 Back
BackChemistry for Microbiology: Essential Principles and Biological Molecules
Study Guide - Smart Notes

Chemistry for Microbiology
The Structure of Atoms
Chemistry is foundational to microbiology, as it explains the interactions and composition of matter at the atomic and molecular levels. Matter is anything that occupies space and has mass, and its fundamental unit is the atom.
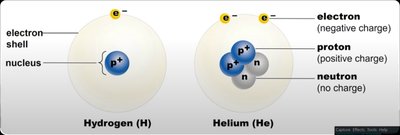
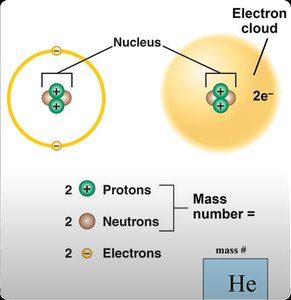
Atom: The smallest unit of matter, composed of a nucleus (protons and neutrons) and electron shells.
Proton: Positively charged particle in the nucleus.
Neutron: Neutral particle in the nucleus.
Electron: Negatively charged particle orbiting the nucleus.
Atomic number: Number of protons in the nucleus, unique to each element.
Mass number: Sum of protons and neutrons in the nucleus.
The Elements and Isotopes
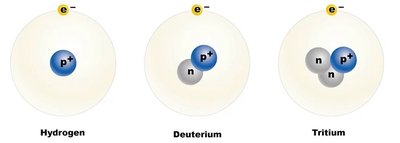
An element is a substance that cannot be broken down by chemical means. Each element is defined by its atomic number. Some elements have isotopes, which are atoms with the same number of protons but different numbers of neutrons.
Isotope: Variant of an element with a different number of neutrons; some are radioactive (e.g., 14C).
Essential elements: Life requires about 25 elements, including trace elements like iron and iodine.
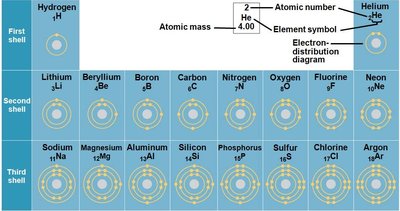

Electron Shells and Chemical Bonding
Electrons are arranged in shells around the nucleus. The arrangement of electrons determines the chemical properties and reactivity of an atom. Atoms seek stability by filling their outermost shell, often through chemical bonding.
Valence shell: The outermost electron shell.
Valence electrons: Electrons in the outer shell, involved in chemical bonding.
Chemical bonds: Attractions between atoms to achieve stability; types include covalent, ionic, and hydrogen bonds.
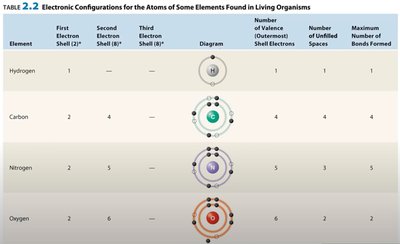
Chemical Bonds
Covalent Bonds
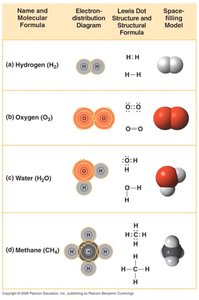
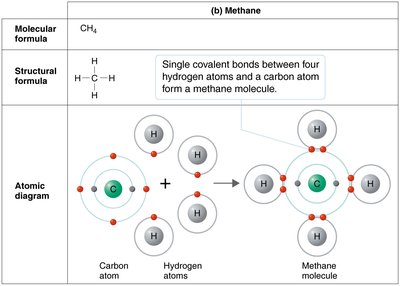
Covalent bonds form when two atoms share one or more pairs of electrons. Molecules are formed when atoms are held together by covalent bonds.
Single covalent bond: Sharing one pair of electrons.
Double covalent bond: Sharing two pairs of electrons.
Electronegativity: The tendency of an atom to attract electrons; affects bond polarity.
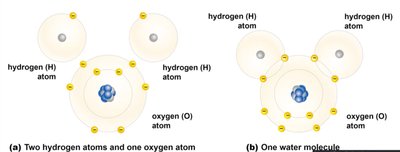
Polar covalent bond: Unequal sharing of electrons, resulting in partial charges (e.g., water).
