 Back
BackControlling Microbial Growth in the Body: Antimicrobial Drugs
Study Guide - Smart Notes

Controlling Microbial Growth in the Body: Antimicrobial Drugs
The History of Antimicrobial Agents
Antimicrobial drugs are essential tools in modern medicine, used to treat infections by targeting microbial physiology. The development of these agents has revolutionized the treatment of infectious diseases.
Drugs: Chemicals that affect physiology in any manner.
Chemotherapeutic agents: Drugs that act against diseases.
Antimicrobial agents (antimicrobials): Drugs that treat infections.
Key historical figures:
Paul Ehrlich: Developed "magic bullets"—arsenic compounds that killed microbes.
Alexander Fleming: Discovered penicillin released from Penicillium.
Gerhard Domagk: Discovered sulfanilamide.
Selman Waksman: Coined the term "antibiotics" for naturally produced antimicrobial agents.
Semisynthetics: Chemically altered antibiotics for improved efficacy and administration.
Synthetics: Antimicrobials synthesized entirely in the laboratory.
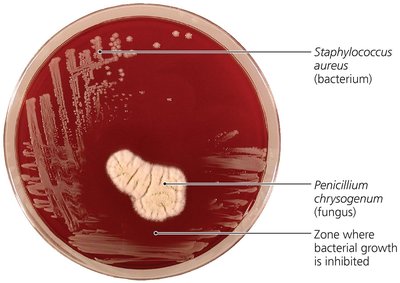
Sources of Common Antibiotics and Semisynthetics
Antibiotics and semisynthetic antimicrobials are derived from various microorganisms, primarily fungi and bacteria. The following table summarizes key sources:
Microorganism | Antimicrobial |
|---|---|
Penicillium chrysogenum (Fungi) | Penicillin G |
Penicillium griseofulvum (Fungi) | Griseofulvin |
Acremonium spp. (Fungi) | Cephalosporin |
Amycolatopsis orientalis (Bacteria) | Vancomycin |
Bacillus polymyxa (Bacteria) | Polymyxin |
Streptomyces griseus (Bacteria) | Streptomycin |
Streptomyces aureofaciens (Bacteria) | Tetracycline |
Streptomyces nodosus (Bacteria) | Amphotericin B |
Streptomyces avermitilis (Bacteria) | Ivermectin |
Additional info: Many antibiotics are produced by Streptomyces species. |
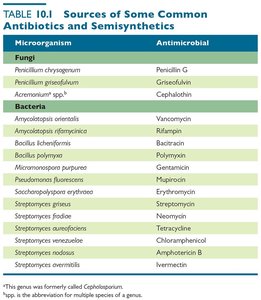
Mechanisms of Antimicrobial Action
Successful chemotherapy relies on selective toxicity, targeting microbial processes without harming the host. Antibacterial drugs are the most numerous, with fewer options for fungi, protozoa, and viruses.
Major mechanisms:
Inhibition of cell wall synthesis
Inhibition of protein synthesis
Disruption of cytoplasmic membranes
Inhibition of metabolic pathways
Inhibition of nucleic acid synthesis
Inhibition of pathogen's attachment or entry into host cell
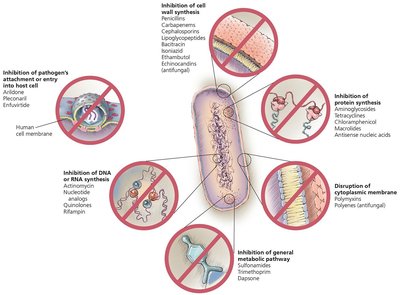
Inhibition of Cell Wall Synthesis
Many antimicrobial agents target the synthesis of bacterial cell walls, particularly peptidoglycan. This mechanism is highly effective against actively growing bacteria.
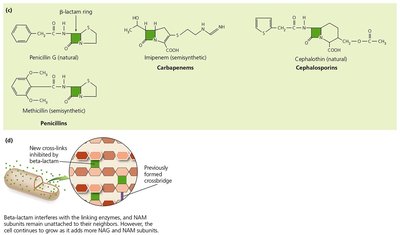
Beta-lactams: Prevent cross-linkage of NAM subunits by binding to enzymes responsible for peptidoglycan synthesis. The functional group is the beta-lactam ring.
Vancomycin and cycloserine: Interfere with bridges linking NAM subunits in Gram-positive bacteria.
Bacitracin: Blocks transport of NAG and NAM from cytoplasm.
Isoniazid and ethambutol: Disrupt mycolic acid formation in mycobacterial species.
Fungal cell wall inhibitors: Echinocandins inhibit glucan synthesis.
