 Back
BackCulture Media and Quantification of Microbial Growth
Study Guide - Smart Notes

Culture Media
Definition and Criteria
Culture media are nutrient materials prepared for the growth of microorganisms in laboratory settings. The selection and preparation of culture media are critical for successful microbial cultivation and experimentation.
Essential Nutrients: Must provide all nutrients required for microbial growth.
Moisture: Sufficient water content is necessary for metabolic processes.
pH: The medium must have an appropriate pH for the target organism.
Oxygen Level: The medium must support the oxygen requirements (aerobic or anaerobic) of the organism.
Sterility: The medium must be sterile before inoculation to prevent contamination.
Incubation Temperature: The medium must be able to be incubated at the correct temperature for the organism.
Types of Media: Physical State
Liquid Media: Broths that do not solidify.
Solid Media: Contain agar, a polysaccharide derived from red algae (seaweed), which solidifies the medium.

Agar Properties: Most organisms cannot degrade agar. It liquefies above 100°C and remains liquid until cooled below 40°C.
Complex Media
Complex media contain nutrients in the form of extracts or digests from natural sources such as yeasts, meat, or plants. The exact chemical composition is not known, making them suitable for a wide range of organisms.
Energy and Elemental Sources: Generally provided by proteins and their digests.
Ease of Use: Most organisms can grow on complex media.

Example: Nutrient agar is a common complex medium used for heterotrophic bacteria.
Chemically Defined Media
In chemically defined media, all chemical components and their exact concentrations are known. These media are essential for studying the nutritional requirements of specific microorganisms.
Applications: Used for research on metabolic pathways and for growing fastidious organisms with known requirements.

Example: A defined medium for Escherichia coli includes glucose, ammonium phosphate, sodium chloride, magnesium sulfate, potassium phosphate, and water.
Selective and Differential Media
Specialized media are used to isolate or differentiate between microorganisms based on their physiological properties.
Selective Media: Suppress or kill unwanted microbes and encourage the growth of desired organisms. Examples include bismuth sulfite agar (for Salmonella typhi) and Sabouraud’s Dextrose agar (for fungi).
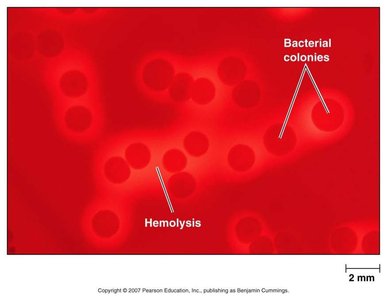
Differential Media: Allow visual distinction between different types of colonies based on specific biochemical reactions. They do not inhibit growth but reveal differences, such as hemolysis on blood agar.
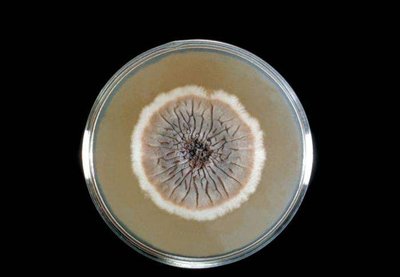
Multipurpose Media
Some media serve both selective and differential functions, aiding in the identification and isolation of specific groups of bacteria.
Eosin Methylene Blue (EMB): Selective for Gram-negative bacteria; differentiates E. coli (metallic green sheen).
MacConkey Agar: Selective for Gram-negative enterics; differentiates lactose fermenters (pink colonies) from non-fermenters.
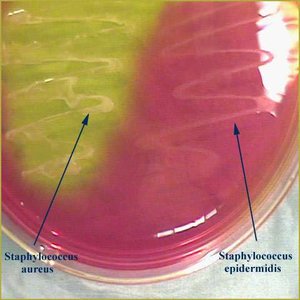
Mannitol Salt Agar (MSA): Selective for high salt-tolerant Gram-positives; differentiates mannitol fermenters (yellow) from non-fermenters (pink).
Hektoen Enteric (HE): Selective for Gram-negatives; differentiates Shigella and Salmonella from other Enterobacteriaceae.
Enrichment Culture
Enrichment cultures are used to increase the numbers of a specific microorganism present in low abundance by providing favorable conditions for its growth over others.
Application: Useful for isolating organisms from environmental or clinical samples where they are present in small numbers.
Special Growth Conditions
Anaerobes
Anaerobic bacteria require environments devoid of oxygen due to oxygen toxicity. Special media and equipment are used to cultivate these organisms.
Reducing Media: Contain chemicals like sodium thioglycolate to remove dissolved oxygen.
Anaerobic Jars: Use chemical reactions to remove oxygen and create an anaerobic environment for culture plates.
OxyPlate: Contains oxyrase enzyme to reduce oxygen to water, creating anaerobic conditions.
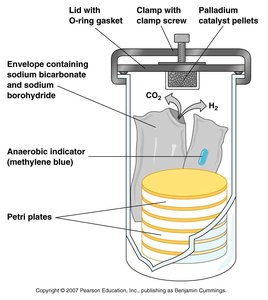
Anaerobic Growth Chambers: Large chambers filled with inert gases, allowing manipulation of cultures without oxygen exposure.

