Back
BackControl of Microbial Growth: Principles, Methods, and Applications
Study Guide - Smart Notes

Control of Microbial Growth
Introduction
The control of microbial growth is essential in preventing infections, reducing food spoilage, and maintaining public health. Infectious diseases remain a leading cause of death globally, and about one-third of food is lost to spoilage, especially fruits and vegetables. Understanding the principles and methods of microbial control is fundamental in microbiology and its applications in healthcare, food safety, and industry.
Terminology of Microbial Control
Definitions and Concepts
Sepsis: Bacterial contamination.
Asepsis: Absence of significant contamination; aseptic techniques are crucial in laboratory and surgical settings to prevent microbial contamination.
Sterilization: Removal and destruction of all microbial life.
Commercial Sterilization: Killing Clostridium botulinum endospores in canned goods.
Disinfection: Destruction of harmful microorganisms on inanimate objects.
Antisepsis: Destruction of harmful microorganisms on living tissue.
Degerming: Mechanical removal of microbes from a limited area.
Sanitization: Lowering microbial counts to safe levels.

Suffixes in Microbial Control
-cide: Treatments that kill microbes (e.g., bactericidal, fungicidal, sporicidal, virucidal).
-stasis: Inhibiting growth without killing (e.g., bacteriostatic, fungistatic).

Microbial Characteristics and Resistance
Factors Affecting Microbial Control
Gram-negative bacteria are generally more resistant than gram-positive bacteria to disinfectants and antiseptics.
Mycobacteria, endospores, and protozoan cysts are highly resistant.
Naked viruses are more resistant than enveloped viruses.
Prions are extremely resistant to disinfection and autoclaving.
The Rate of Microbial Death
Principles
Effectiveness of treatment depends on:
Number of microbes present
Environmental conditions (e.g., presence of organic matter)
Time of exposure
Microbial characteristics (e.g., cell wall structure, spore formation)
Microbes typically die at a constant rate when exposed to antimicrobial agents.
Actions of Microbial Control Agents
Mechanisms
Alteration of membrane permeability: Disrupts cell integrity and function.
Damage to proteins (enzymes): Denaturation or destruction of protein structure.
Damage to nucleic acids: Disrupts DNA and RNA, inhibiting replication and function.
Physical Methods of Microbial Control
Overview
Heat (moist and dry)
Filtration
Low temperatures
Desiccation
Osmotic pressure
Radiation
Heat
Denatures enzymes and proteins.
Thermal death point (TDP): Lowest temperature at which all cells in a culture are killed in 10 minutes.
Thermal death time (TDT): Minimum time for all bacteria in a culture to be killed at a given temperature.
Decimal reduction time (DRT): Minutes to kill 90% of a population at a given temperature.

Dry Heat Sterilization
Kills by oxidation (e.g., flaming, incineration, hot-air ovens).
Hot air requires longer exposure than moist heat for sterilization.

Pasteurization
Short, high-heat treatment to reduce spoilage organisms and pathogens.
Common methods:
63°C for 30 min
High-temperature short-time (HTST): 72°C for 15 sec
Ultra-high-temperature (UHT): 140°C for 4 sec

Moist Heat Sterilization
Denatures proteins using boiling, free-flowing steam, or autoclaving.
Autoclave: Steam under pressure (121°C at 15 psi for 15 min) kills all organisms and endospores.
Large containers require longer sterilization times (e.g., test tube: 15 min; 1-liter flask: 75 min).
Test strips are used to indicate sterility.
Filtration
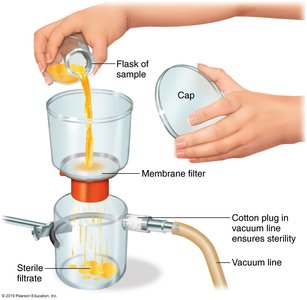
Passage of a substance through a screenlike material to remove microbes.
Used for heat-sensitive materials.
Membrane filters remove microbes >0.22 µm; 0.01 µm filters can retain viruses and some proteins.
HEPA filters remove microbes >0.3 µm from air.
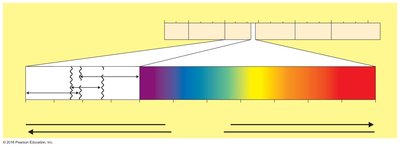
Radiation
Ionizing radiation (X rays, gamma rays, electron beams): Ionizes water to create damaging radicals, causes DNA mutations, used in food industry.
Nonionizing radiation (UV, 260 nm): Damages DNA by creating thymine dimers, used for disinfecting surfaces and vaccines.
Microwaves: Kill by heat; not especially antimicrobial due to uneven heating.
Other Physical Methods
Low temperature: Bacteriostatic effect (refrigeration, deep-freezing, lyophilization).
Desiccation: Absence of water prevents metabolism; microbes can remain viable.
Osmotic pressure: High salt or sugar creates hypertonic environment, causing plasmolysis.
Chemical Methods of Microbial Control
Major Classes of Chemical Agents
Phenols and phenolics
Biguanides (e.g., chlorhexidine)
Halogens (iodine, chlorine)
Alcohols (ethanol, isopropanol)
Heavy metals (silver, copper, mercury, zinc, gold)
Surface-active agents (soaps, quats)
Aldehydes
Chemical food preservatives
Gaseous chemosterilants (ethylene oxide, chlorine dioxide)
Peroxygens (hydrogen peroxide, peracetic acid, ozone)
Principles of Effective Disinfection
Distinguish between disinfectants (inanimate objects) and antiseptics (living tissue).
Follow label instructions for concentration, time, and conditions.
Remove organic matter before disinfection.
Consider pH and other environmental factors.
Evaluating Disinfectants: The Disk-Diffusion Method
This method evaluates the efficacy of chemical agents by placing filter paper disks soaked in chemicals on a microbial culture. The zone of inhibition around the disks indicates effectiveness, but the size of the zone is not always directly proportional to efficacy.

