 Back
BackChemical Principles in Microbiology: Atoms, Bonds, Reactions, and Biological Molecules
Study Guide - Smart Notes

Chemical Principles in Microbiology
Introduction
Chemistry is fundamental to understanding microbiology because the structure and function of microorganisms are determined by chemical interactions at the molecular level. Many pathogenic effects of infectious diseases occur due to molecular changes, making it essential to grasp basic chemical principles to understand infection processes and cellular functions.
The Structure of Atoms and Elements
Subatomic Particles and Atomic Structure
Atoms are the smallest units of elements, retaining the properties of those elements. Each atom consists of subatomic particles:
Protons: Positively charged, located in the nucleus
Neutrons: Neutral, located in the nucleus
Electrons: Negatively charged, orbiting the nucleus in electron shells
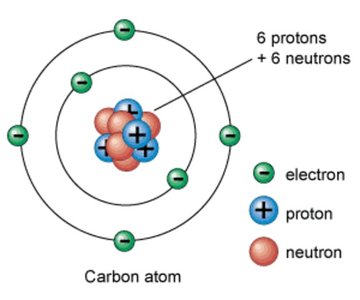
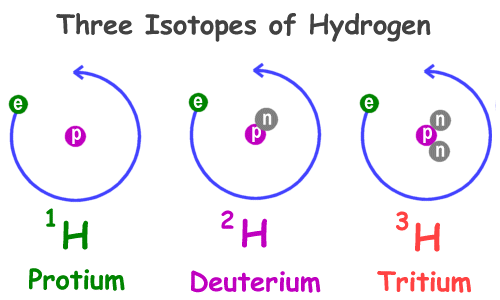
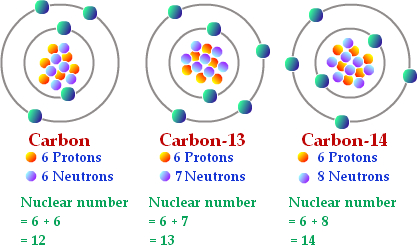
The atomic number is the number of protons in the nucleus, while the atomic weight is the sum of protons and neutrons. Atoms of the same element have the same number of protons but may have different numbers of neutrons, forming isotopes.
Electron Configuration and Valence
Electrons occupy energy levels or shells around the nucleus. The outermost shell, or valence shell, determines the chemical properties and reactivity of the atom. Atoms are most stable when their valence shell is full.
Chemical Bonds
Types of Chemical Bonds
Atoms form molecules by gaining, losing, or sharing electrons to achieve stable electron configurations. The main types of chemical bonds are:
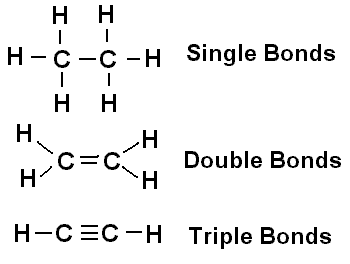
Covalent Bonds: Atoms share pairs of electrons. These are strong bonds and form molecules such as H2 and O2.
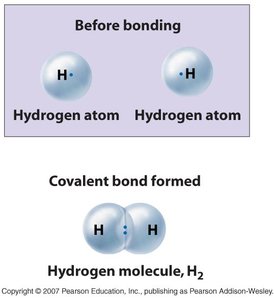
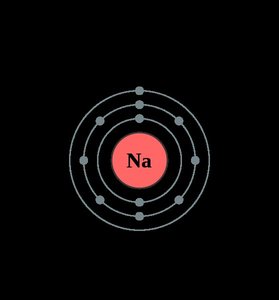
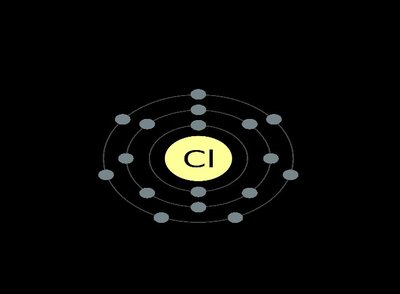
Ionic Bonds: Atoms transfer electrons, resulting in positively charged cations and negatively charged anions that attract each other.

