 Back
BackChemical Principles in Microbiology: Structured Study Notes
Study Guide - Smart Notes

Chemical Principles
Atoms: The Smallest Unit of Matter
Atoms are the fundamental building blocks of matter, forming the basis of all chemical elements and compounds. Understanding atomic structure is essential for comprehending biological molecules and their interactions in microbiology.
Matter: Anything that occupies space and has mass (e.g., organisms, rocks, water).
Chemical Elements: Pure substances made of only one type of atom.
Atom: The smallest unit of an element, and thus the smallest unit of matter.
Atoms make up both living and non-living matter.
Example: Atoms are the smallest units of matter, such as carbon atoms in a diamond or glucose molecule.

Atomic Structure
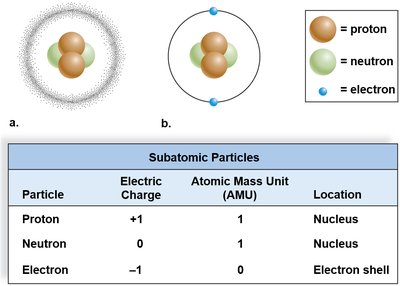
Atoms consist of three main subatomic particles, each with distinct properties and locations within the atom.
Protons: Positively charged, located in the nucleus, mass = 1 AMU.
Neutrons: No charge, located in the nucleus, mass = 1 AMU.
Electrons: Negatively charged, orbiting the nucleus, mass ≈ 0 AMU.
Example: Electrons are negatively charged particles with almost no mass.
Subatomic Particle | Electric Charge | Atomic Mass Unit (AMU) | Location |
|---|---|---|---|
Proton | +1 | 1 | Nucleus |
Neutron | 0 | 1 | Nucleus |
Electron | -1 | 0 | Electron shell |
Elements of Life
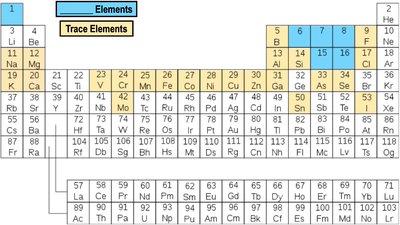
Only a small subset of elements are found in living organisms, and these are essential for life. The periodic table organizes these elements based on their properties.
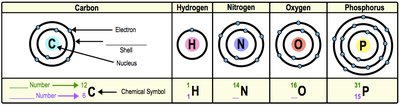
About 97% of the mass of most life is composed of Carbon, Hydrogen, Nitrogen, Oxygen, Phosphorus, and Sulfur (CHNOPS).
Bulk Elements: Required in large amounts.
Trace Elements: Required in small amounts.
Atomic Properties
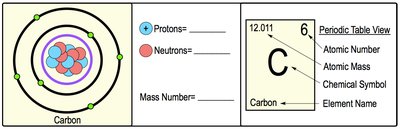
Each atom has unique properties that distinguish it from others, including atomic number, mass number, and atomic mass.
Atomic Number: Number of protons in the nucleus; defines the element.
Mass Number: Sum of protons and neutrons in the nucleus.
Atomic Mass: Weighted average mass of all isotopes of an element.
Example: Carbon atom has 6 protons (atomic number 6), mass number varies with isotope.

Electron Orbitals & Energy Shells
Electrons occupy specific regions around the nucleus called orbitals or energy shells. The arrangement of electrons determines chemical reactivity.
Electron Orbitals: 3D regions where electrons are likely found.
Energy Shells: 2D representations; shells closer to the nucleus are lower in energy.
Valence Electrons: Electrons in the outermost shell; critical for chemical bonding.
1st shell holds up to 2 electrons; 2nd shell up to 8 electrons.
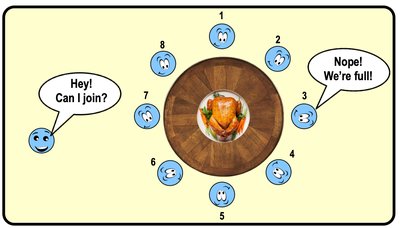
Octet Rule
The octet rule states that atoms are most stable when their valence shell is fully occupied, typically with eight electrons.
