 Back
BackControlling Microbial Growth in the Body: Antimicrobial Drugs
Study Guide - Smart Notes

Controlling Microbial Growth in the Body: Antimicrobial Drugs
Introduction to Antimicrobial Agents
Antimicrobial drugs are essential tools in modern medicine, used to treat infections by targeting and inhibiting the growth of microorganisms. Their development and use are based on principles of selective toxicity, aiming to harm pathogens while minimizing effects on the host.
Drugs: Chemicals that affect physiology in any manner.
Chemotherapeutic agents: Drugs that act against diseases.
Antimicrobial agents (antimicrobials): Drugs that treat infections caused by microorganisms.
History and Types of Antimicrobial Agents
The discovery and development of antimicrobial agents have revolutionized the treatment of infectious diseases. Key historical figures and milestones include:
Paul Ehrlich: Developed "magic bullets" (arsenic compounds) to kill microbes.
Alexander Fleming: Discovered penicillin from Penicillium mold.
Gerhard Domagk: Discovered sulfanilamide, the first widely used antimicrobial.
Selman Waksman: Coined the term "antibiotics" for naturally produced antimicrobial agents.
Semisynthetics: Chemically altered antibiotics with improved properties.
Synthetics: Completely synthesized antimicrobials in the laboratory.
Sources of Common Antibiotics
Many antibiotics are derived from microorganisms such as fungi and bacteria. The following table summarizes some important sources:
Microorganism | Antimicrobial |
|---|---|
Penicillium chrysogenum | Penicillin G |
Streptomyces griseus | Streptomycin |
Bacillus polymyxa | Polymyxin |
Streptomyces aureofaciens | Tetracycline |
Micromonospora purpurea | Gentamicin |
Streptomyces nodosus | Amphotericin B |
Streptomyces noursei | Nystatin |
Amycolatopsis orientalis | Vancomycin |
Mechanisms of Antimicrobial Action
Selective Toxicity and Drug Targets
Successful antimicrobial therapy relies on selective toxicity, where drugs target features unique to pathogens. The main mechanisms include:
Inhibition of cell wall synthesis
Inhibition of protein synthesis
Disruption of cytoplasmic membranes
Inhibition of metabolic pathways
Inhibition of nucleic acid synthesis
Prevention of virus attachment, entry, or uncoating
Inhibition of Cell Wall Synthesis
Many antibiotics, such as beta-lactams, inhibit the synthesis of peptidoglycan in bacterial cell walls, leading to cell lysis. Other drugs target unique features in fungal or mycobacterial cell walls.
Beta-lactams: Bind to enzymes that cross-link NAM subunits in peptidoglycan.
Vancomycin and cycloserine: Interfere with bridges between NAM subunits in Gram-positive bacteria.
Bacitracin: Blocks transport of NAG and NAM from cytoplasm.
Isoniazid and ethambutol: Disrupt mycolic acid formation in mycobacteria.
Echinocandins: Inhibit glucan synthesis in fungal cell walls.
Inhibition of Protein Synthesis
Antimicrobials can selectively target prokaryotic ribosomes (70S) without affecting eukaryotic ribosomes (80S), though mitochondrial ribosomes may be affected.
Drugs may bind to the 30S or 50S subunits, blocking translation.
Mupirocin inhibits isoleucyl-tRNA synthetase in Gram-positive bacteria.
Disruption of Cytoplasmic Membranes
Some drugs disrupt membrane integrity, leading to cell death.
Nystatin and amphotericin B: Bind to ergosterol in fungal membranes.
Polymyxin: Disrupts membranes of Gram-negative bacteria; toxic to human kidneys.
Azoles and allylamines inhibit ergosterol synthesis in fungi.
Inhibition of Metabolic Pathways
Antimetabolic agents target unique metabolic processes in pathogens.
Atovaquone interferes with electron transport in protozoa and fungi.
Sulfonamides and trimethoprim inhibit folic acid synthesis in bacteria.
Antiviral agents may block viral uncoating or protease activity.
Inhibition of Nucleic Acid Synthesis
Some drugs block DNA replication or RNA transcription, often affecting both prokaryotic and eukaryotic cells.
Quinolones and fluoroquinolones inhibit DNA gyrase in bacteria.
Nucleotide/nucleoside analogs distort nucleic acid structure, blocking replication.
Reverse transcriptase inhibitors target HIV replication.
Prevention of Virus Attachment, Entry, or Uncoating
Attachment antagonists block viral proteins or host receptors, preventing infection.
Pleconaril blocks viral attachment.
Arildone prevents viral uncoating.
Clinical Considerations in Prescribing Antimicrobial Drugs
Properties of Ideal Antimicrobial Agents
The ideal antimicrobial agent is effective, safe, stable, and easy to administer. Key properties include:
Readily available and inexpensive
Chemically stable and easily administered
Nontoxic, nonallergenic, and selectively toxic
Spectrum of Action
The spectrum of action refers to the range of pathogens a drug affects.
Narrow-spectrum: Effective against a few organisms.
Broad-spectrum: Effective against many organisms, but may disrupt normal flora and cause superinfections.
