 Back
BackChemical Components of Cells: Structure, Function, and Biological Importance
Study Guide - Smart Notes

Chemical Components of Cells
Overview
This chapter explores the fundamental chemical components of cells, focusing on the types of atoms, chemical bonds, and the properties of water that are essential for life. It also covers the calculation of pH, concentrations, and the structure/function relationships in biological macromolecules.
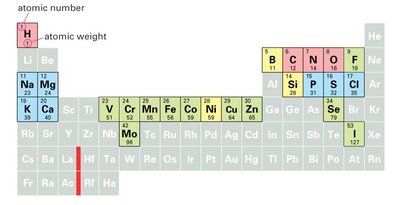
Atoms and Elements in Cells
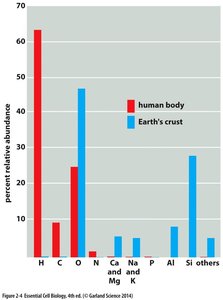
Major Elements in Biological Systems
Cells are composed of a limited set of elements, with a few dominating biological chemistry. These elements are critical for cellular structure and function.
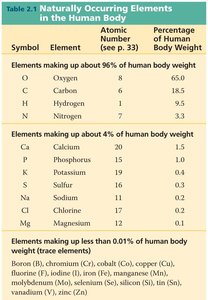
Key Elements: Oxygen, Carbon, Hydrogen, Nitrogen, Calcium, Phosphorus, Potassium, Sulfur, Sodium, Chlorine, Magnesium.
Trace Elements: Boron, Chromium, Cobalt, Copper, Fluorine, Iodine, Iron, Manganese, Molybdenum, Selenium, Silicon, Vanadium, Zinc.
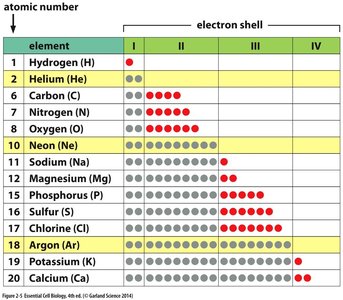
Atomic Structure: Atoms consist of protons (positive charge), neutrons (neutral), and electrons (negative charge).
Atomic Number: Number of protons; determines the element.
Atomic Weight: Sum of protons and neutrons.
Chemical Bonds
Types of Chemical Bonds
Chemical bonds are the forces that hold atoms together in molecules. The nature of these bonds determines the stability and function of biological molecules.
Covalent Bonds: Sharing of electrons between atoms; strongest type of bond in biological systems.
Ionic Bonds: Transfer of electrons from one atom to another, resulting in charged ions.
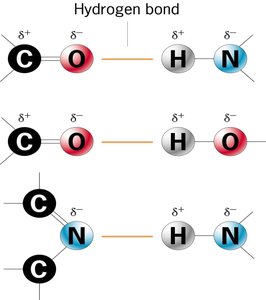
Hydrogen Bonds: Weak electrostatic attraction between a hydrogen atom in a polar bond and another electronegative atom.
Hydrophobic Interactions: Nonpolar molecules aggregate to avoid water.
Van der Waals Attractions: Weak, transient interactions due to temporary dipoles.
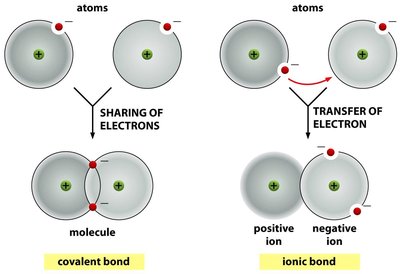
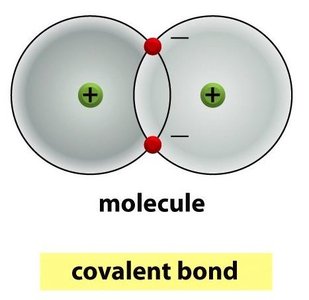
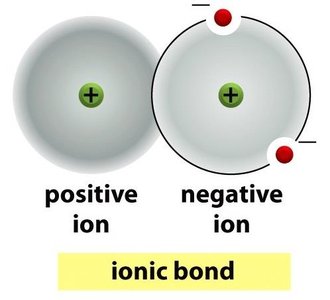
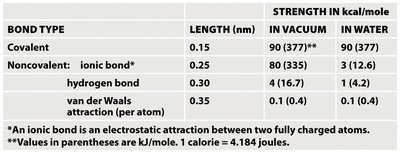
Bond Strengths and Properties
The strength of chemical bonds varies depending on the environment (vacuum vs water) and the type of bond.
Covalent bonds: ~90 kcal/mol in vacuum and water.