Hydrogen Bonds: Weak attractions between a hydrogen atom covalently bonded to an electronegative atom (like O or N) and another electronegative atom. These are crucial in stabilizing the structures of DNA and proteins.
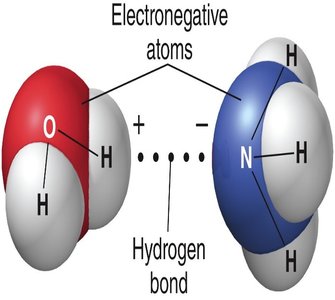
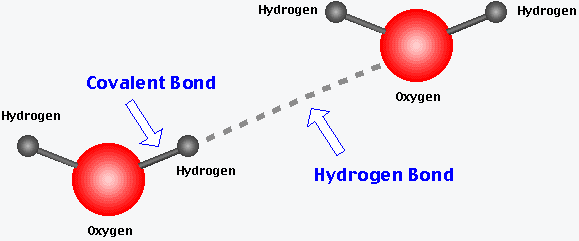
Chemical Reactions
Types of Chemical Reactions
Chemical reactions involve the making or breaking of bonds, resulting in new substances with different properties. The main types are:
Synthesis Reactions (Anabolism): Atoms or molecules combine to form larger molecules. Example: formation of proteins from amino acids.
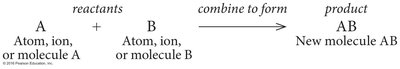
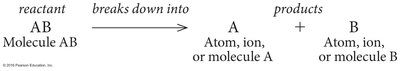
Decomposition Reactions (Catabolism): Molecules are broken down into smaller units. Example: breakdown of sucrose into glucose and fructose.
Exchange Reactions: Involve both synthesis and decomposition. Example: NaOH + HCl → NaCl + H2O.

Reversible Reactions: Products can revert to original reactants under certain conditions.
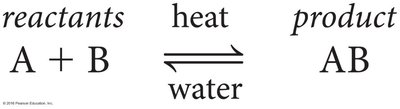
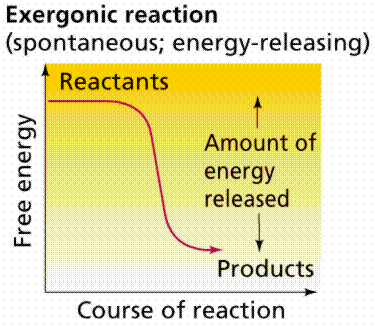
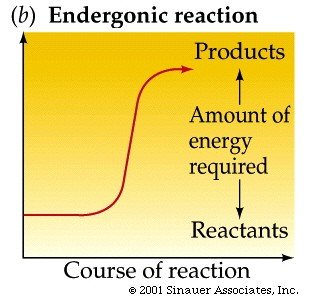
Energy in Chemical Reactions
Reactions may absorb (endergonic) or release (exergonic) energy. Activation energy is required to initiate reactions.
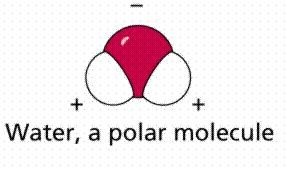
Water and Its Properties
Importance of Water
Water is the most abundant inorganic molecule in living organisms, making up 65-75% of cell mass. Its unique properties are essential for life:
Polarity: Water is a polar molecule, making it an excellent solvent and temperature buffer.
Hydrogen Bonding: Enables water molecules to form networks, contributing to its high heat capacity and solvent abilities.
