 Back
BackChpt 10 Controlling Microbial Growth in the Body: Antimicrobial Drugs
Study Guide - Smart Notes

Controlling Microbial Growth in the Body: Antimicrobial Drugs
Introduction to Antimicrobial Agents
Antimicrobial drugs are essential tools in the treatment of infectious diseases. Their development and use have revolutionized medicine by enabling the control of microbial growth within the human body. This chapter explores the history, mechanisms, clinical considerations, and resistance associated with antimicrobial drugs.
The History of Antimicrobial Agents
Key Definitions
Drugs: Chemicals that affect physiology in any manner.
Chemotherapeutic agents: Drugs that act against diseases, including but not limited to anticancer drugs.
Antimicrobial agents (antimicrobials): Drugs that treat infections caused by microorganisms.
Pioneers in Antimicrobial Discovery
Paul Ehrlich: Proposed the concept of "magic bullets"—chemicals that selectively kill pathogens without harming host cells. Developed arsenic compounds effective against microbes.
Alexander Fleming: Discovered penicillin, the first natural antibiotic, produced by the fungus Penicillium chrysogenum.
Gerhard Domagk: Discovered sulfanilamide, the first widely used antibiotic.
Selman Waksman: Discovered streptomycin and other antibiotics from soil bacteria, earning a Nobel Prize.
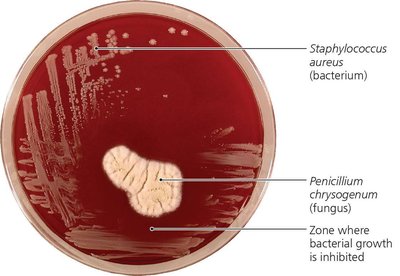
Example: The discovery of penicillin demonstrated the ability of certain fungi to inhibit bacterial growth, as seen in the clear zone (zone of inhibition) around the fungus on an agar plate.
Types of Antimicrobial Agents
Semisynthetics: Chemically modified antibiotics that are more effective, longer-lasting, or easier to administer than natural antibiotics (e.g., doxycycline, minocycline).
Synthetics: Completely synthesized in the laboratory (e.g., sulfonamides, cephalosporins).
Sources of Antibiotics
Fungi: Penicillium and Acremonium species produce penicillins and cephalosporins.
Bacteria: Streptomyces, Bacillus, and others produce most clinically useful antibiotics (e.g., streptomycin, tetracycline, erythromycin).
Mechanisms of Antimicrobial Action
Principle of Selective Toxicity
Successful chemotherapy relies on selective toxicity, where the drug targets the pathogen without harming the host. Antibacterial drugs are the most diverse, while fewer drugs are available for eukaryotic pathogens and viruses due to similarities with host cells.
Major Mechanisms of Action
Inhibition of cell wall synthesis
Inhibition of protein synthesis
Disruption of cytoplasmic membrane
Inhibition of metabolic pathways
Inhibition of nucleic acid synthesis
Prevention of virus attachment, entry, or uncoating
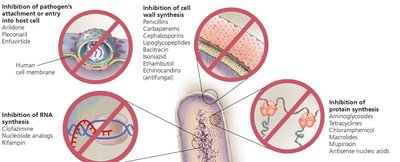
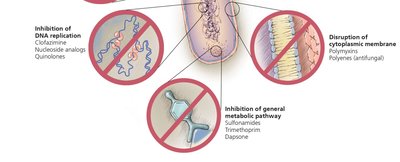
Inhibition of Cell Wall Synthesis
Most bacterial cell walls contain peptidoglycan, which is targeted by several antibiotics:
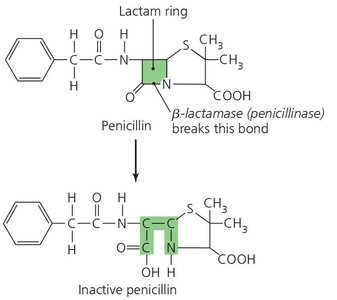
Beta-lactams (e.g., penicillins, cephalosporins): Prevent cross-linkage of NAM subunits, weakening the cell wall and causing lysis.
Vancomycin and cycloserine: Interfere with bridges between NAM subunits in Gram-positive bacteria.
Bacitracin: Blocks transport of NAG and NAM from the cytoplasm.
Isoniazid and ethambutol: Disrupt mycolic acid formation in mycobacteria.
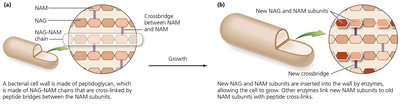
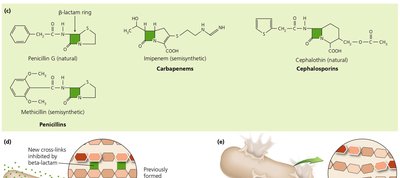
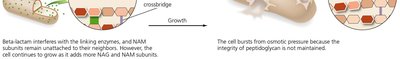
Note: These drugs are only effective against growing cells, as they do not affect existing peptidoglycan.
