 Back
BackChemical Principles in Microbiology: Atoms, Bonds, and Biological Molecules
Study Guide - Smart Notes

Ch. 2 - Chemical Principles
The Structure of Atoms
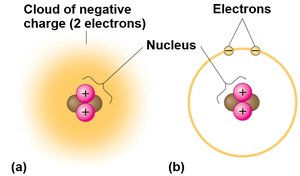
Chemistry is fundamental to understanding microbiology, as it explains how atoms and molecules interact to form the basis of life. Atoms are the smallest units of matter, composed of subatomic particles: protons, neutrons, and electrons. The arrangement and properties of these particles determine the physical and chemical characteristics of elements.
Protons (p+): Positively charged, located in the nucleus, mass ~1 dalton.
Neutrons (n0): No charge, located in the nucleus, mass ~1 dalton.
Electrons (e–): Negatively charged, orbit the nucleus, mass ~1/2000 dalton.
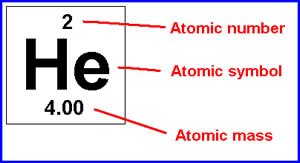
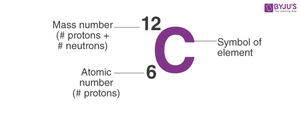
Atomic number: Number of protons in the nucleus; defines the element.
Atomic mass: Total number of protons and neutrons.
Isotopes: Atoms of the same element with different numbers of neutrons.
Chemical Elements and the Elements of Life
Living organisms require a subset of the 92 naturally occurring elements. The most abundant elements in life are carbon, hydrogen, oxygen, and nitrogen, which make up 96% of living matter. Other elements, such as calcium, phosphorus, potassium, and sulfur, are also essential, while trace elements are needed in minute quantities.
Essential elements: Required for life; include C, H, O, N, Ca, P, K, S.
Trace elements: Needed in small amounts for enzyme function and other processes.
Element | Symbol | Atomic Number | Approximate Atomic Mass |
|---|---|---|---|
Hydrogen | H | 1 | 1 |
Carbon | C | 6 | 12 |
Nitrogen | N | 7 | 14 |
Oxygen | O | 8 | 16 |
Sodium | Na | 11 | 23 |
Magnesium | Mg | 12 | 24 |
Phosphorus | P | 15 | 31 |
Element | Symbol | Atomic Number | Approximate Atomic Mass |
|---|---|---|---|
Sulfur | S | 16 | 32 |
Chlorine | Cl | 17 | 35 |
Potassium | K | 19 | 39 |
Calcium | Ca | 20 | 40 |
Iron | Fe | 26 | 56 |
Iodine | I | 53 | 127 |

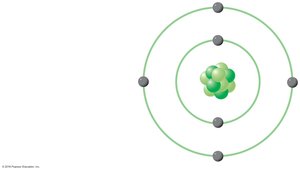
Electronic Configurations and Energy Levels
Electrons are arranged in shells around the nucleus, each corresponding to a specific energy level. Electrons farther from the nucleus possess higher potential energy. The distribution of electrons determines how atoms interact and form bonds.
Energy level: The state of potential energy of an electron based on its location.
Electron shell: The region where electrons are likely to be found.
Valence electrons: Electrons in the outermost shell; crucial for chemical bonding.