Microaerophiles
Microaerophiles require lower oxygen concentrations than are present in the atmosphere. Special incubation methods, such as candle jars or CO2-generating packets, are used to create suitable environments.
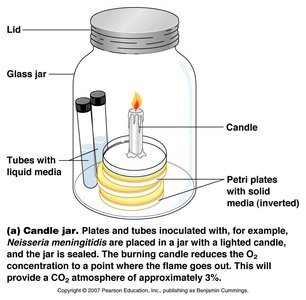
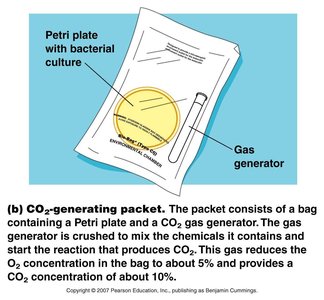
Obligate Intracellular Parasites
Certain bacteria, such as Rickettsia, Chlamydia, and Mycobacterium leprae, cannot be grown on artificial media because they require host cells for nutrients and energy due to their reduced genomes.
Quantifying Bacteria
Counting Methods
Quantification of bacteria is essential for microbiological studies and can be achieved through direct and indirect methods.
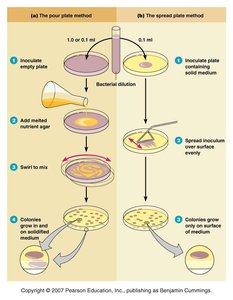
Plate Counts: Involves serial dilution and plating to count colonies. Valid counts are between 30-300 colonies per plate.
Pour Plate Method: Diluted bacteria are mixed with melted agar and poured into plates.
Spread Plate Method: Diluted bacteria are spread on the surface of solid agar.
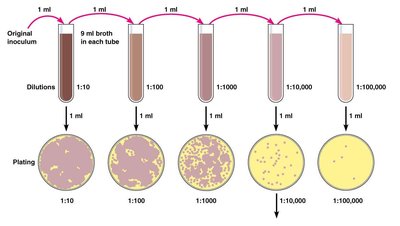
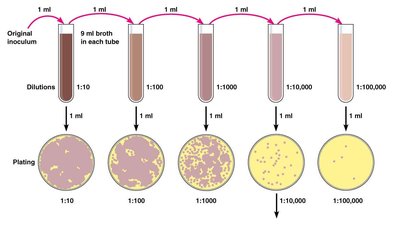
Calculation Formula:
Filtration
Used for samples with low bacterial counts, such as environmental water samples. A known volume is filtered, and the membrane is incubated to count colonies.

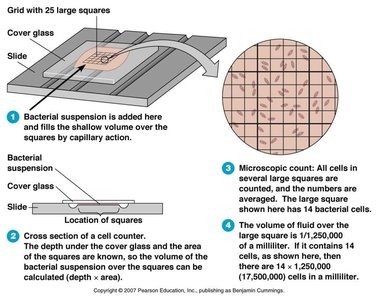
Direct Microscopic Count
Special slides with grids (hemocytometers) are used to count bacteria in a known volume under a microscope. This method is rapid but does not distinguish between live and dead cells.
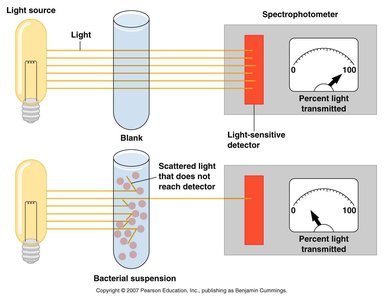
Indirect Methods
Turbidity: Measured using a spectrophotometer; increased turbidity correlates with higher bacterial numbers.
Metabolic Activity: Quantifies metabolic products (e.g., acid, CO2) as a proxy for cell number.
Dry Weight: Bacteria are dried and weighed, often used in industrial microbiology.
Example: Filtering 10 liters of ocean water and finding 55 coliform colonies means there are 5.5 coliform bacteria per liter.
Additional info: These methods are foundational for microbial enumeration in research, clinical, and industrial settings.
