 Back
BackEnvironmental Microbiology: Microbial Diversity, Biogeochemical Cycles, and Water Treatment
Study Guide - Smart Notes

Environmental Microbiology
Microbial Diversity and Habitats
Microbial populations are highly diverse and occupy a wide range of ecological niches. They compete with other organisms and adapt to various environmental conditions, including extreme environments.
Extremophiles: Microorganisms that thrive in extreme pH, temperature, or salinity. Most extremophiles are Archaea.
Symbiosis
Symbiosis refers to a close association between two unlike organisms that benefits one or both partners. In environmental microbiology, symbiotic relationships are crucial for nutrient cycling and plant health.
Ruminants and Digestive Bacteria: Ruminants (e.g., sheep, cows) rely on digestive bacteria in the rumen to break down cellulose.
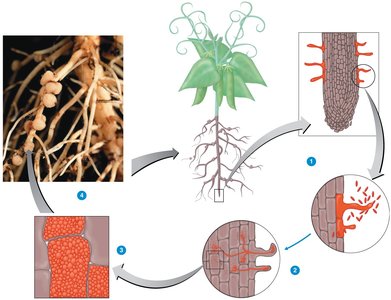
Mycorrhizae: Symbiotic associations between plant roots and fungi, enhancing nutrient and water uptake. Two main types are:
Endomycorrhizae: Fungi penetrate root cells.
Ectomycorrhizae: Fungi form a sheath around roots (e.g., truffles with oak trees).
Commercial Value: Mycorrhizal infection can significantly improve plant growth and is commercially valuable for agriculture and forestry.

Soil Microbiology and Biogeochemical Cycles
Microbial Populations in Soil
Soil contains millions of bacteria per gram, most of which cannot be cultured in the laboratory. Metagenomics allows for the identification and sequencing of novel microbial genes directly from soil samples. The largest microbial populations are found in the top few centimeters of soil.
Biogeochemical Cycles
Microorganisms drive the cycling of essential elements by oxidizing and reducing them to meet their metabolic needs. The main biogeochemical cycles include the carbon, nitrogen, sulfur, and phosphorus cycles.
The Carbon Cycle
The carbon cycle describes the movement of carbon among the atmosphere, biosphere, and geosphere, largely mediated by microorganisms.
Photoautotrophs: Fix CO2 into organic matter using sunlight (e.g., plants, algae, cyanobacteria).
Chemoheterotrophs: Use organic matter for energy, releasing CO2 via respiration.
Decomposers: Oxidize organic compounds from dead plants and animals, returning CO2 to the atmosphere.
Human Impact: Burning fossil fuels releases additional CO2, contributing to global warming.
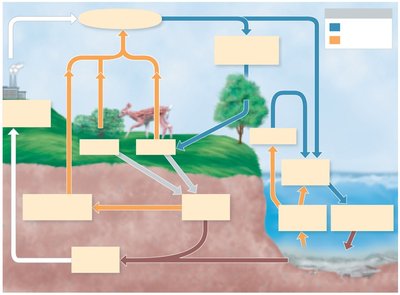
The Nitrogen Cycle
Nitrogen is essential for proteins and nucleic acids. Although N2 makes up 80% of the atmosphere, it must be fixed into organic compounds by microorganisms before it can be used by most living organisms.
Decomposition: Microbial breakdown of proteins into amino acids, followed by deamination (removal of amino groups) and ammonification (release of ammonia, NH3).
Nitrification: Oxidation of ammonium ions (NH4+) to nitrite (NO2-) and then to nitrate (NO3-) by autotrophic bacteria (Nitrosomonas and Nitrobacter).
Denitrification: Reduction of nitrate to nitrogen gas (N2) by denitrifying bacteria (e.g., Pseudomonas), returning nitrogen to the atmosphere.
Nitrogen Fixation: Conversion of atmospheric N2 to ammonia by nitrogenase enzyme in free-living (e.g., Azotobacter, Beijerinckia, Clostridium, cyanobacteria) and symbiotic bacteria (e.g., Rhizobium in root nodules of legumes).
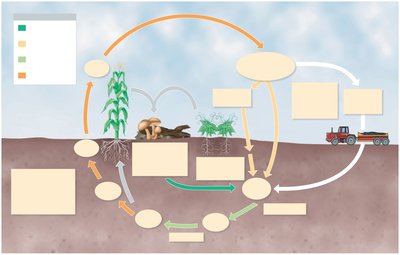
The Sulfur Cycle
Sulfur is cycled through the environment by microbial processes, including the formation of hydrogen sulfide (H2S) under anaerobic conditions, which serves as an energy source for autotrophic bacteria. Sulfates (SO42-) are incorporated into amino acids by plants and bacteria.
Dissimilation: Decomposition of proteins releases H2S.
Oxidation: H2S is oxidized to elemental sulfur and sulfates by bacteria such as Thiomargarita and Acidithiobacillus.
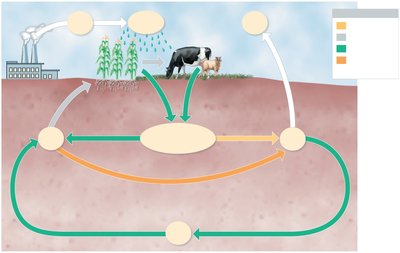
The Phosphorus Cycle
Phosphorus cycles between soluble and insoluble forms, and between organic and inorganic states. It exists mainly as phosphate ions (PO43-), and its availability is influenced by pH. Certain bacteria (e.g., Acidithiobacillus) can solubilize phosphate from rocks.
Degradation of Synthetic Chemicals in Soil and Water
Biodegradation and Xenobiotics
Natural organic matter is readily degraded by microbes, but xenobiotics (synthetic chemicals not naturally found in nature) are often resistant to microbial degradation. Examples include pesticides and industrial chemicals such as Agent Orange.
Bioremediation and Bioaugmentation
Bioremediation: The use of microbes to detoxify or degrade pollutants. The process can be enhanced by adding nitrogen and phosphorus fertilizers.
Bioaugmentation: The addition of specific pollutant-degrading microbes to contaminated environments.
Composting: The controlled microbial degradation of organic waste by thermophiles, converting plant remains into humus.