Inhibition of Fungal Cell Wall Synthesis
Fungal cell walls contain unique polysaccharides. Echinocandins inhibit glucan synthesis, disrupting fungal cell walls.
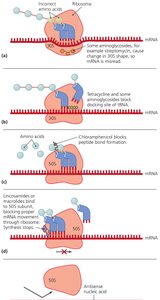
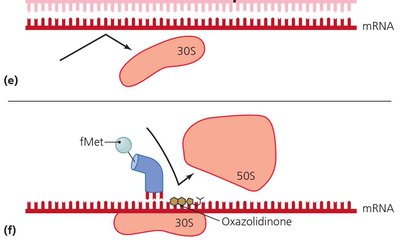
Inhibition of Protein Synthesis
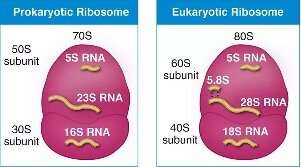
Prokaryotic ribosomes (70S) differ from eukaryotic ribosomes (80S), allowing selective targeting:
Drugs can bind to the 30S or 50S subunits, blocking translation.
Examples: Aminoglycosides, tetracyclines, chloramphenicol, macrolides, oxazolidinones.
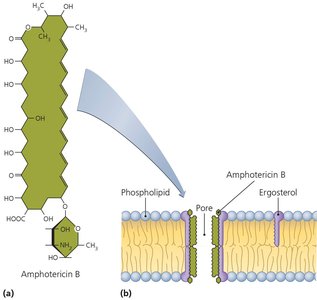
Disruption of Cytoplasmic Membranes
Some drugs (e.g., polymyxin, amphotericin B) form channels in membranes, compromising integrity.
Amphotericin B binds ergosterol in fungal membranes; polymyxin targets Gram-negative bacteria but is toxic to human kidneys.
Inhibition of Metabolic Pathways
Antimetabolic agents exploit differences in pathogen and host metabolism.
Examples: Atovaquone (electron transport), heavy metals (enzyme inactivation), sulfonamides (folic acid synthesis inhibition).
Some drugs prevent viral uncoating (amantadine, rimantadine) or inhibit viral enzymes (protease inhibitors for HIV).
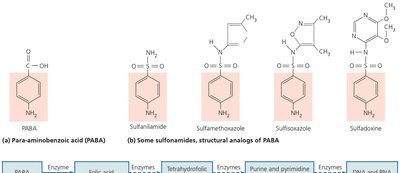
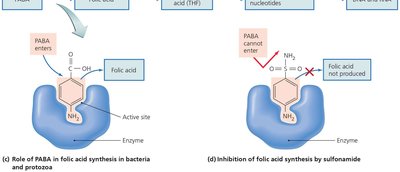
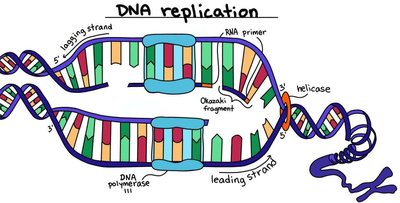
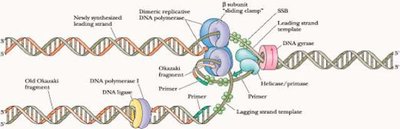
Inhibition of Nucleic Acid Synthesis
Some drugs block DNA replication or RNA transcription (e.g., quinolones, fluoroquinolones, nucleotide/nucleoside analogs).
Quinolones target bacterial DNA gyrase, not affecting human helicase.
Nucleotide analogs distort nucleic acid shapes, preventing replication—useful against viruses and cancer cells.
Reverse transcriptase inhibitors target HIV-specific enzymes.
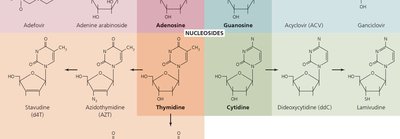
Prevention of Virus Attachment, Entry, or Uncoating
Attachment antagonists block viral binding to host cells (e.g., pleconaril).
Some drugs prevent viral uncoating (e.g., arildone).
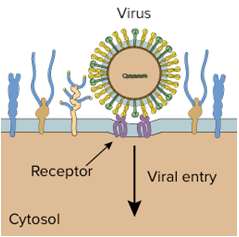
Clinical Considerations in Prescribing Antimicrobial Drugs
Ideal Properties of Antimicrobial Agents
Readily available, inexpensive, chemically stable, easily administered, nontoxic, nonallergenic, and selectively toxic against a wide range of pathogens.
Spectrum of Action
Narrow-spectrum: Effective against a few organisms.
Broad-spectrum: Effective against many organisms but may cause secondary infections by disrupting normal flora.

