 Back
BackChemical Principles in Microbiology: Structure, Bonds, and Biological Molecules
Study Guide - Smart Notes

Chemical Principles of Microbiology
Introduction to Chemistry in Microbiology
Chemistry forms the foundation for understanding the structure and function of microbial cells. Atoms, molecules, and their interactions underpin all biological processes, including metabolism, genetic information transfer, and cellular structure.
The Structure of Atoms
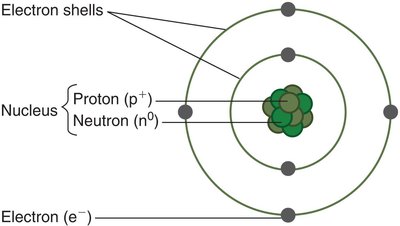
Atomic Structure and Subatomic Particles
Atom: The smallest unit of matter that retains the properties of an element.
Subatomic particles: Atoms are composed of protons (positively charged), neutrons (neutral), and electrons (negatively charged).
Protons and neutrons form the nucleus, while electrons move in regions called electron shells around the nucleus.
Chemical Elements and Isotopes
Chemical element: Defined by the number of protons in the nucleus (atomic number).
Atomic mass: The sum of protons and neutrons.
Isotopes: Atoms of the same element with different numbers of neutrons (e.g., 16O, 17O, 18O).
Major Elements in Living Organisms
The most abundant elements in living organisms are hydrogen, carbon, nitrogen, and oxygen. Other important elements include sodium, magnesium, phosphorus, sulfur, chlorine, potassium, calcium, iron, and iodine.
Element | Symbol | Atomic Number | Approximate Atomic Mass |
|---|---|---|---|
Hydrogen | H | 1 | 1 |
Carbon | C | 6 | 12 |
Nitrogen | N | 7 | 14 |
Oxygen | O | 8 | 16 |
Sodium | Na | 11 | 23 |
Magnesium | Mg | 12 | 24 |
Phosphorus | P | 15 | 31 |
Sulfur | S | 16 | 32 |
Chlorine | Cl | 17 | 35 |
Potassium | K | 19 | 39 |
Calcium | Ca | 20 | 40 |
Iron | Fe | 26 | 56 |
Iodine | I | 53 | 127 |
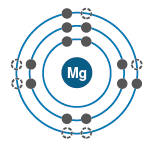
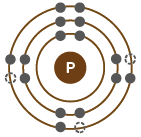
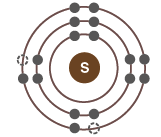
Electronic Configurations and Chemical Behavior
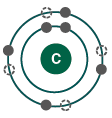
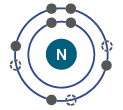
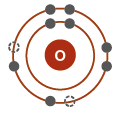
Electron Shells and Valence
Electrons are arranged in shells around the nucleus, each with a characteristic energy level.
The valence shell (outermost shell) determines an atom's chemical reactivity.
Atoms are most stable when their valence shell is full.
Chemical Bonds
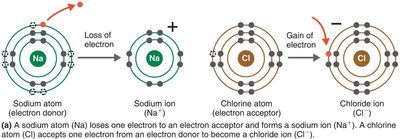
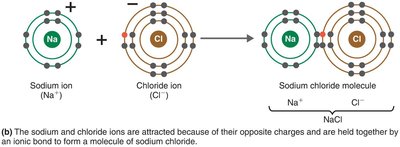
Ionic Bonds
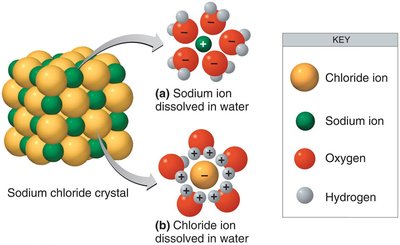
Ionic bonds are formed when one atom donates electrons to another, resulting in the formation of oppositely charged ions that attract each other.
