 Back
BackCH. 7 Fundamentals of Microbial Growth and Decontamination
Study Guide - Smart Notes

Fundamentals of Microbial Growth and Decontamination
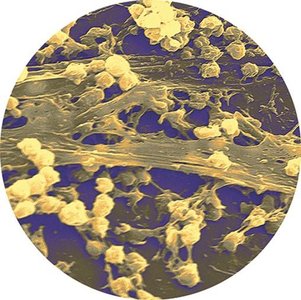
Microbial Growth in Nature and Biofilms
Microbial growth refers to the increase in the number of cells through cell division, resulting in a larger population. In nature, microbes often exist in complex communities, forming biofilms where cells communicate and collaborate for survival. Biofilms are especially problematic in healthcare settings due to their resistance to treatment and role in persistent infections. Biofilm formation begins when planktonic (free-floating) bacteria adhere to surfaces, such as indwelling medical devices.
Biofilm: A structured community of microbial cells enclosed in a self-produced polymeric matrix attached to a surface.
Clinical relevance: Biofilms on catheters and heart valves can lead to chronic infections.
Example pathogens: Pseudomonas aeruginosa, Methicillin-resistant Staphylococcus aureus (MRSA).
Mechanisms of Microbial Cell Division
Bacteria primarily reproduce by binary fission, an asexual process resulting in two genetically identical daughter cells. Some microbes use alternative methods such as budding or spore formation.
Binary fission: Chromosome replication, septum formation, and cell division.
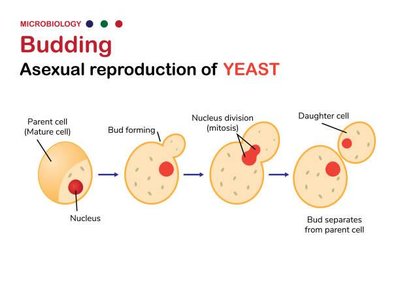
Budding: Cell elongates, forms a bud, and separates; common in some bacteria and fungi.
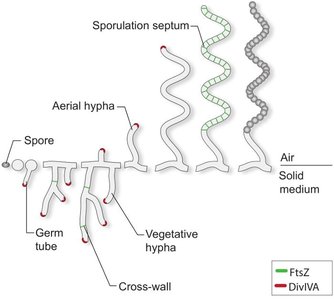
Spore formation: Some bacteria and fungi produce spores for survival and reproduction.
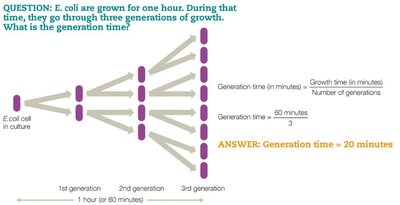
Generation Time and Exponential Growth
Generation time is the period required for a cell to divide. Bacterial populations grow exponentially under optimal conditions, with generation times varying by species and environment.
Generation time: Time for one cell division (e.g., E. coli: 20 minutes; Mycobacterium tuberculosis: 15–20 hours).
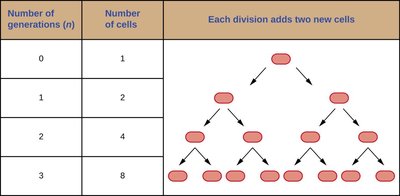
Exponential growth: Population doubles with each generation.
Equation:
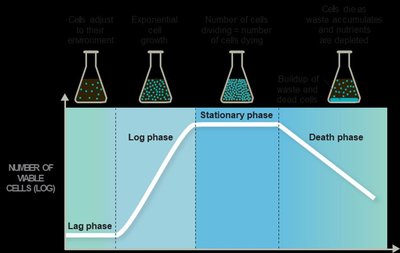
Growth Phases in Closed Batch Culture
When bacteria are cultured in a closed system, they exhibit four distinct growth phases: lag, log, stationary, and death. The duration and characteristics of each phase depend on the species, growth conditions, and initial inoculum size.
Lag phase: Cells adapt to new environment; no immediate division.
Log (exponential) phase: Rapid cell division and population growth.
Stationary phase: Nutrient depletion and waste accumulation halt growth; some cells form endospores.
Death phase: Cells die at an exponential rate due to harsh conditions.
Industrial Microbiology: Chemostat
In industrial settings, maintaining cells at a specific growth phase is crucial for optimizing product yield. A chemostat continuously supplies fresh medium and removes waste, keeping cells in the desired phase.

Environmental Factors Affecting Microbial Growth
Microbes adapt to various environmental conditions, including temperature, pH, salinity, and oxygen availability. These factors determine the ecological niche and pathogenic potential of microbes.
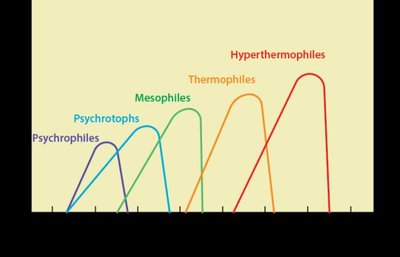
Temperature: Psychrophiles, psychrotrophs, mesophiles, thermophiles, and extreme thermophiles are classified by their optimal growth temperatures.
pH: Acidophiles, neutralophiles, and alkaliphiles thrive in different pH ranges.
Salinity: Halophiles and facultative halophiles tolerate high salt concentrations.
Oxygen: Microbes are classified by their oxygen requirements and tolerance.