Non-polar covalent bond: Equal sharing of electrons, no partial charges (e.g., hydrocarbons).
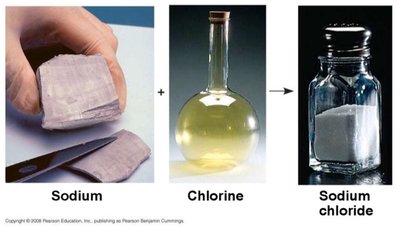
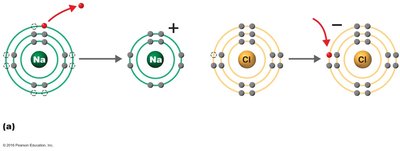
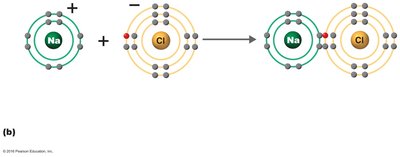
Ionic Bonds
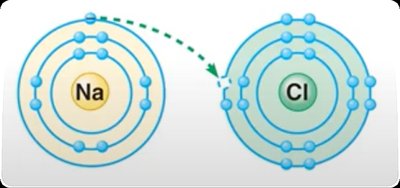
Ionic bonds occur when one atom donates electrons to another, resulting in the formation of ions. Oppositely charged ions attract each other to form ionic compounds.
Ion: Atom or molecule with a net electrical charge due to loss or gain of electrons.
Cation: Positively charged ion (loss of electron).
Anion: Negatively charged ion (gain of electron).
Ionic compound: Substance formed from ionic bonds (e.g., NaCl).
Hydrogen Bonds
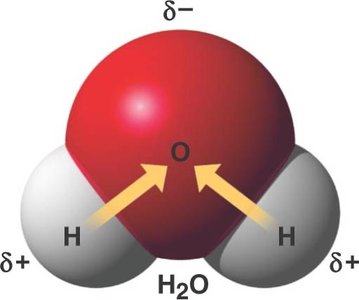
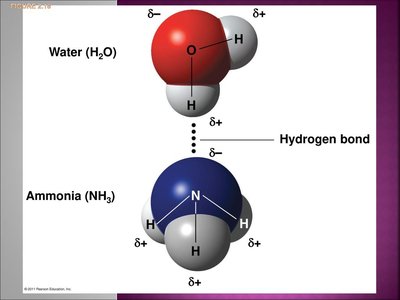
Hydrogen bonds are weak attractions between a covalently bonded hydrogen atom (slightly positive) and an electronegative atom (slightly negative) in another molecule. These bonds are crucial for the structure of water and biological macromolecules.
Hydrogen bonds stabilize the structure of proteins and nucleic acids.
They are responsible for many unique properties of water.
Chemical Reactions
Types of Chemical Reactions
Chemical reactions involve the making or breaking of bonds between atoms, resulting in changes in chemical energy.
Synthesis (Anabolism): Formation of bonds, requires energy (endergonic).
Decomposition (Catabolism): Breaking of bonds, releases energy (exergonic).
Exchange: Combination of synthesis and decomposition.
Reversible reactions: Can proceed in both directions under suitable conditions.
Properties of Water
Structure and Polarity of Water
Water is essential for life, making up 70-95% of cell composition. Its molecular structure and polarity give rise to unique properties.
Polar molecule: Unequal distribution of charges due to electronegativity difference between oxygen and hydrogen.
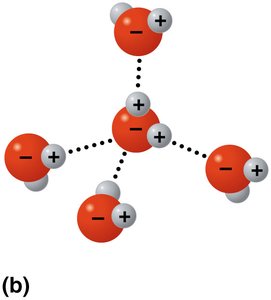
Hydrogen bonding: Water molecules form up to four hydrogen bonds, leading to cohesion and other properties.
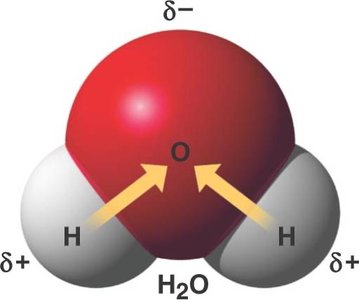
Unique Properties of Water
Cohesion: Water molecules stick together due to hydrogen bonding, enabling water transport in plants and surface tension.
