 Back
BackChemistry of Living Things: Foundations for Microbiology
Study Guide - Smart Notes

Chemical Principles of Microbiology
Biochemistry is important because it helps is udnerstand how living things work at the smallest level.
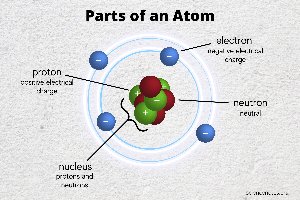
Atoms and Their Structure
Atoms are the fundamental units of matter, forming the basis for all chemical reactions in living organisms. Understanding atomic structure is essential for grasping how molecules interact in microbiology.
Atom: The smallest unit of an element, retaining its chemical properties.
Nucleus: Contains protons (positive charge) and neutrons (neutral).
Electron cloud: Electrons (negative charge) move in shells around the nucleus, determining chemical reactivity.
Ions: Atoms that gain or lose electrons, acquiring a charge.
Isotopes: Atoms with the same number of protons but different numbers of neutrons.
Importance of Isotopes in Medicine and Research
Isotopes, especially radioisotopes, are crucial in modern medicine and biological research. Their unique properties allow for imaging, targeted therapies, and sterilization.
Imaging: PET and SPECT scans use radioisotopes to visualize internal processes.
Targeted treatment: Radioisotopes can deliver radiation to specific cells (e.g., thyroid cancer).
Theranostics: Combines diagnosis and treatment using the same molecule.
Sterilization: Used to sterilize medical equipment and trace drug movement.
Molecules of Microbiology
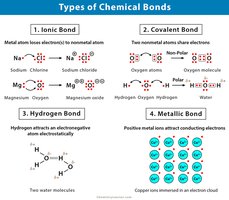
Chemical Bonds
Chemical bonds are the forces that hold atoms together, forming molecules essential for life. The type of bond influences molecular properties and biological functions.
Covalent bonds: Atoms share electrons. Can be non-polar (equal sharing) or polar (unequal sharing).
Ionic bonds: One atom donates electrons to another, resulting in charged ions that attract each other.
Metallic bonds: Electrons are shared among many atoms, typical in metals.
Secondary Bonds: help make weak, temporary connections and help biological molecules fold into their correct 3D shape.
Hydrogen bonds: Weak attractions between a hydrogen atom and electronegative atoms (O, N).
Van der Waals forces: Weak, temporary attractions due to shifting electron clouds.
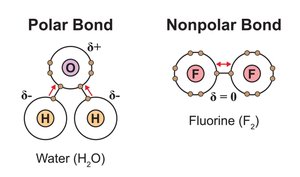
Covalent Bonds: Polar vs. Nonpolar
Covalent bonds are classified based on how electrons are shared:
Non-polar covalent: Electrons are shared equally (e.g., O2, F2).
Polar covalent: Electrons are shared unequally, creating partial charges (e.g., H2O).
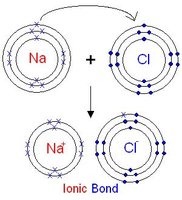
Ionic Bonds
Ionic bonds form when one atom transfers electrons to another, resulting in oppositely charged ions that attract each other. Example: sodium chloride (NaCl).
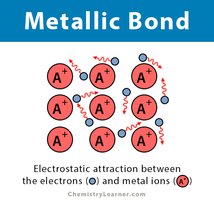
Metallic Bonds
Metallic bonds involve a 'sea of electrons' shared among metal atoms, giving metals their conductivity and malleability.
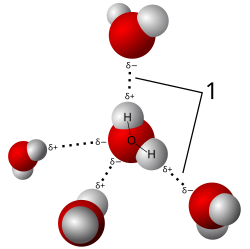
Hydrogen Bonds
Hydrogen bonds are weak but crucial for stabilizing biological molecules, such as DNA and proteins.
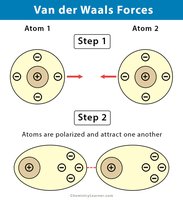
Van der Waals Forces
Van der Waals forces are weak, short-lived attractions that help large biological molecules maintain their structure.
Biological Importance of Chemical Bonds
Understanding chemical bonds is essential for drug design, materials science, and biological function. Manipulating these bonds allows scientists to create targeted therapies and new materials.
Drug Design: Specific bond interactions ensure drugs fit into protein active sites.
Materials Science: Knowledge of bond strengths enables development of advanced materials.
Biological Function: Life processes depend on breaking and forming bonds (e.g., ATP hydrolysis).
Molecules of Microbiology
Organic Macromolecules (Biomolecules)
Organic macromolecules are large, carbon-based molecules essential for life. They are formed by joining monomers into polymers via dehydration synthesis.
Carbohydrates
Lipids (Fats)
Proteins
Nucleic acids
Carbohydrates
Carbohydrates provide energy and structural support. They typically have a 1:2:1 ratio of carbon, hydrogen, and oxygen (CH2O).
Monosaccharides (its simplest form): Simple sugars (e.g., glucose, fructose, galactose).
Functions: Immediate energy, short-term storage, structural support.