Cation: Positively charged ion (e.g., Na+).
Anion: Negatively charged ion (e.g., Cl-).
Covalent Bonds
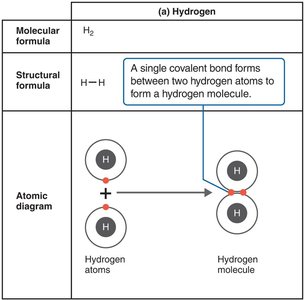
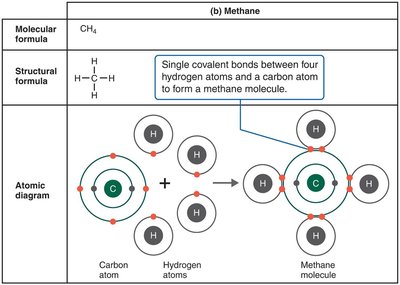
Covalent bonds involve the sharing of one or more pairs of electrons between atoms. These are the strongest and most common bonds in biological molecules.
Single covalent bond: One pair of electrons shared (e.g., H2).
Double covalent bond: Two pairs of electrons shared (e.g., O2).
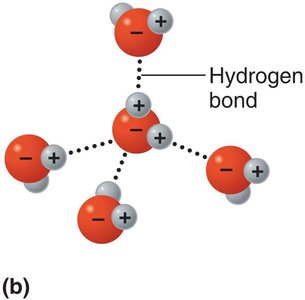
Hydrogen Bonds
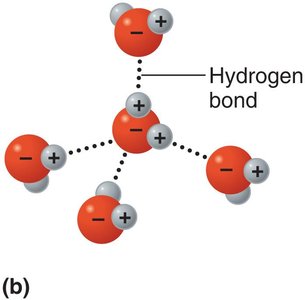
Hydrogen bonds are weak attractions between a hydrogen atom covalently bonded to an electronegative atom (O or N) and another electronegative atom. They are crucial for stabilizing the structures of proteins and nucleic acids.
Hydrogen bonds do not form molecules but serve as bridges between molecules or within large molecules.
Comparison of Bond Types
Type of Bond | Definition and Importance |
|---|---|
Ionic | Attraction between ions of opposite charge; important in biochemical reactions. |
Covalent | Atoms share electrons; most common in organisms, forming stable molecules. |
Hydrogen | Weak attraction involving hydrogen; stabilizes large molecules like proteins and DNA. |
Chemical Reactions in Microbiology
Types of Chemical Reactions
Synthesis reactions: Atoms or molecules combine to form larger molecules (anabolism).
Decomposition reactions: Molecules are broken down into smaller components (catabolism).
Exchange reactions: Involve both synthesis and decomposition; atoms are rearranged between molecules.
Reversible reactions: Can proceed in both directions under different conditions.
Activation Energy and Energy Changes
Activation energy: The energy required to initiate a chemical reaction.
Endergonic reactions: Absorb energy.
Exergonic reactions: Release energy.
Water and Its Importance
Properties of Water
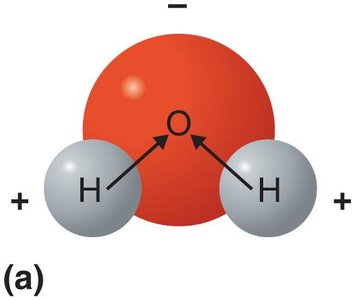
Water is an inorganic, polar molecule essential for life. Its polarity and ability to form hydrogen bonds give it unique properties:
Excellent temperature buffer
Ice is less dense than liquid water
Excellent solvent for polar and ionic substances
Participates in many chemical reactions
Acids, Bases, and Salts
Definitions and Examples
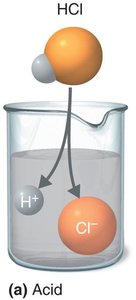
Acids: Substances that dissociate into hydrogen ions (H+) and anions; proton donors.