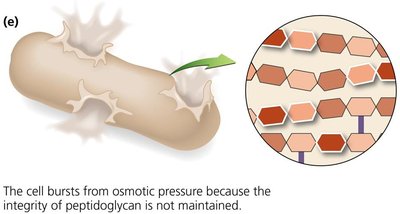
Effectiveness: Only affects growing cells; no effect on existing peptidoglycan.
Inhibition of Protein Synthesis
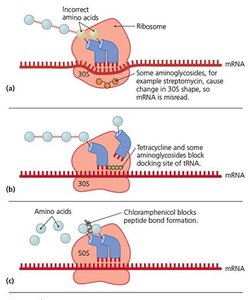
Antimicrobial drugs can selectively target prokaryotic ribosomes (70S) without affecting eukaryotic ribosomes (80S), disrupting translation and protein production.
Aminoglycosides: Cause misreading of mRNA by altering 30S subunit.
Tetracyclines: Block docking site of tRNA on 30S subunit.
Chloramphenicol: Blocks peptide bond formation on 50S subunit.
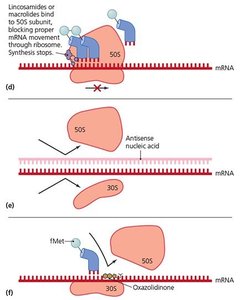
Macrolides and lincosamides: Bind to 50S subunit, blocking mRNA movement.
Oxazolidinones: Block initiation of translation.
Mupirocin: Inhibits aminoacyl-tRNA synthetase, preventing loading of isoleucine in Gram-positive bacteria.
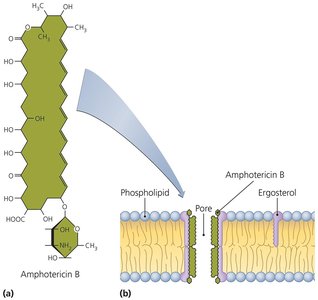
Disruption of Cytoplasmic Membranes
Some drugs compromise the integrity of microbial membranes, leading to cell death.
Nystatin and amphotericin B: Attach to ergosterol in fungal membranes, forming pores.
Azoles and allylamines: Inhibit ergosterol synthesis.
Polymyxin: Disrupts membranes of Gram-negative bacteria; toxic to human kidneys.
Antiparasitic drugs: Target cytoplasmic membranes of parasites.
Humans: Somewhat susceptible due to cholesterol similarity to ergosterol; bacteria lack sterols and are not susceptible.
Inhibition of Metabolic Pathways
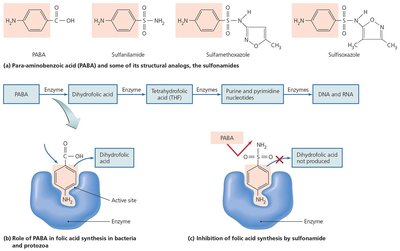
Antimetabolic agents exploit differences between pathogen and host metabolism.
Atovaquone: Interferes with electron transport in protozoa and fungi.
Heavy metals: Inactivate enzymes.
Agents disrupting tubulin polymerization and glucose uptake: Target protozoa and parasitic worms.
Drugs blocking viral activation: Metabolic antagonists.
Trimethoprim: Interferes with nucleotide synthesis.
Antiviral agents: Amantadine, rimantadine, and weak organic bases prevent viral uncoating; protease inhibitors block HIV replication.
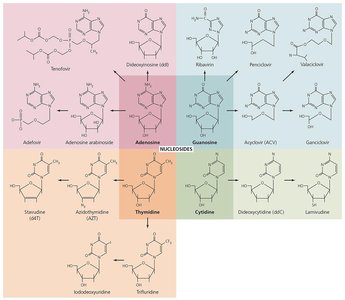
Inhibition of Nucleic Acid Synthesis
Drugs that block DNA replication or RNA transcription are often used in research or to slow cancer cell replication, as they can affect both prokaryotic and eukaryotic cells.
Quinolones and fluoroquinolones: Act against prokaryotic DNA gyrase.
Nucleotide or nucleoside analogs: Distort nucleic acid shapes, preventing replication, transcription, or translation; most often used against viruses and rapidly dividing cancer cells.
Inhibitors of RNA polymerase: Block transcription.
Reverse transcriptase inhibitors: Target HIV replication; do not harm humans as we lack reverse transcriptase.
Additional info:
Selective toxicity is the foundation of antimicrobial drug design, aiming to minimize harm to the host while effectively targeting pathogens.
Resistance to antimicrobial drugs is a growing concern, requiring careful use and ongoing research into new agents.