Testing Antimicrobial Effectiveness
Several laboratory tests are used to evaluate the efficacy of antimicrobial agents:
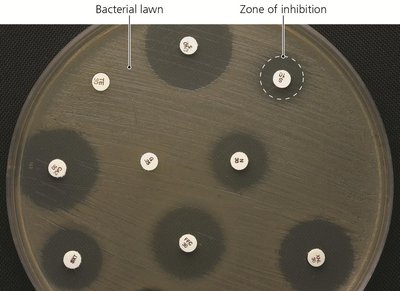
Diffusion susceptibility (Kirby-Bauer) test: Measures zones of inhibition around antibiotic disks on an agar plate.
Minimum inhibitory concentration (MIC) test: Determines the lowest concentration of drug that inhibits visible growth.
Minimum bactericidal concentration (MBC) test: Identifies the lowest concentration that kills the organism.
Routes of Administration
Antimicrobial drugs can be administered in several ways, affecting their distribution and effectiveness:
Topical: For external infections.
Oral: Self-administered, no needles required.
Intramuscular (IM): Injected into muscle.
Intravenous (IV): Delivered directly to bloodstream for rapid effect.
Safety and Side Effects
Antimicrobial drugs may cause adverse effects, including toxicity, allergies, and disruption of normal microbiota.
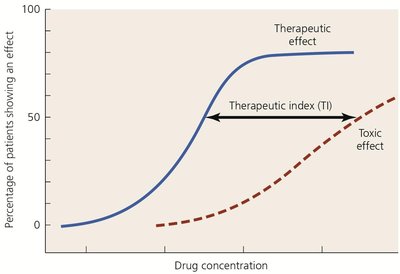
Toxicity: May affect kidneys, liver, or nerves; therapeutic index (TI) is the ratio of tolerated dose to effective dose.
Allergies: Rare but potentially life-threatening (e.g., anaphylactic shock).
Disruption of normal microbiota: Can lead to secondary infections or overgrowth of normal flora.
Resistance to Antimicrobial Drugs
Development of Resistance
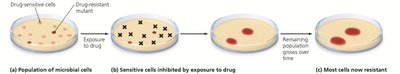
Microbial resistance to drugs can arise through mutations or acquisition of resistance genes (R plasmids). Overuse and misuse of antibiotics accelerate this process.
Resistance can be acquired by mutation or horizontal gene transfer (transformation, transduction, conjugation).
Persister cells and resistant mutants survive drug exposure and proliferate.
Mechanisms of Resistance
Bacteria employ several strategies to resist antimicrobial drugs:
Produce enzymes (e.g., beta-lactamase) that destroy or deactivate drugs.
Prevent drug entry or pump drugs out of the cell (efflux pumps).
Alter drug targets to reduce binding.
Change metabolic pathways or protect target enzymes.
Biofilm formation increases resistance.
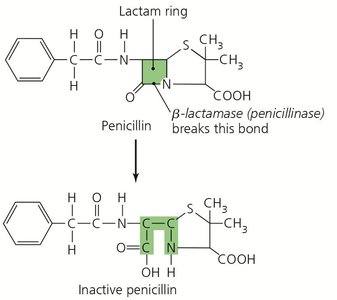
Multiple Resistance and Cross Resistance
Pathogens may become resistant to multiple drugs, especially in healthcare settings where antibiotics are frequently used. Cross resistance can occur among drugs with similar structures.
Multiple-drug-resistant pathogens resist at least three antimicrobial agents.
Cross resistance: Resistance to one drug confers resistance to others in the same class.
Retarding Resistance
Strategies to slow the development of resistance include:
Maintaining high drug concentrations to ensure pathogen elimination.
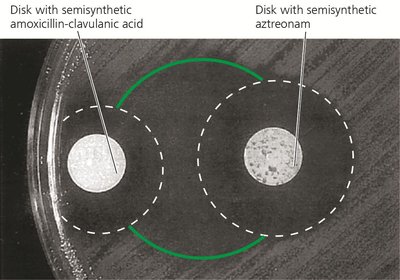
Using drug combinations (synergism enhances effect; antagonism reduces efficacy).
Limiting antimicrobial use to necessary cases.
Developing new drugs and modifying existing ones.
Summary Table: Mechanisms of Antimicrobial Action
Mechanism | Example Drugs | Target/Effect |
|---|---|---|
Cell Wall Synthesis Inhibition | Penicillins, Cephalosporins, Vancomycin | Peptidoglycan cross-linking |
Protein Synthesis Inhibition | Tetracyclines, Aminoglycosides | Ribosomal subunits |
Membrane Disruption | Polymyxin, Amphotericin B | Membrane integrity |
Metabolic Pathway Inhibition | Sulfonamides, Trimethoprim | Folic acid synthesis |
Nucleic Acid Synthesis Inhibition | Quinolones, Rifampin | DNA/RNA synthesis |
Attachment/Entry Inhibition | Pleconaril, Arildone | Viral entry/uncoating |
Additional info: Some table entries and explanations have been expanded for clarity and completeness.
