 Back
BackChapter 7 Fundamentals of Microbial Growth and Decontamination
Study Guide - Smart Notes

Fundamentals of Microbial Growth
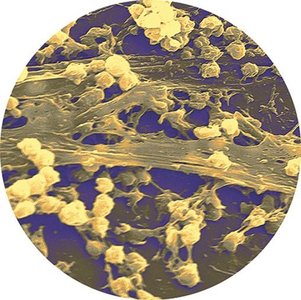
Microbial Growth in Nature and Biofilms
Microbial growth refers to the increase in cell number through cell division, resulting in new daughter cells and a larger population. While laboratory studies often use pure cultures, microbes in nature interact with other species and environmental factors, forming complex communities such as biofilms. Biofilms are structured microbial communities that adhere to surfaces and are difficult to treat, especially in healthcare settings where they contribute to persistent infections.
Biofilm formation: Begins when planktonic bacteria adhere to surfaces, such as catheters or heart valves.
Clinical relevance: Biofilms are resistant to treatment and can cause blood infections.
Example: Pseudomonas aeruginosa and Staphylococcus aureus are common biofilm-forming pathogens.
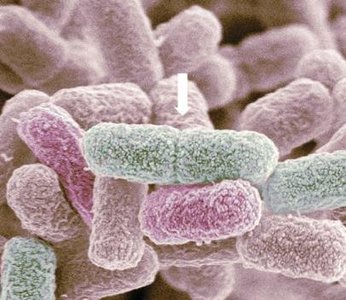
Methods of Microbial Cell Division
Bacteria primarily divide by binary fission, an asexual process resulting in two genetically identical daughter cells. Some microbes use budding or spore formation as alternative reproductive strategies.
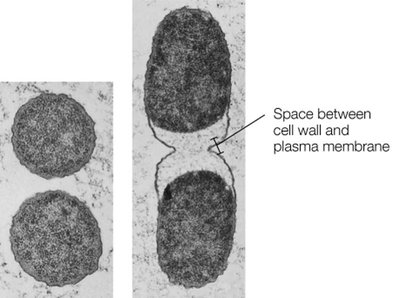
Binary fission: Chromosome replication, cell pinching, septum formation, and separation.
Budding: Cell elongates, forms a bud, duplicates chromosome, and separates. Common in certain fungi and bacteria.
Spore formation: Some fungi and bacteria produce spores for reproduction or survival. Bacterial spores differ from endospores, which are dormant and resistant structures.
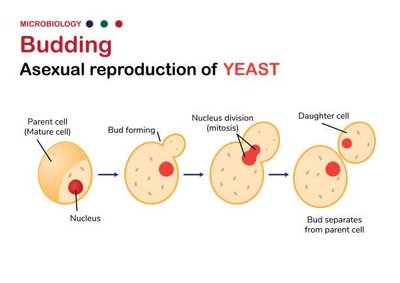
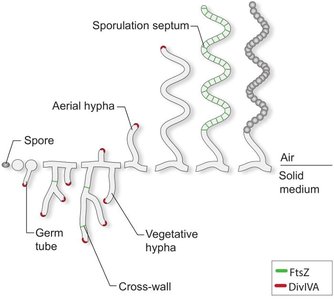
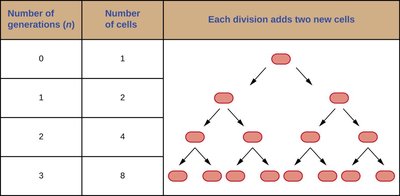
Generation Time and Exponential Growth
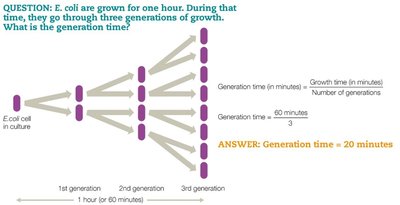
Generation time is the period required for a cell to divide. Bacterial populations grow exponentially, with generation times varying by species and environmental conditions.
Generation time: E. coli divides every 20 minutes; Mycobacterium tuberculosis takes 15–20 hours.
Formula:
Bacterial Growth Phases in Closed Batch Systems
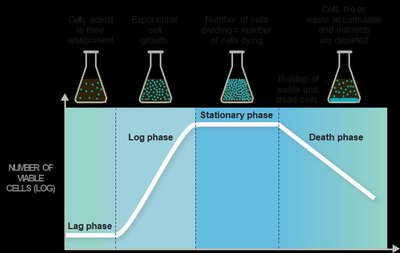
When cultured in closed systems, bacteria progress through four distinct growth phases: lag, log, stationary, and death. Each phase is influenced by species, conditions, and inoculum size.
Lag phase: Cells adjust to environment and alter gene expression.
Log phase: Rapid exponential growth; nutrients are abundant.
Stationary phase: Nutrients deplete, waste accumulates, growth rate levels off.
Death phase: Cells die exponentially as conditions deteriorate.
Industrial Maintenance of Growth Phases
Industries often use chemostats to maintain cells at specific growth phases for optimal production of products like ethanol or pharmaceuticals. Chemostats continuously add fresh medium and remove waste and excess cells.

