 Back
BackHost Resistance to Viral Infection: Mechanisms and Immune Responses
Study Guide - Smart Notes

Host Resistance to Viral Infection
Overview of Host Resistance
Host resistance to viral infection refers to the ability of a host organism to limit or prevent viral replication, spread, and disease. This resistance is achieved through a multi-layered defense system, including both innate (nonspecific) and adaptive (specific) immunity. The effectiveness of these mechanisms varies among species, individuals, and populations, and is influenced by genetic, immunological, and environmental factors.
Resistance: Reduces viral pathogen burden.
Tolerance: Minimizes damage caused by infection without necessarily reducing viral load.
Mechanisms of Host Resistance
Nonspecific Host Defenses (Innate Immunity)
Innate immunity provides the body's first line of defense against any pathogen, regardless of its type. It does not retain memory from prior encounters and acts rapidly upon infection.
Physical and Chemical Barriers: Skin, mucus, and saliva prevent viruses from entering the body.
Cellular and Molecular Responses: Includes interferons, natural killer (NK) cells, and antigen-presenting cells (APCs).
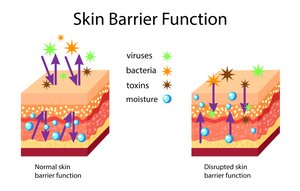
Physical and Chemical Barriers
The skin acts as a robust barrier, preventing entry of viruses, bacteria, toxins, and moisture. Disruption of this barrier increases susceptibility to infection.
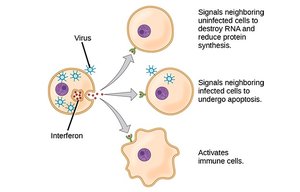
Interferons
Interferons are signaling proteins released by infected cells to alert neighboring cells and inhibit viral replication. They establish an antiviral state and activate immune cells.
Interferons signal uninfected cells to destroy viral RNA and reduce protein synthesis.
They induce apoptosis in infected cells.
They activate immune cells to enhance antiviral responses.
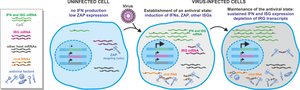
Cellular Antiviral Defenses: Induction of Interferon Stimulated Genes (ISGs)
Upon viral detection, infected cells produce interferons, which signal neighboring cells to upregulate ISGs. These genes encode proteins that interfere with various stages of the viral life cycle, such as degrading viral RNA and inhibiting viral protein synthesis. 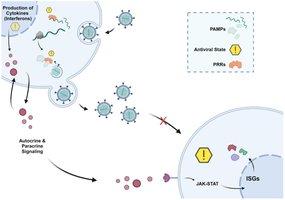
Natural Killer (NK) Cells
NK cells are a critical component of innate immunity, directly killing infected cells and releasing pro-inflammatory cytokines.
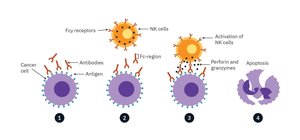
Cytotoxic Killing: NK cells release perforin and granzymes to induce cell lysis.
Antibody-Dependent Cell-Mediated Cytotoxicity (ADCC): NK cells bind to antibody-coated infected cells, leading to their destruction.
Cytokine Production: NK cells produce cytokines to coordinate immune responses and limit viral replication.
Tissue Damage: Excessive NK cell activity can cause tissue damage, highlighting the need for balance.
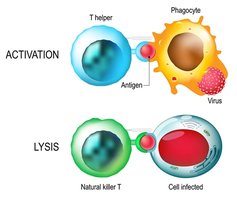
Antigen-Presenting Cells (APCs)
APCs, such as dendritic cells and macrophages, act as messengers between the innate and adaptive immune systems. They internalize viruses, digest them, and present viral peptides on MHC molecules to activate T cells. 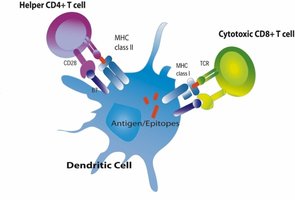
Adaptive Immunity
Specific Immune System Responses
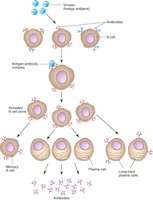
Adaptive immunity is slower to develop but highly specific to the particular virus. It involves B cells and T cells, which provide immunological memory for rapid response upon re-exposure.