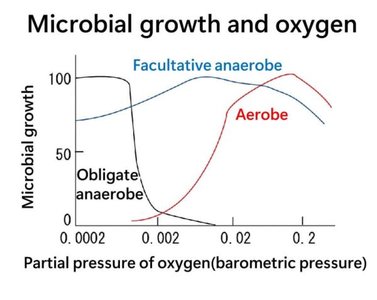
Oxygen Requirements and Tolerance
Microbes are categorized based on their oxygen use and tolerance. Oxygen can be toxic due to reactive oxygen species (ROS), so microbes have evolved various strategies to detoxify ROS.
Obligate aerobes: Require oxygen for growth.
Obligate anaerobes: Cannot tolerate oxygen.
Facultative anaerobes: Grow with or without oxygen.
Aerotolerant anaerobes: Tolerate oxygen but do not use it.
Microaerophiles: Require low levels of oxygen.
Type | Oxygen Use | ROS Detoxification |
|---|---|---|
Obligate Aerobe | Yes | Yes |
Obligate Anaerobe | No | No |
Facultative Anaerobe | Yes/No | Yes |
Aerotolerant Anaerobe | No | Yes |
Microaerophile | Low | Yes (low) |
Microbial Nutritional Requirements
Microbes require essential nutrients, growth factors, and energy sources for survival and proliferation. Nutrients are classified as macronutrients (needed in large amounts) and micronutrients (trace elements).
Heterotrophs: Require organic carbon sources.
Autotrophs: Use inorganic carbon (CO2) via carbon fixation.
Growth factors: Essential organic molecules that microbes cannot synthesize (e.g., amino acids, vitamins).
Fastidious organisms: Require multiple growth factors and enriched media.

Culture Media and Microbial Growth
Microbes are grown in various types of media, classified by physical state, chemical composition, and function.
Physical state: Liquid (broth), solid (agar plates), semisolid (motility testing).
Chemical composition: Defined (synthetic) media have known components; complex (enriched) media contain unknown quantities of nutrients.
Function: Differential media distinguish microbes visually; selective media favor the growth of specific microbes.
Media Type | Example | Purpose |
|---|---|---|
Defined | Glucose Minimal Salts | Known composition, for autotrophs/heterotrophs |
Complex | Luria–Bertani | Unknown composition, for fastidious organisms |
Microbial Enumeration Methods
Counting microbes is essential in clinical, industrial, and research settings. Methods are classified as direct or indirect.
Direct methods: Microscopic count, Coulter counter, flow cytometry, viable plate count (CFU/mL).
Indirect methods: Turbidity measurement (spectrophotometry), dry weight, biochemical activity.
Method | Description |
|---|---|
Microscopic count | Manual count using microscope |
Coulter counter | Automated, counts cells passing through tube |
Flow cytometer | Laser-based, differentiates cell types |
Viable plate count | Counts colonies from diluted samples |
Turbidity | Measures culture cloudiness |
Decontamination and Microbial Control
Decontamination strategies aim to reduce or eliminate microbial contamination. Methods include physical (temperature, radiation, filtration) and chemical (germicides) controls.
Sterilization: Eliminates all microbes, including endospores.
Disinfection: Reduces microbial numbers on surfaces and equipment.
Physical methods: Autoclaving, boiling, pasteurization, dry heat, radiation, filtration.
Chemical methods: Germicides (disinfectants, antiseptics) classified by efficacy (low, intermediate, high).
Germicide | Level | Mode of Action | Pros/Cons |
|---|---|---|---|
Alcohols | Intermediate | Denature proteins, disrupt membranes | Cheap, flammable |
Aldehydes | High | React with proteins, nucleic acids | Toxic, sterilant |
Phenols | Intermediate | Disrupt cell walls, proteins | Irritant, residue |
Halogens | High | Oxidize cell components | Corrosive, inactivated by organics |
Peroxygens | High | Oxidize proteins, nucleic acids | Corrosive, effective sterilant |
Ethylene oxide | High | Damages proteins, nucleic acids | Toxic, for heat-sensitive items |
Special Considerations in Microbial Control
Different microbes require specific control strategies due to their unique resistance mechanisms.
Mycobacterium: Waxy cell wall; requires airborne control and strong disinfectants.
Endospores: Highly resistant; best eliminated by autoclaving or sporicides.
Viruses: Enveloped viruses are sensitive to detergents; naked viruses require chlorine-based agents.
Protozoa: Resistant life stages; require filtration, UV, or ozone treatments.
Prions: Extremely resistant; require combined chemical and physical treatments.
Clinical Application: Case Study
Case studies highlight the importance of proper decontamination and aseptic technique in preventing healthcare-associated infections. For example, improper handling of piercing tools and antiseptics can lead to outbreaks of Pseudomonas aeruginosa infections.
Aseptic technique: Prevents contamination during clinical procedures.
Sample collection: Requires sterile materials and proper handling to maintain sample integrity.
Streak plate technique: Used to isolate pure colonies for identification.