Environmental Factors Affecting Microbial Growth
Temperature
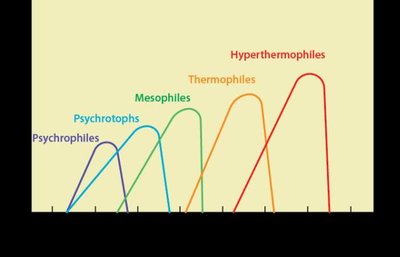
Temperature affects enzymatic reactions and microbial growth rates. Microbes are classified by their preferred temperature ranges:
Psychrophiles: Thrive at -20°C to 10°C.
Psychrotrophs: Grow at 0–30°C; associated with foodborne illness.
Mesophiles: Grow best at 10–50°C; most pathogens.
Thermophiles: Grow at 40–75°C; found in compost and hot springs.
Extreme thermophiles: Grow at 65–120°C; adapted to high pressure and temperature.

pH
Microbes are classified by their optimal pH ranges:
Neutralophiles: pH 5–8; majority of microorganisms.
Alkaliphiles: pH 9–11; found in soda lakes.
Acidophiles: pH 1–5; live in sulfur springs and volcanic vents.

High-Salt Conditions
Halophiles thrive in high-salt environments, overcoming osmotic stress by maintaining high concentrations of organic materials and ions in their cytoplasm. Facultative halophiles tolerate higher salt but may not grow well.
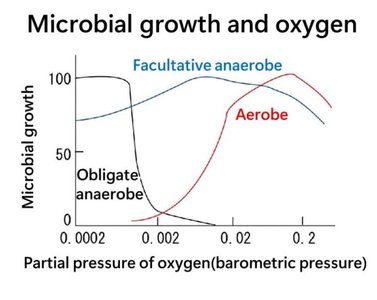
Oxygen Requirements
Microbes vary in their oxygen requirements and tolerance. Oxygen can generate reactive oxygen species (ROS) that damage cellular components. Microbes have evolved mechanisms to detoxify ROS.
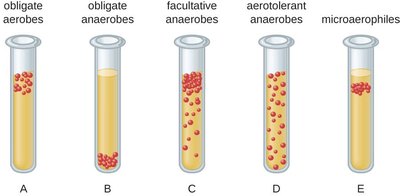
Obligate aerobes: Require oxygen for metabolism.
Microaerophiles: Use small amounts of oxygen.
Facultative anaerobes: Can grow with or without oxygen.
Anaerobes: Do not use oxygen.
Aerotolerant anaerobes: Tolerate oxygen but do not use it.
Obligate anaerobes: Cannot survive in oxygen.
Classification | Oxygen Use | ROS Management |
|---|---|---|
Obligate Aerobe | Absolute dependence | Yes |
Obligate Anaerobe | Not used | No |
Microaerophile | Small amounts | Yes (low amounts) |
Aerotolerant Anaerobe | Not used | Yes |
Facultative Anaerobe | Prefer oxygen, can survive without | Yes |
Microbial Nutritional Requirements
Essential Nutrients
Microbes require macronutrients (e.g., carbon, hydrogen, nitrogen, oxygen) and micronutrients (e.g., iron, zinc) to build cells and enzymes. They are classified by how they obtain organic carbon:
Heterotrophs: Require external organic carbon sources.
Autotrophs: Use carbon fixation to convert inorganic carbon into organic carbon.