Bases: Substances that dissociate into hydroxide ions (OH-) and cations; proton acceptors.
Salts: Substances that dissociate into cations and anions, neither of which is H+ or OH-.
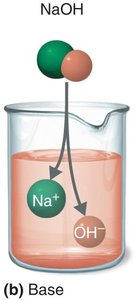
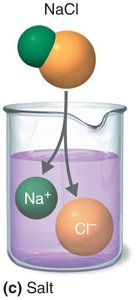
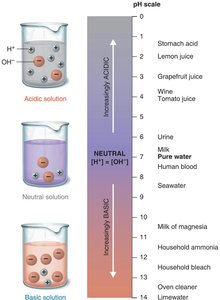
The Concept of pH
pH measures the concentration of hydrogen ions in a solution.
pH is calculated as
pH scale ranges from 0 (acidic) to 14 (basic), with 7 being neutral.
Organisms require a stable pH for optimal biochemical function; buffers help maintain pH balance.
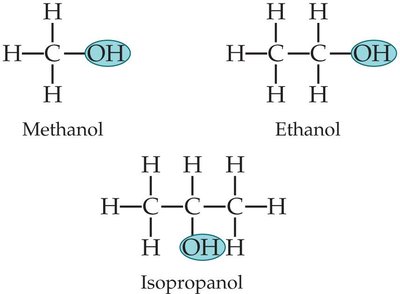
Organic Molecules and Functional Groups
Structure and Functional Groups
Organic molecules contain carbon skeletons with various functional groups attached, determining their chemical properties.
Common functional groups include hydroxyl, carboxyl, amino, methyl, phosphate, and others.
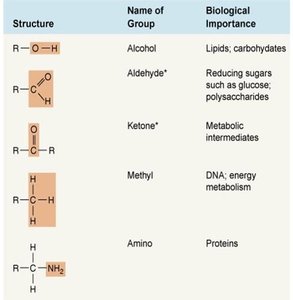
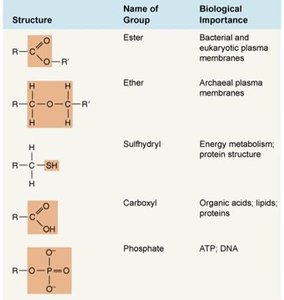
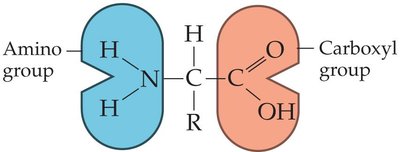
Amino Acids and Macromolecules
Amino acids contain an amino group, carboxyl group, and a variable side chain (R group).
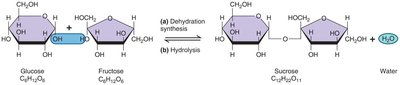
Macromolecules are polymers formed by joining monomers through dehydration synthesis.
Carbohydrates
Types and Functions
Carbohydrates serve as energy sources and structural components.
Monosaccharides: Simple sugars (e.g., glucose, fructose, deoxyribose).
Disaccharides: Two monosaccharides joined by dehydration synthesis (e.g., maltose, sucrose, lactose).
Polysaccharides: Long chains of monosaccharides (e.g., starch, glycogen, cellulose).
Lipids
Simple and Complex Lipids
Lipids are nonpolar, hydrophobic molecules composed of C, H, and O.
Simple lipids (fats/triglycerides): Glycerol + fatty acids, formed by dehydration synthesis.
Saturated fats: No double bonds in fatty acids.
Unsaturated fats: One or more double bonds (cis or trans configuration).
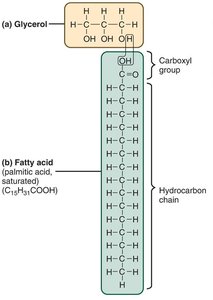
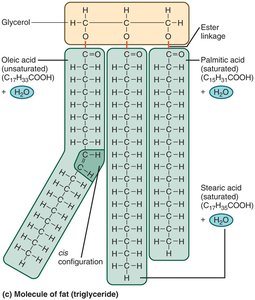
Complex Lipids and Steroids
Phospholipids: Contain glycerol, two fatty acids, and a phosphate group; major component of cell membranes.