Atoms are less reactive when their outer shells are full.
1st shell: 2 electrons; 2nd shell: 8 electrons.
Example: Neon is unreactive because its valence shell is full.
Isotopes
Isotopes are atoms of the same element with different numbers of neutrons, resulting in different mass numbers.
All isotopes have the same atomic number (protons) but different mass numbers.
Example: Carbon-12, Carbon-13, and Carbon-14 are isotopes of carbon.
Introduction to Chemical Bonding
Chemical bonds are attractive forces that hold atoms together, forming molecules and compounds. Understanding these bonds is crucial for studying biological macromolecules.
Molecule: Substance containing two or more chemically bound atoms (e.g., O2).
Compound: Molecule composed of two or more different elements (e.g., H2O).
Chemical Formula: Shows the types and numbers of atoms in a molecule (e.g., C6H12O6).
Intramolecular vs. Intermolecular Bonds
Bonds can occur within a molecule (intramolecular) or between molecules (intermolecular).
Intramolecular Bonds: Interactions within a single molecule.
Intermolecular Bonds: Interactions between different molecules.
Covalent Bonds
Covalent bonds involve the sharing of electrons between atoms. They are fundamental to the structure of biological molecules.
Nonpolar Covalent Bonds: Equal sharing of electrons (similar electronegativities).
Polar Covalent Bonds: Unequal sharing of electrons (different electronegativities), resulting in partial charges.
Electronegativity: Measure of an atom's attraction for electrons (scale 0-4).
Example: Water (H2O) has polar covalent bonds between hydrogen and oxygen.
Noncovalent Bonds
Noncovalent bonds result from full or partial charges and do not involve electron sharing. They are important for molecular interactions in biology.
Ionic Bonds: Electrical attractions between oppositely charged ions.
Hydrogen Bonds: Weak interactions between a highly electronegative atom (F, O, N) and a hydrogen atom.
Van der Waals Bonds: Very weak, transient interactions.
Ions: Anions vs. Cations
Ions are atoms or molecules with a net electrical charge due to the gain or loss of electrons.
Anions: Negatively charged ions (gain of electrons).
Cations: Positively charged ions (loss of electrons).
Ionic Bonding
Ionic bonds form when electrons are transferred between atoms, resulting in oppositely charged ions that attract each other.
Transfer of electrons fills valence shells and creates charges.
Example: Sodium chloride (NaCl) forms via ionic bonding between Na+ and Cl-.
Hydrogen Bonding
Hydrogen bonds are weak individually but strong collectively, playing a crucial role in the properties of water and the structure of biological macromolecules.
Form between a highly electronegative atom and hydrogen.
Important for water's properties and macromolecular structure.
Introduction to Water
Water is a small, polar molecule with unique properties due to hydrogen bonding. It is essential for life and acts as a universal solvent.
Water molecules have partial negative and positive charges.
Hydrogen bonds form between water molecules.
Emergent Properties of Water
Water's hydrogen bonding gives rise to several emergent properties critical for life.
Cohesion & Adhesion: Water molecules stick to each other and to other substances.
Moderation of Temperature: Water resists temperature changes.
Density: Ice is less dense than liquid water, allowing it to float.
Universal Solvent: Water dissolves many substances.
Properties of Water: Cohesion & Adhesion
Cohesion allows water molecules to stick together, while adhesion allows them to stick to other substances. Surface tension is a result of these properties.
Cohesion: Water molecules stick to each other via hydrogen bonds.
Adhesion: Water molecules stick to other polar substances.
Surface Tension: Difficulty in breaking the surface of water.
Properties of Water: Density
Liquid water is more densely packed than solid ice, which forms a lattice structure due to stable hydrogen bonds. This allows ice to float and insulates aquatic life.
Ice is less dense than liquid water.
Allows water to freeze from the top, sustaining life below.
Properties of Water: Thermal
Water has high specific heat and high heat of vaporization, allowing it to moderate temperature and resist rapid changes.