Moderation of temperature: Water has high specific heat, absorbing and retaining heat, which moderates climate.
Expansion upon freezing: Ice is less dense than liquid water due to a rigid hydrogen-bonded structure, allowing aquatic life to survive under ice.
Solvent of life: Water dissolves many substances due to its polarity, forming hydration shells around ions and molecules.
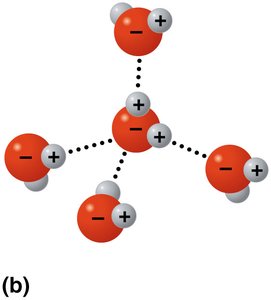
Hydrophilic vs Hydrophobic Substances
Hydrophilic: Substances that interact with water (ionic and polar compounds).
Hydrophobic: Substances that do not interact with water (non-polar compounds).
Acids, Bases, and pH
pH Scale and Biological Importance
The pH scale measures the acidity or basicity of a solution, ranging from 0 (most acidic) to 14 (most basic). Biological processes are sensitive to pH changes.
Acid: Substance that increases H+ concentration (pH < 7).
Base: Substance that increases OH- concentration (pH > 7).
Salt: Ionic compound that dissociates in water but does not release H+ or OH-.
Buffer: Substance that resists changes in pH by donating or accepting H+.
Carbon and Organic Molecules
Carbon: The Backbone of Life
Carbon is central to organic chemistry due to its ability to form four covalent bonds, resulting in diverse structures such as chains, rings, and branches. Organic molecules are primarily composed of carbon and hydrogen, with other elements like oxygen, nitrogen, sulfur, and phosphorus.
Isomers: Compounds with the same molecular formula but different structures.
Functional groups: Groups of atoms that confer specific properties to organic molecules (e.g., hydroxyl, carboxyl, amino).
Macromolecules of Life
Polymers and Monomers
Macromolecules are large, complex molecules essential for life. Most are polymers, made by linking monomers through dehydration reactions.
Monomer: Single building block of a polymer.
Polymer: Chain of monomers linked by covalent bonds.
Dehydration reaction: Joins monomers by removing water.
Carbohydrates
Carbohydrates are primary energy sources and structural components. They consist of carbon, hydrogen, and oxygen, typically in the formula Cn(H2O)n.
Monosaccharides: Simple sugars (e.g., glucose, fructose).
Disaccharides: Two monosaccharides linked (e.g., sucrose, lactose).
Polysaccharides: Complex carbohydrates (e.g., starch, glycogen, cellulose, chitin).
Lipids
Lipids are hydrophobic molecules, including fats, phospholipids, and steroids. They are important for energy storage, membrane structure, and signaling.
Triglycerides: Composed of glycerol and three fatty acids.
Saturated fats: No double bonds, solid at room temperature.
Unsaturated fats: One or more double bonds, liquid at room temperature.
Phospholipids: Amphipathic molecules forming cell membranes.
Steroids: Four-ring carbon skeleton (e.g., cholesterol).
Proteins
Proteins are polymers of amino acids, performing diverse functions such as catalysis, transport, and structural support. Protein structure is hierarchical:
Primary structure: Sequence of amino acids.
Secondary structure: Coiling and folding (alpha helix, beta sheet) stabilized by hydrogen bonds.
Tertiary structure: 3D folding due to interactions among R-groups.
Quaternary structure: Association of multiple polypeptide chains.
Denaturation: Loss of protein structure and function due to environmental changes.
Nucleic Acids
Nucleic acids (DNA, RNA, ATP) store and transmit genetic information. They are polymers of nucleotides, each consisting of a pentose sugar, phosphate group, and nitrogenous base.
DNA: Deoxyribonucleic acid, stores hereditary information.
RNA: Ribonucleic acid, involved in protein synthesis.
ATP: Adenosine triphosphate, energy currency of the cell.
Base pairing: Complementary hydrogen bonding between bases (A-T, G-C in DNA; A-U, G-C in RNA).
Example: Sickle Cell Anemia
Sickle cell anemia is caused by a single base change in DNA, leading to a single amino acid substitution in the beta-globin protein, which alters the protein's structure and function.
Additional info: This chapter provides foundational chemical principles essential for understanding microbial structure, metabolism, and genetics, which are critical for all subsequent topics in microbiology.