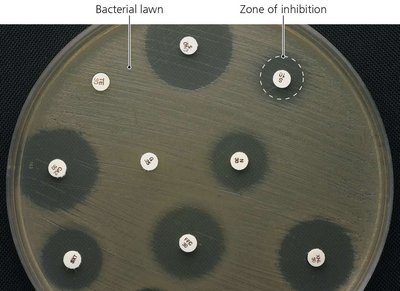
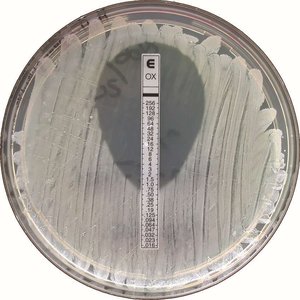
Testing Antimicrobial Effectiveness
Diffusion susceptibility (Kirby-Bauer) test: Measures zones of inhibition around antibiotic disks.
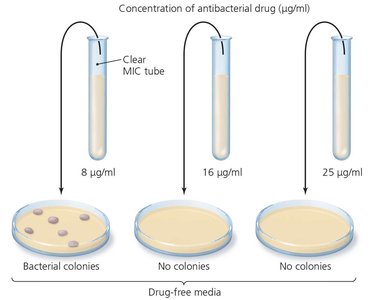
Minimum inhibitory concentration (MIC) test: Determines the lowest drug concentration that inhibits growth.
Minimum bactericidal concentration (MBC) test: Identifies the lowest concentration that kills bacteria.
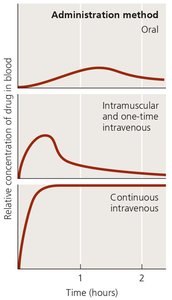
Routes of Administration
Topical: For external infections.
Oral: Easiest, but may result in lower blood concentrations.
Intramuscular: Injection into muscle.
Intravenous: Directly into bloodstream for highest concentration.
Safety and Side Effects
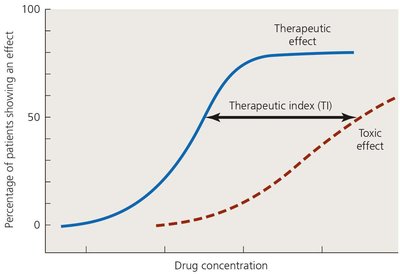
Toxicity: May affect kidneys, liver, or nerves; special caution for pregnant women. Therapeutic index (TI) is the ratio of tolerated dose to effective dose; higher TI means safer drug.
Therapeutic window: Range of concentrations effective without excessive toxicity.
Disruption of normal microbiota: Can lead to secondary infections (e.g., yeast infections after antibiotics).
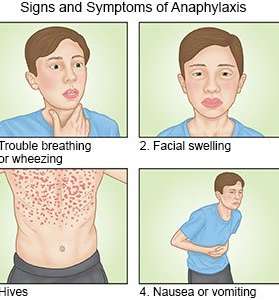
Allergies: Rare but can cause life-threatening anaphylaxis; treated with epinephrine (EpiPen).
Resistance to Antimicrobial Drugs
Development of Resistance
Some pathogens are naturally resistant; others acquire resistance via mutations or horizontal gene transfer (transformation, transduction, conjugation).
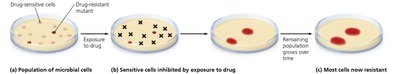
Mechanisms of Resistance
Enzyme production that destroys or deactivates the drug (e.g., beta-lactamases).
Prevention of drug entry into the cell.
Alteration of drug target.
Efflux pumps expel the drug.
Biofilm formation.
Production of proteins that protect target enzymes (e.g., MfpA in Mycobacterium tuberculosis).
Multiple Resistance and Cross Resistance
Pathogens may become resistant to multiple drugs, especially in healthcare settings.
Cross resistance occurs when drugs share similar structures or mechanisms.
Retarding Resistance
Maintain high drug concentrations to inhibit pathogens.
Use combinations of drugs (synergism enhances effect; antagonism reduces efficacy).
Prescribe antimicrobials only when necessary and complete the full course.
Develop new generations of drugs to overcome resistance.

Summary Table: Key Antimicrobial Mechanisms and Examples
Mechanism | Drug Examples | Target Pathogen |
|---|---|---|
Cell wall synthesis inhibition | Penicillins, cephalosporins, vancomycin | Bacteria |
Protein synthesis inhibition | Tetracyclines, macrolides, aminoglycosides | Bacteria |
Membrane disruption | Polymyxin, amphotericin B | Bacteria, fungi |
Metabolic pathway inhibition | Sulfonamides, trimethoprim | Bacteria, protozoa |
Nucleic acid synthesis inhibition | Quinolones, nucleotide analogs | Bacteria, viruses |
Viral attachment/entry inhibition | Pleconaril, arildone | Viruses |
Additional info: This guide covers the core concepts and clinical considerations for antimicrobial drugs, including mechanisms of action, resistance, and best practices for use. Understanding these principles is essential for effective infection control and combating antimicrobial resistance.