Ionic bonds: Strong in vacuum (~80 kcal/mol), much weaker in water (~3 kcal/mol).
Hydrogen bonds: ~4 kcal/mol in vacuum, ~1 kcal/mol in water.
Van der Waals: Very weak (~0.1 kcal/mol).
Properties of Water
Unique Properties of Water
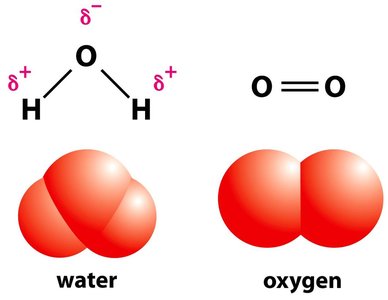
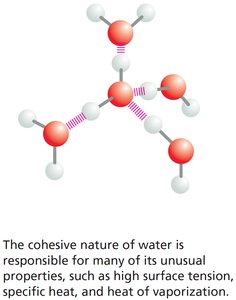
Water is essential for life due to its polar nature and ability to form hydrogen bonds, which give rise to several unique properties.
High specific heat and boiling point
Liquid at room temperature
Lower density as solid (ice floats)
Surface tension, adhesion, and cohesion

Hydration and Solubility
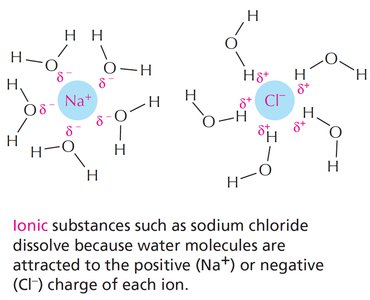
Water's polarity allows it to dissolve ionic and polar substances by forming hydration shells around ions and molecules.
Sphere of Hydration: Water molecules surround ions, partially shielding their charges.
Ionic substances: Dissolve due to attraction between water and ions.
Polar substances: Dissolve by forming hydrogen bonds with water.

Weak Interactions: Van der Waals and Hydrophobic Forces
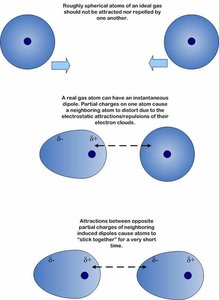
Van der Waals Attractions
Van der Waals forces are weak interactions caused by transient electrical charges. They are significant when many such interactions occur between macromolecular surfaces.
Transient dipoles: Cause attraction between atoms.
Importance: Stabilize protein and nucleic acid structures.
Hydrophobic Interactions
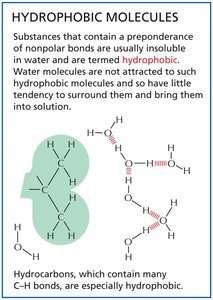
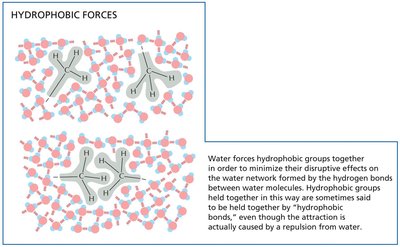
Hydrophobic interactions occur when nonpolar molecules aggregate to minimize their exposure to water, driven by water's tendency to exclude nonpolar substances.
Hydrophobic molecules: Insoluble in water; often hydrocarbons.
Hydrophobic forces: Cause nonpolar groups to cluster together.
Solutions, Concentrations, and pH
The Mole and Molarity
Concentration is a critical concept in cell chemistry. The mole is a standard unit for quantifying atoms and molecules.
Avogadro's number: particles per mole.
Molarity (M): Moles per liter of solution.
Example: 1 M NaCl solution requires 58 g NaCl per liter.
pH and Ionization of Water
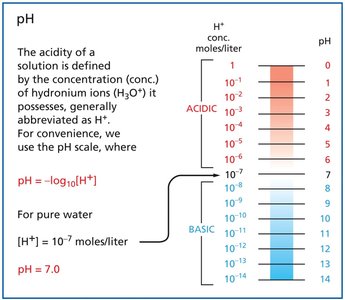
pH measures the concentration of hydrogen ions in solution, which is crucial for biological processes.
Ionization of water:
Equilibrium constant:
pH formula:
Pure water: , pH = 7
Acids: Donate protons (H+)
Bases: Accept protons (H+)
Buffers: Resist changes in pH; consist of weak acid/base and their conjugate forms.


Functional Groups in Biological Molecules
Common Functional Groups
Functional groups are specific groups of atoms within molecules that confer characteristic chemical properties.
