Topic: Protein Structure and Function
This question tests your understanding of amino acids as the building blocks of proteins and the diversity of protein structure and function.
Key Terms and Concepts:
Amino Acid: Monomer of proteins.
Peptide Bond: Linkage between amino acids.
Primary, Secondary, Tertiary, Quaternary Structure: Levels of protein structure.
Step-by-Step Guidance
Identify the basic building block (amino acid) and the bond that links them (peptide bond).
List examples of proteins (e.g., enzymes, antibodies).
Describe the levels of protein structure and their importance for function.
Try solving on your own before revealing the answer!
Q12. Identify the building blocks of nucleic acids and give examples of molecules.
Background
Topic: Nucleic Acid Structure and Function
This question focuses on the monomers of nucleic acids and examples of DNA and RNA.
Key Terms and Concepts:
Nucleotide: Monomer of nucleic acids.
DNA: Deoxyribonucleic acid.
RNA: Ribonucleic acid.
Step-by-Step Guidance
Identify the building block (nucleotide) and its components (sugar, phosphate, base).
List examples of nucleic acids (DNA, RNA).
Explain the role of nucleic acids in cells.
Try solving on your own before revealing the answer!
Q13. Describe Adenosine Triphosphate (ATP) as a macromolecule and its role in cellular activities.
Background
Topic: ATP and Cellular Energy
This question is about the structure of ATP and its central role as the energy currency of the cell.
Key Terms and Concepts:
ATP: Adenosine triphosphate, a nucleotide derivative.
Energy Currency: ATP stores and transfers energy for cellular processes.
Hydrolysis: Breaking ATP to ADP releases energy.
Step-by-Step Guidance
Describe the structure of ATP (adenine, ribose, three phosphates).
Explain how ATP stores energy in its phosphate bonds.
Discuss how ATP is used in cellular activities (e.g., muscle contraction, active transport).
Try solving on your own before revealing the answer!
