 Back
BackControlling Microbial Growth in the Environment: Physical and Chemical Methods
Study Guide - Smart Notes

Controlling Microbial Growth in the Environment
Introduction
Controlling microbial growth is essential in healthcare, food preservation, laboratory work, and various industries to prevent infection, spoilage, and contamination. This chapter reviews the physical and chemical methods used to control microbial populations in the environment, focusing on their mechanisms, effectiveness, and applications.
Physical Methods of Microbial Control
Radiation
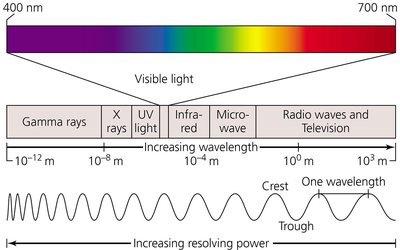
Radiation is a physical method used to control microbial growth by damaging cellular components, particularly DNA. There are two main types: ionizing and nonionizing radiation.
Ionizing Radiation: Involves wavelengths shorter than 1 nm (e.g., electron beams, gamma rays, X-rays). It ejects electrons from atoms, creating ions that disrupt hydrogen bonding, oxidize double covalent bonds, and generate hydroxyl radicals. These ions denature molecules such as DNA, leading to microbial death.
Nonionizing Radiation: Involves wavelengths greater than 1 nm (e.g., ultraviolet [UV] light). It excites electrons, causing the formation of new covalent bonds and affecting the 3-D structure of proteins and nucleic acids. UV light induces pyrimidine dimers in DNA, inhibiting replication and transcription.
Applications: Ionizing radiation is used for sterilizing medical equipment and food preservation, while nonionizing radiation is suitable for disinfecting air, transparent fluids, and surfaces.

Other Physical Methods
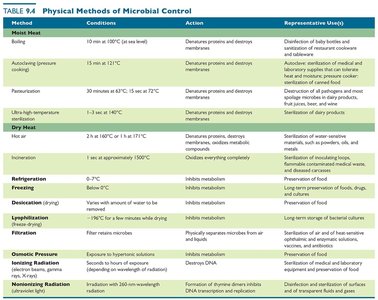
Additional physical methods include heat (moist and dry), refrigeration, freezing, desiccation, filtration, and osmotic pressure. Each method targets microbial structures or metabolism to inhibit or destroy microorganisms.
Method | Conditions | Action | Representative Use(s) |
|---|---|---|---|
Boiling | 10 min at 100°C | Denatures proteins, destroys membranes | Disinfection of baby bottles and sanitization of restaurant cookware |
Autoclaving | 15 min at 121°C | Denatures proteins, destroys membranes | Sterilization of medical and laboratory supplies |
Pasteurization | 30 min at 63°C or 15 sec at 72°C | Denatures proteins, destroys membranes | Destruction of pathogens and spoilage microbes in dairy products, fruit juices, beer, and wine |
Ultra-high-temperature sterilization | 1–3 sec at 140°C | Denatures proteins, destroys membranes | Sterilization of dairy products |
Dry heat | 2 hr at 160°C or 1 hr at 171°C | Denatures proteins, destroys membranes, oxidizes metabolic compounds | Sterilization of water-sensitive materials such as powders, oils, and metals |
Refrigeration | 0–7°C | Inhibits metabolism | Preservation of food, drugs, and cultures |
Desiccation | Varies with amount of water to be removed | Inhibits metabolism | Preservation of food |
Filtration | Filter retains microbes | Physically separates microbes from air and liquids | Sterilization of air and heat-sensitive ophthalmic and enzymatic solutions, vaccines, antibiotics |
Osmotic pressure | Exposure to hypertonic solutions | Inhibits metabolism | Preservation of food (e.g., jerky, jams, jellies, salted fish) |
Ionizing radiation | Seconds to hours of exposure (depending on intensity) | Destroys DNA | Sterilization of medical and laboratory equipment and preservation of food |
Nonionizing radiation | Seconds to hours of exposure (depending on wavelength) | Formation of thymine dimers inhibits DNA transcription and replication | Disinfection and sterilization of surfaces and of transparent fluids and gases |
Relative Susceptibility of Microbes
Microorganisms vary in their resistance to antimicrobial agents. Prions and bacterial endospores are among the most resistant, while enveloped viruses are the most susceptible.
Most resistant: Prions, bacterial endospores, protozoan cysts, mycobacteria
Most susceptible: Enveloped viruses, most Gram-positive bacteria