Methyl (-CH3): Nonpolar hydrocarbon
Hydroxyl (-OH): Alcohols
Carboxyl (-COOH): Weak acid
Carbonyl (C=O): Ketones and aldehydes
Amino (-NH2): Weak base
Amide (O=C-NH2): Carboxyl + amine
Phosphate (-PO3): Esters and anhydrides
Sulfhydryl (-SH): Forms disulfide bonds
Biological Macromolecules
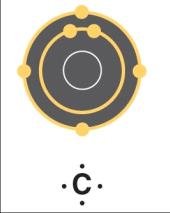
Carbon and Organic Chemistry
Carbon is the backbone of organic molecules due to its valency of four, allowing for diverse structures and isomerism.
Hydrocarbons: Chains and rings of carbon and hydrogen.
Isomers: Structural, geometric, and optical variations.
Major Classes of Biological Molecules
Cells contain four major classes of macromolecules: carbohydrates, lipids, nucleic acids, and proteins.
Carbohydrates: Sugars and polysaccharides; energy storage and structural roles.
Lipids: Fatty acids, triacylglycerols, phospholipids, cholesterol; energy storage, membrane structure, signaling.
Nucleic Acids: DNA and RNA; information storage and transmission.
Proteins: Polymers of amino acids; enzymatic, structural, signaling functions.
Condensation and Hydrolysis Reactions
Macromolecules are assembled by condensation reactions (removal of water) and broken down by hydrolysis (addition of water).
Condensation: Formation of bonds between monomers, releasing water.
Hydrolysis: Breaking bonds by adding water.
Carbohydrates: Structure and Function
Monosaccharides and Polysaccharides
Carbohydrates are composed of monosaccharides (simple sugars) and polysaccharides (complex carbohydrates).
General formula: (CH2O)n
Glucose: Most important hexose; exists in two ring forms.
Polysaccharides: Energy storage (starch, glycogen) and structural (cellulose).
Glycosidic bonds: Link monosaccharides in polysaccharides.
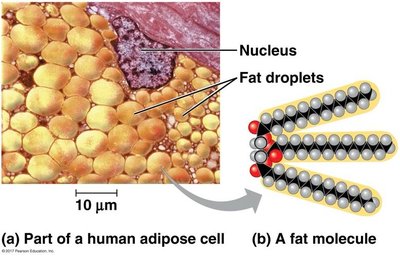
Lipids: Structure and Function
Fatty Acids and Triacylglycerols
Lipids are hydrophobic molecules used for energy storage and membrane structure.
Fatty acids: Long hydrocarbon chains with a carboxyl group.
Triacylglycerols: Fatty acids linked to glycerol by ester bonds.
Phospholipids: Glycerol, two fatty acids, and a phosphate group; major membrane component.
Cholesterol: Four-ring structure; membrane fluidity and precursor to steroid hormones.
Nucleic Acids: Structure and Function
Nucleotides and Nucleic Acid Assembly
Nucleic acids are polymers of nucleotides, essential for genetic information storage and transmission.
Nucleotide: Base + pentose sugar + phosphate.
Phosphodiester linkage: Joins nucleotides in DNA/RNA.
Structural polarity: 5' phosphate and 3' hydroxyl ends.
Complementary base pairing: A-T/U (2 H bonds), G-C (3 H bonds).
DNA vs RNA: DNA is double-stranded, RNA is mostly single-stranded.
Proteins: Structure and Function
Amino Acids and Protein Assembly
Proteins are polymers of amino acids, each with unique side chains (R-groups) that determine their properties and functions.
20 common amino acids: Classified as nonpolar, acidic, basic, or uncharged polar.
Peptide bonds: Amide linkage formed by condensation.
Structural polarity: N-terminus (amino) and C-terminus (carboxyl).
Sequence defines structure and function: Vast diversity due to different combinations.
Summary Table: Bond Types and Strengths
BOND TYPE | LENGTH (nm) | STRENGTH IN VACUUM (kcal/mol) | STRENGTH IN WATER (kcal/mol) |
|---|---|---|---|
Covalent | 0.15 | 90 (377) | 90 (377) |
Ionic (noncovalent) | 0.25 | 80 (335) | 3 (12.6) |
Hydrogen bond | 0.30 | 4 (16.7) | 1 (4.2) |
Van der Waals | 0.35 | 0.1 (0.4) | 0.1 (0.4) |
Key Equations
pH calculation:
Water ionization:
Equilibrium constant:
Conclusion
Understanding the chemical components of cells, including the types of atoms, chemical bonds, and properties of water, is fundamental to microbiology. These principles underpin the structure and function of biological macromolecules and the processes essential for life.