Steroids: Four carbon rings with an -OH group; maintain membrane fluidity (e.g., cholesterol in animals, ergosterol in fungi).
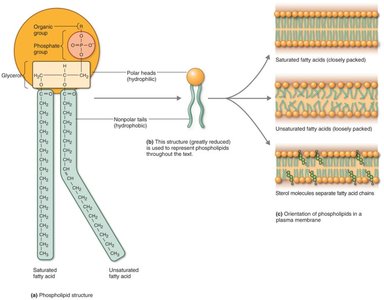
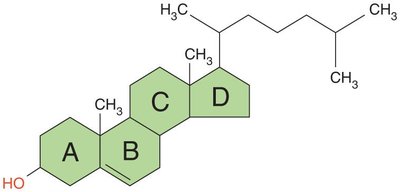
Proteins
Structure and Function
Proteins are polymers of amino acids (contain C, H, O, N, sometimes S).
Functions include enzymes, transport, movement, toxins, and structural roles.
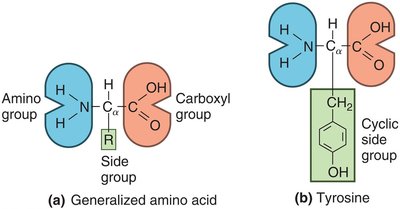
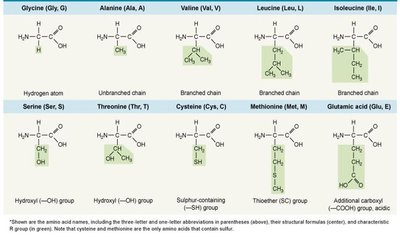
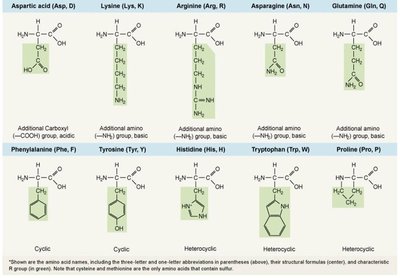
Peptide Bonds and Protein Structure
Peptide bonds: Link amino acids via dehydration synthesis.
Primary structure: Linear sequence of amino acids.
Secondary structure: Alpha helices and beta sheets stabilized by hydrogen bonds.
Tertiary structure: 3D folding stabilized by disulfide bridges, hydrogen bonds, ionic bonds, and hydrophobic interactions.
Quaternary structure: Association of multiple polypeptide chains.
Denaturation: Loss of structure and function due to environmental changes.
Conjugated proteins: Contain amino acids plus other organic molecules (e.g., glycoproteins, lipoproteins).
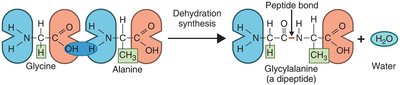
Nucleic Acids
DNA and RNA
Nucleic acids: Store and transmit genetic information (DNA and RNA).
Nucleotides: Building blocks composed of a pentose sugar, phosphate group, and nitrogenous base.
DNA: Double helix, deoxyribose sugar, bases A-T and C-G.
RNA: Single-stranded, ribose sugar, bases A-U and C-G; includes mRNA, rRNA, tRNA.
Adenosine Triphosphate (ATP)
Structure and Function
ATP is the primary energy carrier in cells.
Composed of ribose, adenine, and three phosphate groups.
Hydrolysis of ATP releases energy for cellular processes.
Additional info: This summary covers the chemical principles essential for understanding microbial structure and function, including atomic structure, bonding, water, acids and bases, organic molecules, and the macromolecules critical to life.
