 Back
BackControlling Microbial Growth in the Body: Antimicrobial Drugs
Study Guide - Smart Notes

Controlling Microbial Growth in the Body: Antimicrobial Drugs
Mechanisms of Antimicrobial Action
Antimicrobial drugs are designed to inhibit or kill pathogenic microorganisms within the body. Their effectiveness depends on their ability to target microbial processes without causing excessive harm to the host.
Inhibition of Cell Wall Synthesis: Drugs such as penicillins and cephalosporins prevent the formation of peptidoglycan, weakening bacterial cell walls and causing cell lysis.
Inhibition of Protein Synthesis: Agents like tetracyclines and erythromycin interfere with ribosomal function, halting protein production in microbes.
Disruption of Cytoplasmic Membranes: Some drugs, such as polymyxins, compromise membrane integrity, leading to cell death.
Inhibition of Metabolic Pathways: Sulfonamides block essential metabolic reactions, such as folic acid synthesis.
Inhibition of Nucleic Acid Synthesis: Drugs like fluoroquinolones inhibit DNA replication or transcription.
Prevention of Virus Attachment, Entry, or Uncoating: Antiviral drugs may block viral proteins or prevent uncoating, stopping infection at early stages.
Example: Pleconaril blocks viral attachment, while arildone prevents viral uncoating.
Clinical Considerations in Prescribing Antimicrobial Drugs
Ideal Antimicrobial Agent
The ideal antimicrobial drug should be readily available, inexpensive, chemically stable, easily administered, nontoxic, nonallergenic, and selectively toxic against a wide range of pathogens.
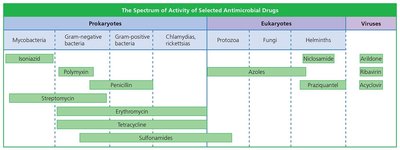
Spectrum of Action
The spectrum of action refers to the range of pathogens a drug can affect. Narrow-spectrum drugs target specific organisms, while broad-spectrum drugs act against many types. Broad-spectrum drugs may lead to secondary infections or superinfections by disrupting normal flora.
Effectiveness
Drug effectiveness is determined by laboratory tests:
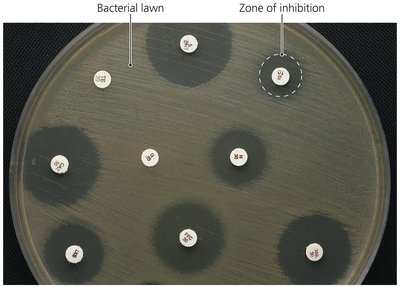
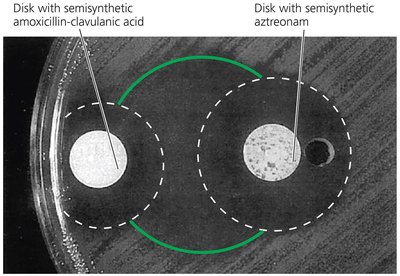
Diffusion Susceptibility Test (Kirby-Bauer): Measures the zone of inhibition around drug disks on a bacterial lawn.
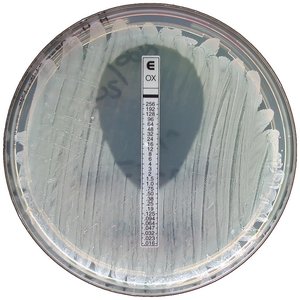
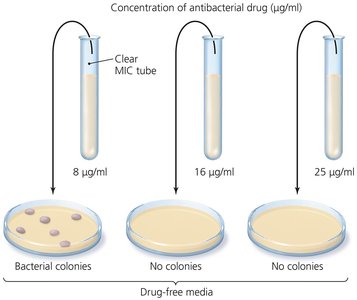
Minimum Inhibitory Concentration (MIC) Test: Identifies the lowest concentration of drug that prevents visible growth.
Minimum Bactericidal Concentration (MBC) Test: Determines the lowest concentration that kills the bacteria.

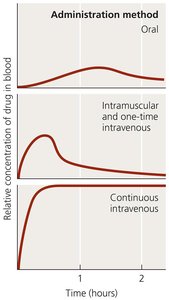
Routes of Administration
Antimicrobial drugs can be administered topically, orally, intramuscularly, or intravenously. The route affects drug distribution and concentration in the blood.
Safety and Side Effects
Toxicity: Some drugs may harm kidneys, liver, or nerves. The therapeutic index (ratio of tolerated dose to effective dose) and therapeutic window (range of effective concentrations) are important safety measures.
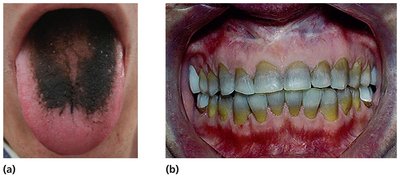
Allergies: Rare but potentially life-threatening reactions, such as anaphylactic shock, can occur.
Disruption of Normal Microbiota: May cause secondary infections or superinfections, especially in hospitalized patients.
Resistance to Antimicrobial Drugs
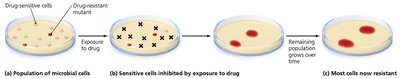
Development of Resistance in Populations
Microbial resistance arises naturally or through acquired mechanisms:
New Mutations: Changes in chromosomal genes can confer resistance.
Acquisition of R Plasmids: Horizontal gene transfer via transformation, transduction, or conjugation spreads resistance.
Mechanisms of Resistance
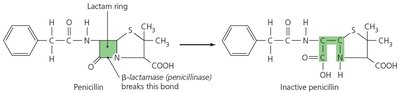
Enzyme production that destroys or deactivates the drug (e.g., beta-lactamase).
Prevention of drug entry into the cell.
Alteration of drug targets.
Modification of metabolic pathways.
Efflux pumps expel drugs from the cell.
Bacteria in biofilms resist antimicrobials.
Specific proteins (e.g., MfpA in Mycobacterium tuberculosis) bind drug targets and prevent drug action.
Multiple Resistance and Cross Resistance
Pathogens may develop resistance to multiple drugs, especially in healthcare settings where R plasmids are exchanged. Multiple-drug-resistant pathogens resist at least three agents. Cross resistance occurs when drugs are structurally similar.
Retarding Resistance
Maintain high drug concentration for sufficient time to inhibit pathogens.
Use combination therapy (synergism enhances effects; antagonism reduces efficacy).
Use antimicrobials only when necessary.
Develop new drug variations (second- and third-generation drugs).
Search for new antibiotics, semisynthetics, and synthetics.
Design drugs complementary to microbial protein shapes.
Key Terms and Concepts
Zone of Inhibition: Area around an antimicrobial disk where bacteria do not grow, indicating drug effectiveness.
MIC (Minimum Inhibitory Concentration): Lowest concentration of drug preventing visible microbial growth.
MBC (Minimum Bactericidal Concentration): Lowest concentration of drug that kills the microbe.
Therapeutic Index:
Synergism: Combined effect of drugs is greater than the sum of their individual effects.
Antagonism: Drugs interfere with each other's action.
Example: Beta-lactamase breaks the beta-lactam ring of penicillin, rendering it inactive.
Additional info: Combination therapy and prudent use of antimicrobials are essential strategies to slow resistance development and preserve drug efficacy.