Chemical Methods of Microbial Control
Overview
Chemical agents control microbial growth by affecting cell walls, membranes, proteins, or DNA. Their effectiveness depends on environmental conditions and the type of microorganism targeted. They are often more effective against enveloped viruses and vegetative cells.
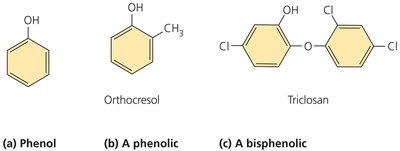
Phenol and Phenolics
Phenol and its derivatives (phenolics) denature proteins and disrupt cell membranes. They are effective in the presence of organic matter and remain active for prolonged periods. Commonly used in healthcare and laboratories, they have a strong odor and potential side effects.
Alcohols
Alcohols are intermediate-level disinfectants that denature proteins and disrupt cytoplasmic membranes. They are more effective than soap for hand hygiene but less effective against some viruses. Alcohol swabs are commonly used before injections to reduce microbial load.
Halogens
Halogens (e.g., iodine, chlorine, bromine) are intermediate-level antimicrobial chemicals that damage enzymes by denaturation. They are widely used for disinfecting water, surfaces, and medical equipment.
Oxidizing Agents
Oxidizing agents such as peroxides, ozone, and peracetic acid kill microbes by oxidizing their enzymes. They are high-level disinfectants and antiseptics, useful for sterilizing surfaces and equipment. Hydrogen peroxide is less effective on open wounds due to catalase activity in tissues.
Surfactants
Surfactants are surface-active chemicals that reduce surface tension, aiding in the removal of microbes. Soaps are good degerming agents but not antimicrobial, while detergents (especially quaternary ammonium compounds, or quats) disrupt cellular membranes and are used in medical and industrial settings.
Heavy Metals
Heavy-metal ions (e.g., silver, mercury, copper) denature proteins and act as low-level bacteriostatic and fungistatic agents. Examples include silver nitrate for preventing neonatal blindness and copper for controlling algal growth.
Aldehydes
Aldehydes (e.g., glutaraldehyde, formalin) contain terminal —CHO groups and cross-link functional groups to denature proteins and inactivate nucleic acids. They are used for disinfecting and sterilizing medical equipment and preserving biological specimens.
Gaseous Agents
Gaseous agents are microbicidal and sporicidal gases used in closed chambers to sterilize items. They denature proteins and DNA but can be hazardous, explosive, poisonous, and potentially carcinogenic.
Enzymes
Antimicrobial enzymes, such as lysozyme in human tears, digest peptidoglycan in bacterial cell walls. Enzymes are used to reduce microbial contamination in food and on medical instruments (e.g., prionzyme for prion removal).
Antimicrobial Drugs
Antibiotics, semisynthetic, and synthetic chemicals are primarily used to treat disease but can also be used for environmental microbial control.
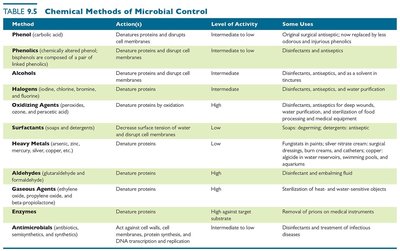
Method | Action(s) | Level of Activity | Some Uses |
|---|---|---|---|
Phenol | Denatures proteins and disrupts cell membranes | Intermediate to low | Original surgical antiseptic, now replaced by less odorous alternatives |
Phenolics | Denature proteins and disrupt cell membranes | Intermediate to low | Disinfectants and antiseptics |
Alcohols | Denature proteins and disrupt cell membranes | Intermediate | Disinfectants, antiseptics, and as a solvent in tinctures |
Halogens | Denature proteins | Intermediate | Disinfectants, antiseptics, and water purification |
Oxidizing Agents | Denature proteins by oxidation | High | Disinfectants, antiseptics for deep wounds, water purification, and sterilization of food-processing and medical equipment |
Surfactants | Decrease surface tension of water and disrupt cell membranes | Low | Soaps, detergents, and antiseptics |
Heavy Metals | Denature proteins | Low | Fungistats in paints, silver nitrate cream, surgical dressings, burn creams, and catheters |
Aldehydes | Denature proteins | High | Disinfectant and embalming fluid |
Gaseous Agents | Denature proteins and DNA by cross-linking functional groups | High | Sterilization of heat- and water-sensitive objects |
Enzymes | Denature proteins | High against target substrate | Removal of prions on medical instruments |
Antimicrobials | Act against cell walls, cell membranes, protein synthesis, and DNA transcription and replication | Intermediate to low | Disinfectants and treatment of infectious diseases |
Evaluating Disinfectants and Antiseptics
Phenol Coefficient
The phenol coefficient test compares the effectiveness of a chemical agent to phenol under standardized conditions. A coefficient greater than 1.0 indicates higher efficacy than phenol. This method has largely been replaced by newer tests.
Use-Dilution Test
In this test, metal cylinders are contaminated with bacteria, immersed in disinfectant dilutions, and then incubated in growth medium. The most effective agents prevent growth at the highest dilution. This is the current standard in the United States.
Kelsey-Sykes Capacity Test
This European Union-approved test involves adding bacterial suspensions to the chemical, sampling at intervals, and incubating in media with a disinfectant deactivator. The minimum effective time is determined by the absence of bacterial growth.
In-Use Test
Swabs are taken from objects before and after disinfectant application, inoculated into growth medium, and monitored for microbial growth. This test determines the proper strength and application procedure for specific situations.
Development of Resistant Microbes
Overuse of antiseptic and disinfectant chemicals can promote the development of resistant microbes. There is little evidence that these products significantly improve human or animal health, highlighting the importance of prudent use.