Aquatic Microbiology and Water Treatment
Aquatic Microbiology
Aquatic microbiology studies microorganisms in natural waters. High numbers of microbes indicate high nutrient levels. Many aquatic bacteria have appendages and holdfasts for attachment to surfaces.
Water Quality and Pollution
Pathogen Transmission: Some pathogens (e.g., Salmonella, Vibrio cholerae) are transmitted via contaminated water.
Chemical Pollution: Many chemicals are resistant to biodegradation. Eutrophication, caused by excess nitrogen and phosphorus, leads to algal blooms and oxygen depletion.

Water Purity Tests
Indicator Organisms: Coliforms (e.g., Escherichia coli) are used to detect fecal contamination. They are aerobic or facultatively anaerobic, gram-negative rods that ferment lactose with acid and gas production within 48 hours at 35°C.
Testing Methods: The Most Probable Number (MPN) method is commonly used.
Limitations: Some pathogens (e.g., viruses, Giardia intestinalis, Cryptosporidium) are resistant to chemical disinfection and may not be detected by coliform tests.
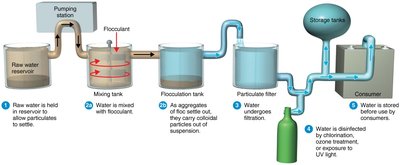
Water Treatment
Municipal water treatment involves several steps to ensure water safety:
Coagulation and Flocculation: Removal of particulates and colloidal materials by adding alum (flocculant).
Filtration: Water passes through sand or coal filters, removing microorganisms by adsorption.
Disinfection: Water is treated with ozone, chlorine, or UV light to kill remaining pathogens.
Sewage (Wastewater) Treatment
Primary Treatment
Primary treatment removes solids from sewage by sedimentation. Sludge collects in sedimentation tanks. This process removes 25–35% of the biochemical oxygen demand (BOD), which measures the amount of biodegradable organic matter in water.
Secondary Treatment
Secondary treatment uses aerobic microbes to oxidize organic matter, removing 75–95% of BOD.
Activated Sludge System: Air is passed through sewage, promoting microbial growth.
Trickling Filters: Sewage is sprayed over rocks or plastic, forming a biofilm of aerobic microbes.
Rotating Biological Contactors: Disks rotate to aerate wastewater and support microbial biofilms.
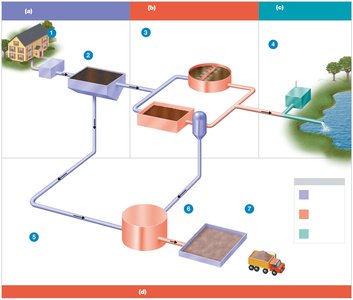
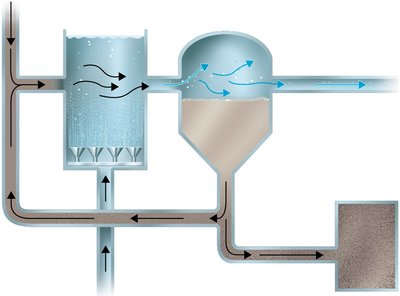
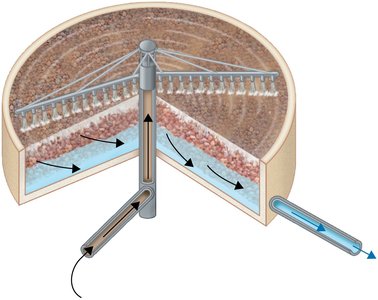
Sludge Digestion and Disinfection
Sludge Digestion: Anaerobic bacteria degrade organic solids in sludge digesters, producing methane and carbon dioxide.
Disinfection: Treated sewage is disinfected (usually by chlorination) before release into the environment.
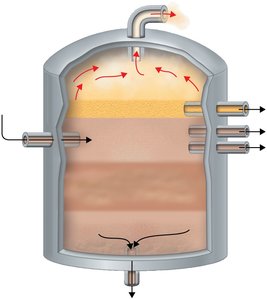
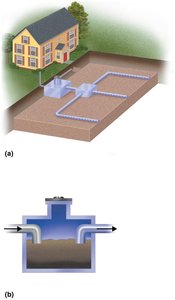
Alternative Sewage Treatment Methods
Septic Tanks: Used in low-density areas for primary treatment; effluent is decomposed by soil microbes in drainage fields.
Oxidation Ponds: Used for further treatment; algae produce oxygen for aerobic decomposition.
Tertiary Treatment
Tertiary treatment removes remaining BOD, nitrogen, and phosphorus through physical and chemical processes, including chlorination. The resulting water can be made potable ("toilet to tap").