Phenol and Phenolics
Disrupt plasma membrane lipids, causing leakage.
Phenol is rarely used due to irritation and odor; phenolics are modified for better action and less irritation.
Bisphenols (e.g., hexachlorophene, triclosan) are found in soaps and mouthwash.
Biguanides
Disrupt plasma membranes; used for skin disinfection and surgical hand scrubs (e.g., chlorhexidine).
Halogens
Iodine: Impairs protein synthesis and alters membranes.
Chlorine: Oxidizing agent; effective against cysts and endospores at high concentrations. Bleach is hypochlorous acid (HOCl).
Chloramine (chlorine + ammonia) is used for water treatment.
Alcohols
Denature proteins and dissolve lipids; ineffective against endospores and nonenveloped viruses.
Common types: ethanol and isopropanol (optimal concentration: 70%).
Not effective on wounds due to coagulation under which bacteria may survive.

Heavy Metals and Their Compounds
Oligodynamic action: Small amounts exert antimicrobial activity (especially silver and copper).
Denature enzymes and proteins.
Applications: Silver nitrate for neonatal eye infections, silver in medical devices, mercuric chloride in paint.
Potential toxicity limits use.

Surface-Active Agents
Soaps and detergents: Degerming agents; emulsify and remove microbes.
Acid-anionic sanitizers: Used in food industry; disrupt plasma membranes.
Quaternary ammonium compounds (quats): Cationic detergents, bactericidal, more effective against gram-positive bacteria, used in mouthwash and surface disinfection.
Chemical Food Preservatives
Sulfur dioxide: Prevents wine spoilage.
Organic acids: Sorbic acid, benzoic acid, calcium propionate prevent mold in acidic foods.
Nitrites and nitrates: Prevent botulism in meats, preserve color; can form nitrosamines (carcinogens), but risk is low.
Aldehydes
Inactivate proteins by cross-linking functional groups.
Used for preserving specimens and sterilizing medical equipment (e.g., formaldehyde, glutaraldehyde).
Glutaraldehyde is less irritating and more effective than formaldehyde.
Gaseous Chemosterilants
Cause alkylation, cross-linking nucleic acids and proteins, inhibiting cell function.
Ethylene oxide is highly penetrating, used for heat-sensitive items, but toxic and explosive.
Chlorine dioxide is used for fumigating buildings and water treatment.
Peroxygens and Other Forms of Oxygen
Oxidizing agents used for surface and food packaging disinfection.
Hydrogen peroxide is effective on inanimate objects, rapidly degrades to water and oxygen.
Peracetic acid is a liquid sporicide with no toxic residue.
Ozone neutralizes odors and is used in water treatment.
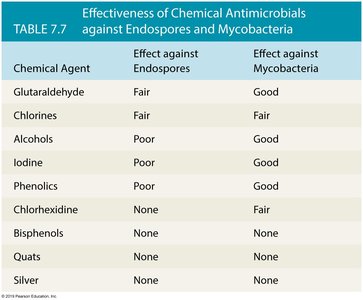
Effectiveness of Chemical Antimicrobials
The following table summarizes the effectiveness of various chemical agents against endospores and mycobacteria:
Chemical Agent | Effect against Endospores | Effect against Mycobacteria |
|---|---|---|
Glutaraldehyde | Fair | Good |
Chlorines | Fair | Fair |
Alcohols | Poor | Good |
Iodine | Poor | Good |
Phenolics | Poor | Poor |
Chlorhexidine | None | Fair |
Bisphenols | None | None |
Quats | None | None |
Silver | None | None |