Specific Heat: Amount of heat required to raise/lower 1 gram of water by 1°C.
Heat of Vaporization: Amount of heat required to convert 1 gram of water to vapor.
Properties of Water: Universal Solvent
Water dissolves many substances due to its polarity, forming aqueous solutions essential for biological processes.
Solvent: Substance that dissolves other substances (usually water).
Solute: Substance dissolved by the solvent.
Solution: Homogeneous mixture of solute and solvent.
Water forms a hydration shell around solute molecules.
Acids and Bases
Acids and bases affect the concentration of hydrogen ions (H+) in solution, influencing biological processes.
Acid: Increases H+ concentration in solution.
Base: Decreases H+ concentration in solution (often by increasing OH-).
pH Scale
The pH scale measures the concentration of hydrogen ions in solution, determining whether it is acidic or basic.
pH ranges from 0 (acidic) to 14 (basic); 7 is neutral.
pH indirectly measures [OH-] as well.
Buffers
Buffers are substances that resist changes in pH when acids or bases are added, helping organisms maintain homeostasis.
Can donate or accept H+ as needed.
Example: Bicarbonate buffer system in blood.
Carbon
Carbon is the most abundant element in living systems (excluding water) and forms the backbone of organic molecules.
Organic Molecules: Contain covalently linked carbon and hydrogen atoms.
Hydrocarbons: Organic molecules made of only carbon and hydrogen.
Carbon can form four bonds, allowing for diverse molecular structures.
Functional Groups
Functional groups are reactive groups of atoms commonly found in biological molecules, extending from the carbon backbone.
Examples include hydroxyl (-OH), carboxyl (-COOH), amino (-NH2), and phosphate (-PO4).
Introduction to Biomolecules
Biomolecules are organic molecules essential to living organisms, classified into four primary groups.
Carbohydrates
Proteins
Nucleic Acids
Lipids
Monomers & Polymers
Monomers are individual building blocks that can be linked to form polymers. Most biomolecules are polymers except lipids.
Dehydration Synthesis: Forms covalent bonds to link monomers and build polymers.
Hydrolysis: Breaks covalent bonds to break down polymers.
Carbohydrates
Carbohydrates are carbon-based molecules hydrated with hydroxyl groups. They are classified by size and function.
Monosaccharide: Single carbohydrate unit (e.g., glucose).
Disaccharide: Two covalently linked monosaccharides.
Polysaccharide: More than 20 covalently linked monosaccharides.
Functions: Structural support (cellulose, chitin) and energy storage (starch in plants, glycogen in animals).
Proteins
Proteins are polymers made of amino acid monomers, linked by peptide bonds. They have a hierarchy of structure and diverse functions.
Amino Acids: Building blocks of proteins; 20 types used in living organisms.
Peptide Bonds: Covalent bonds linking amino acids.
Protein Structure: Primary, secondary, tertiary, and quaternary levels.
Denatured Proteins: Non-functional proteins with altered shape due to environmental changes.
Chaperone Proteins: Assist in protein folding or re-naturing.
Nucleic Acids
Nucleic acids store and encode genetic information. They are polymers of nucleotide monomers.
Nucleotides: Consist of a phosphate group, a pentose sugar, and a nitrogenous base.
DNA: Double helix, stores genetic information.
RNA: Single-stranded, involved in protein synthesis.
Phosphodiester Bonds: Covalent bonds linking nucleotides.
Lipids
Lipids are hydrophobic biomolecules, highly diverse in structure and function. They do not form polymers.
Fatty Acids: Hydrocarbon chains with a carboxylic acid group.
Saturated Fatty Acids: Only single bonds, solid at room temperature.
Unsaturated Fatty Acids: One or more double bonds, liquid at room temperature.
Triglycerides: Three fatty acids linked to glycerol.
Phospholipids: Major component of cell membranes, amphipathic.
Steroids: Four fused carbon rings (e.g., cholesterol).
Waxes: Fatty acids bound to long-chain alcohols, used for protection.