Growth Factors
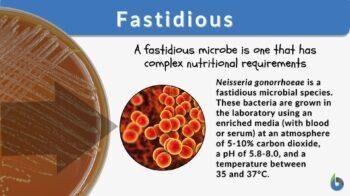
Some microbes cannot synthesize all their organic precursors and must import them from the environment. These substances are called growth factors. Fastidious organisms require multiple growth factors and need enriched media for laboratory growth.
Energy Sources
Phototrophs: Use light energy.
Chemotrophs: Break down chemical compounds for energy.
Microbial Culture Media
Physical State of Media
Media can be liquid, solid, or semisolid, depending on the application. Broth media are used for large batches, solid media for isolating colonies, and semisolid media for motility testing.
Chemical Composition: Complex vs Defined Media
Media are classified as defined (precisely known composition) or complex (contains unknown quantities of nutrients).
Type | Example | Ingredients |
|---|---|---|
Complex Media | Luria–Bertani | Tryptone, yeast extract, NaCl |
Defined Media | Glucose Minimal Salts | Glucose, MgSO4, CaCl2, Na2HPO4, KH2PO4, NH4Cl, NaCl |
Function: Differential and Selective Media
Differential media visually distinguish microbes, while selective media foster growth of certain bacteria and suppress others.
Blood agar: Differentiates hemolytic activity.
Mannitol salt agar: Selective for salt-tolerant bacteria; differentiates based on mannitol fermentation.
Eosin methylene blue agar: Selective for Gram-negative bacteria; differentiates based on lactose fermentation.
Anaerobic Media
Anaerobic media remove molecular oxygen using reducing agents like thioglycate or specialized chambers and jars.
Microbial Enumeration and Identification
Counting Microbes
Microbial counts are important for quality control and clinical diagnostics. Methods are classified as direct or indirect.
Method | Description |
|---|---|
Microscopic count | Manual count using microscope and chamber |
Coulter counter/Flow cytometer | Automated cell counting; flow cytometer distinguishes cell types |
Viable plate count | Colony count from diluted sample |
Turbidity measurement | Spectrophotometer measures cloudiness |
Dry weight | Measures dry mass |
Biochemical activity | Measures byproducts of cell growth |
Microbial Identification
Microbes are identified using physical, biochemical, and genetic analyses. Staining and microscopy reveal morphology, biochemical tests assess metabolic properties, and genetic methods (PCR, DNA fingerprinting) provide rapid identification.
Microbial Decontamination and Control
Decontamination, Sterilization, and Disinfection
Decontamination reduces microbial populations. Sterilization eliminates all microbes, while disinfection reduces numbers. Control measures are essential in healthcare, food, and water sanitation.
Physical Methods of Control
Temperature: Refrigeration slows growth; heat sterilizes or decontaminates.
Radiation: Ionizing (gamma, X-rays) and nonionizing (UV) radiation damage nucleic acids.
Filtration: Removes microbes from air and liquids using filters.
Chemical Methods: Germicides
Germicides are chemicals that kill or inhibit microbes. They are classified as disinfectants (inanimate objects) or antiseptics (living tissue), and ranked as low, intermediate, or high-level agents.
Level | Germicide | Mode of Action | Pros/Cons |
|---|---|---|---|
Low | Detergents | Target lipid membranes | Cheap, low toxicity; activity decreased in hard water |
Intermediate | Alcohols | Target proteins and lipid membranes | Cheap, flammable |
Intermediate | Phenols | Target proteins and lipid membranes | Effective in hard water, irritant |
High | Aldehydes | Target proteins, nucleic acids | Toxic, achieve sterility |
High | Halogens | Oxidizing agents | Sterilants, corrosive |
High | Peroxygens | Oxidizing agents | Effective, corrosive |
High | Ethylene oxide | Targets proteins, nucleic acids | Gentle, toxic, flammable |
Medical Equipment Decontamination Tiers
Critical equipment: Must be sterilized; contacts sterile body sites.
Semicritical equipment: Should be free of microbes; contacts mucous membranes or non-intact skin.
Noncritical equipment: Requires less stringent disinfection; contacts intact skin.
Microbial Control for Specific Pathogens
Mycobacterium: Control airborne particles; waxy cell walls resist many treatments.
Endospores: Autoclaving is most effective; resistant to many treatments.
Viruses: Envelope viruses sensitive to heat and detergents; naked viruses inactivated by chlorine.
Protozoa: Resistant stages require filtration, UV, or ozone.
Prions: Require combined chemical and autoclave treatments.
Clinical Case Study: Piercing-Related Infection
Case Summary
Three patients developed upper ear cartilage infections after piercings at a mall kiosk. The causative agent was Pseudomonas aeruginosa. Oral antibiotics were ineffective due to poor blood flow in cartilage, requiring surgical removal.
Investigation: Health department sampled surfaces and items at the kiosk.
Safety procedures: Included hand sanitization, gloves, and antiseptic cleaning.
Possible transmission: Contaminated surfaces, tools, or antiseptic could have contributed.
Culture methods: Selective and differential media (e.g., EMB, MSA) differentiate P. aeruginosa from S. aureus. Anaerobic conditions not required for these pathogens.
Antiseptic role: If contaminated or expired, could facilitate pathogen transmission.
Decontamination tier: Piercing tool is critical equipment; sterilization recommended.
Prevention: Use fresh antiseptics, proper sterilization, and aseptic techniques.
Lower earlobe case: Likely did not develop infection due to better blood flow and healing.