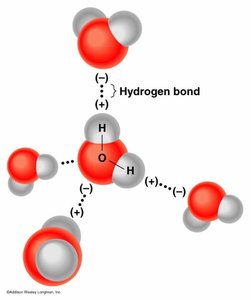
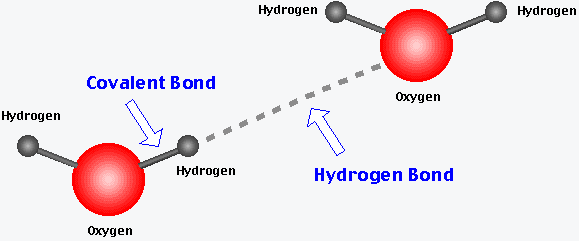
Acids, Bases, Salts, and pH
Definitions and Biological Importance
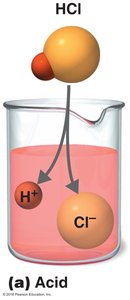
Acids: Substances that dissociate into H+ ions and anions. Example: HCl → H+ + Cl-
Bases: Substances that dissociate into OH- ions and cations. Example: NaOH → Na+ + OH-
Salts: Substances that dissociate into cations and anions, neither of which is H+ or OH-. Example: NaCl → Na+ + Cl-

pH Scale
The pH scale measures the concentration of hydrogen ions in a solution:
pH = –log10[H+]
Most organisms grow best at pH 6.5–8.5
Enzyme activity is highly sensitive to pH changes
Organic Compounds and Biological Macromolecules
Organic vs. Inorganic Compounds
Organic Compounds: Always contain carbon and hydrogen, often complex and large (e.g., carbohydrates, lipids, proteins, nucleic acids).
Inorganic Compounds: Typically lack carbon, usually small and simple (e.g., water, salts, acids, bases).
Functional Groups
Functional groups are specific groups of atoms within molecules that confer characteristic chemical properties. Examples include hydroxyl (–OH), carboxyl (–COOH), amino (–NH2), and phosphate (–PO43-).
Carbohydrates
Carbohydrates serve as energy sources and structural components. They are classified as:
Monosaccharides: Simple sugars (e.g., glucose, fructose)
Disaccharides: Two monosaccharides joined by dehydration synthesis (e.g., sucrose)
Polysaccharides: Long chains of monosaccharides (e.g., starch, cellulose)
Lipids
Lipids are hydrophobic molecules important for cell membranes and energy storage. Types include:
Simple Lipids (Fats/Triglycerides): Glycerol + 3 fatty acids
Complex Lipids (Phospholipids): Glycerol, 2 fatty acids, and a phosphate group
Steroids: Four carbon rings with functional groups (e.g., cholesterol)
Proteins
Proteins are polymers of amino acids and perform diverse functions such as catalysis (enzymes), transport, and structural support. Protein structure is organized into four levels:
Primary: Amino acid sequence
Secondary: Alpha helices and beta sheets
Tertiary: 3D folding
Quaternary: Association of multiple polypeptides
Nucleic Acids
Nucleic acids (DNA and RNA) store and transmit genetic information. They are composed of nucleotides (sugar, phosphate, nitrogenous base).
DNA: Double helix, deoxyribose sugar, bases A-T and G-C
RNA: Single-stranded, ribose sugar, bases A-U and G-C
Adenosine Triphosphate (ATP)
ATP is the primary energy carrier in cells. It stores energy in high-energy phosphate bonds and releases it upon hydrolysis to power cellular activities.
Case Study Application: Microbial Growth and Nutrient Utilization
Example: Oral Thrush in Infants
The overgrowth of Candida albicans in the mouth (thrush) is linked to the availability of organic nutrients such as carbohydrates and proteins, which serve as energy and building blocks for microbial growth. The balance of the oral microbiome and the presence of nutrients directly affect the proliferation of pathogenic microbes.
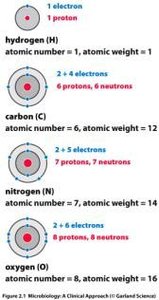
Summary Table: Subatomic Particles
Particle | Charge | Mass (amu) | Location |
|---|---|---|---|
Proton | +1 | 1 | Nucleus |
Neutron | 0 | 1 | Nucleus |
Electron | -1 | ~1/1800 | Electron Shells |
Key Equations
pH calculation:
Check Your Understanding
Distinguish between organic and inorganic compounds.
Define and identify functional groups.
Identify the building blocks of carbohydrates, lipids, proteins, and nucleic acids.
Differentiate between DNA and RNA.
Explain the role of ATP in cellular activities.