Lipids
Lipids are hydrophobic molecules that provide energy, insulation, and are key components of cell membranes.
Fats: Solid at room temperature.
Oils: Liquid at room temperature.
Structure: Composed of glycerol and fatty acids.
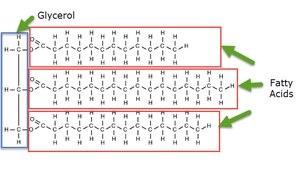
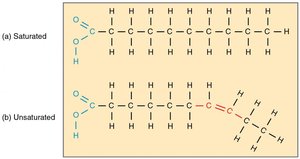
Saturated vs. Unsaturated Fatty Acids
Saturated fatty acids have no double bonds and are typically solid at room temperature, while unsaturated fatty acids have one or more double bonds and are usually liquid.
Saturated: Unhealthy, fully hydrogenated.
Unsaturated: Healthy, contain double bonds.


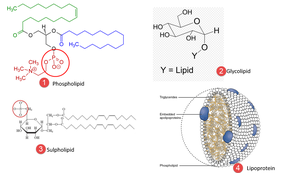
Compound Lipids
Compound lipids contain additional chemical groups, such as phospholipids (phosphate group), glycolipids (hexose units), sulpholipids (sulfated hexose), and lipoproteins (protein subunits).
Sterols
Sterols, such as cholesterol, are characterized by four fused rings and are important for hormone production, bile synthesis, and cell membrane structure.
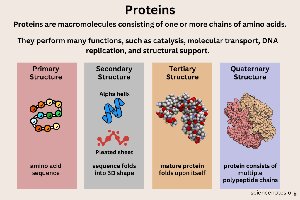
Proteins
Proteins are polymers of amino acids and perform diverse functions, including catalysis, transport, signaling, movement, and defense.
Enzymes: Catalyze biochemical reactions.
Structural: Framework for cells and tissues.
Transport: Carry molecules (e.g., hemoglobin).
Defense: Antibodies.
Hormonal: Chemical messengers.
Movement: Muscle contraction.
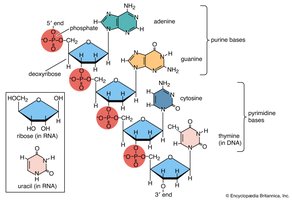
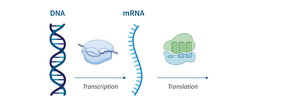
Nucleic Acids
Nucleic acids (DNA and RNA) are polymers of nucleotides, essential for storing and transmitting genetic information and protein synthesis.
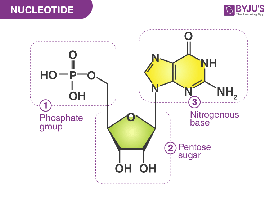
Nucleotide: Composed of a phosphate group, sugar (deoxyribose or ribose), and a nitrogenous base.
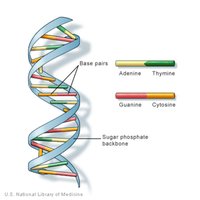
DNA: Double-stranded helix, stores genetic information.
RNA: Single-stranded, involved in protein synthesis.
Summary Table: Macromolecules
Macromolecule | Monomer | Main Functions | Examples |
|---|---|---|---|
Carbohydrates | Monosaccharides | Energy, structure | Glucose, cellulose |
Lipids | Fatty acids, glycerol | Energy, membranes | Triglycerides, phospholipids |
Proteins | Amino acids | Catalysis, structure, transport | Enzymes, collagen |
Nucleic acids | Nucleotides | Genetic information | DNA, RNA |
Additional info: These foundational chemical principles are directly relevant to microbiology, as they underpin cell structure, metabolism, genetic regulation, and molecular interactions essential for microbial life.