B Cells: Produce antibodies that neutralize viruses.
T Cells: Include helper T cells (coordinate responses) and cytotoxic T cells (kill infected cells).
Immunological Memory: Memory B and T cells enable faster and stronger responses to subsequent infections.
Time Course of Immune Responses
Innate defenses act rapidly after infection, while adaptive responses (antibodies) require days to weeks to develop. 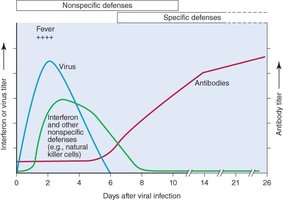
Humoral Response: Antibody Production
B cells produce antibodies that target and neutralize viruses.
Neutralization: Antibodies bind to viruses or their antigens, preventing entry into host cells.
Blocking Attachment: Neutralizing antibodies block viral surface proteins from attaching to host cell receptors.
Inhibiting Entry: Antibodies prevent fusion of viral and host cell membranes.
Opsonization and Phagocytosis
Antibodies coat viral particles (opsonization), marking them for destruction by phagocytic cells such as macrophages. 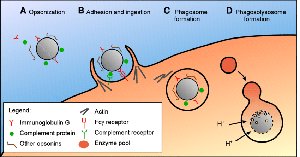
Antibody-Dependent Cellular Cytotoxicity (ADCC)
Antibodies bind to viral antigens on infected cells, attracting NK cells that induce apoptosis via cytotoxic molecules. 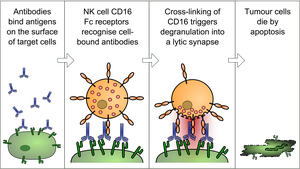
T Cell-Mediated Responses
T cells attack viruses that have already infected host cells.
Cytotoxic T Cells (CD8+): Identify and destroy infected cells displaying viral antigens on MHC class I molecules.
Helper T Cells (CD4+): Coordinate immune responses, activate B cells, and enhance cytotoxic T cell activity.
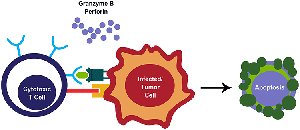
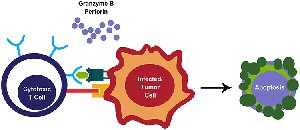
Cytotoxic T Cell Mechanism
Antigen Recognition: Cytotoxic T cells bind to viral antigens presented on MHC class I molecules.
Targeted Killing: Release perforin and granzymes to induce apoptosis in infected cells.
Viral Elimination: Apoptosis destroys infected cells and prevents release of new viral particles.
Helper T Cell Functions
Antigen Presentation: Recognize viral antigens presented by APCs on MHC class II molecules.
Activating B Cells: Help B cells mature and produce neutralizing antibodies.
Enhancing Cytotoxic T Cells: Secrete cytokines to promote proliferation and maturation of cytotoxic T cells.
Boosting Innate Immunity: Release cytokines to activate macrophages and NK cells.
Factors Influencing Resistance
Genetic Factors
Genes within the major histocompatibility complex (MHC) significantly influence immune response and resistance to viruses.
Physiological Factors
Age, nutritional status, stress, and underlying health conditions impact resistance to viral infections.
Co-evolution
Viruses evolve to evade host defenses, resulting in an ongoing evolutionary arms race between host and pathogen.
Distinction from Host Tolerance
Resistance | Tolerance |
|---|---|
Reduces viral pathogen burden | Minimizes tissue damage without reducing viral load |
Clears virus from body | Allows virus to persist but limits harm |
Example: The body may minimize tissue damage even if the virus remains present. Additional info: These mechanisms are central to understanding viral pathogenesis and host defense, and are directly relevant to microbiology topics such as innate and adaptive immunity, viral infection, and immunopathology.