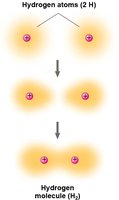
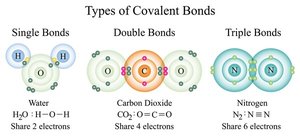
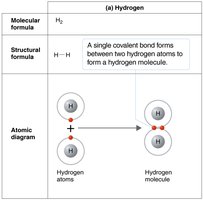
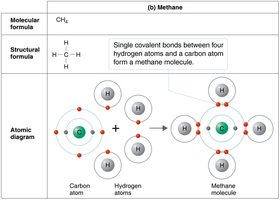
How Atoms Form Molecules: Chemical Bonds
Atoms combine to fill their outermost electron shells, forming molecules through chemical bonds. The type of bond depends on how electrons are distributed between atoms.
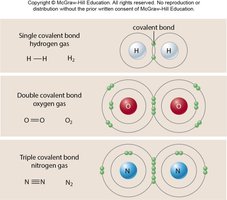
Covalent bonds: Atoms share electrons; strong and common in biological molecules.
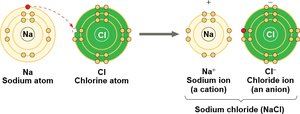
Ionic bonds: Atoms transfer electrons, resulting in oppositely charged ions that attract each other.
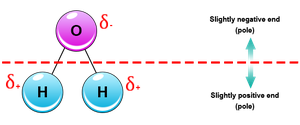
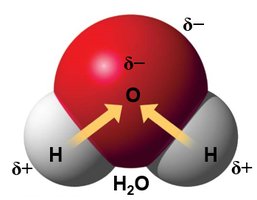
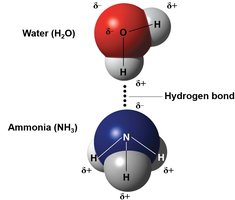
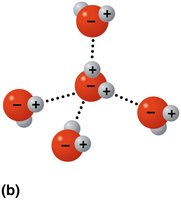
Hydrogen bonds: Weak attractions between a hydrogen atom (partially positive) and an electronegative atom (usually O or N).
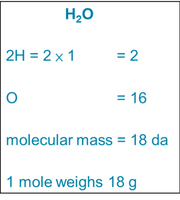
Molecular Mass and Moles
The molecular mass is the sum of the atomic masses of all atoms in a molecule. One mole of a substance is its molecular mass expressed in grams, a fundamental concept for quantifying substances in chemical reactions.
Molecular mass: Sum of atomic masses in a molecule (unit: dalton).
Mole: The molecular mass in grams; used for measuring quantities in reactions.
Chemical Reactions
Chemical reactions involve the making or breaking of bonds between atoms, resulting in changes in chemical energy. Reactions can be classified as synthesis, decomposition, or exchange.
Synthesis reactions: Combine atoms or molecules to form larger molecules (anabolism).
Decomposition reactions: Split molecules into smaller components (catabolism).
Exchange reactions: Involve both synthesis and decomposition.
Reversible reactions: Can proceed in either direction under suitable conditions.
Important Biological Molecules: Inorganic Compounds
Inorganic compounds, such as water, acids, bases, and salts, play crucial roles in biological systems. Water is a polar molecule, an excellent solvent, and acts as a temperature buffer due to hydrogen bonding.
Acids: Dissociate to release hydrogen ions (H+).
Bases: Dissociate to release hydroxide ions (OH–).
Salts: Dissociate into cations and anions, neither of which is H+ or OH–.
pH scale: Measures the concentration of H+ ions; most organisms thrive between pH 6.5 and 8.5.
Organic Compounds and Functional Groups
Organic compounds contain carbon and are fundamental to life. The carbon skeleton forms the backbone, while functional groups confer specific chemical properties. Macromolecules are polymers made of monomers joined by dehydration synthesis.
Functional group: Specific group of atoms attached to the carbon skeleton, responsible for chemical properties.
Macromolecule: Large molecule formed by joining monomers.
Dehydration synthesis: Reaction that joins monomers by removing water.
Carbohydrates
Carbohydrates serve as energy sources and structural components. They include monosaccharides (simple sugars), disaccharides, and polysaccharides.
Monosaccharides: Simple sugars (e.g., glucose, fructose, galactose).
Disaccharides: Formed by joining two monosaccharides (e.g., sucrose, lactose, maltose).
Polysaccharides: Long chains of monosaccharides (e.g., starch, glycogen, cellulose, chitin).
Lipids
Lipids are hydrophobic molecules essential for cell membranes and energy storage. They include fats, phospholipids, and steroids.
Simple lipids: Fats or triglycerides; contain glycerol and fatty acids.
Complex lipids: Include phospholipids with polar and nonpolar regions.
Saturated fats: No double bonds; solid at room temperature.
Unsaturated fats: One or more double bonds; liquid at room temperature.
Steroids: Four carbon rings; important for membrane fluidity.
Proteins
Proteins are polymers of amino acids, essential for structure and function in cells. They have four levels of structure: primary, secondary, tertiary, and quaternary.
Amino acids: Monomers of proteins; contain amino and carboxyl groups.
Peptide bond: Covalent bond joining amino acids.
Primary structure: Sequence of amino acids.
Secondary structure: Coils and folds (α helix, β sheet) due to hydrogen bonding.
Tertiary structure: 3D shape from R group interactions.
Quaternary structure: Multiple polypeptide chains.
Denaturation: Loss of structure and function due to environmental changes.
Nucleic Acids
Nucleic acids (DNA and RNA) store and transmit genetic information. They are polymers of nucleotides, each consisting of a pentose sugar, phosphate group, and nitrogenous base.
DNA: Double helix; contains deoxyribose; bases: A, T, C, G.
RNA: Single-stranded; contains ribose; bases: A, U, C, G.
Adenosine Triphosphate (ATP)
ATP is the primary energy carrier in cells. It consists of ribose, adenine, and three phosphate groups. Hydrolysis of ATP releases energy for cellular activities.
ATP hydrolysis:
Additional info:
Isomers are molecules with the same chemical formula but different structures.
Enzymes are proteins that catalyze biochemical reactions.
Conjugated proteins contain both protein and non-protein components (e.g., glycoproteins, nucleoproteins, lipoproteins).
