 Back
BackHost–Microbe Interactions and Pathogenesis: Principles and Clinical Applications
Study Guide - Smart Notes

Host–Microbe Interactions
Normal Microbiota and Their Roles
Normal microbiota are the diverse community of microorganisms that inhabit various regions of the human body, including the skin, digestive tract, genital, urinary, and respiratory systems. These microbes typically exist in a mutualistic or commensal relationship with the host, contributing to health and homeostasis.
Mutualistic relationships: Microbes manufacture vitamins, compete with potential pathogens, and promote immune system maturation.
Disruption of microbiota: Antibiotic therapy or illness can disturb the balance, leading to dysbiosis and increased susceptibility to opportunistic infections, such as Clostridium difficile overgrowth in the gut.
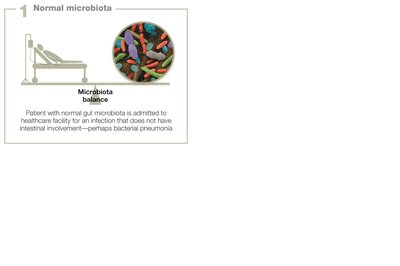
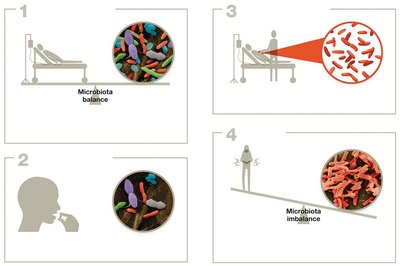
Opportunistic Pathogens
Opportunistic pathogens are typically harmless but can cause disease when the host's defenses are compromised or when they access unusual body sites.
Normal microbiota vs. opportunistic pathogens: Both reside in or on the body; normal microbiota are usually beneficial or neutral, while opportunists cause disease under specific conditions (e.g., immune suppression, anatomical disruption).
Example: Group B streptococci (GBS) are harmless in many women but can cause severe neonatal infections if transmitted during birth.
Host Factors and Pathogenicity
The outcome of host–microbe interactions depends on host factors such as immune status, anatomical barriers, and genetic predisposition. A microbe harmless in one individual may be pathogenic in another.
Pathogenesis and Virulence
Definitions and Concepts
Pathogenicity: The ability of a microbe to cause disease (all-or-nothing concept).
Virulence: The degree or severity of disease caused by a pathogen.
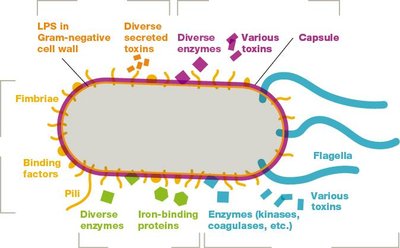
Virulence factors: Molecules or structures that enable pathogens to colonize, invade, evade immune responses, and damage the host (e.g., toxins, adhesins, capsules).
Attenuation: The process by which pathogens lose virulence factors and become less capable of causing disease, often used in vaccine development.
Infectious Dose and Lethal Dose
Infectious dose-50 (ID50): The number of microbes required to infect 50% of exposed hosts. Lower ID50 indicates higher infectivity.
Lethal dose-50 (LD50): The amount of toxin needed to kill 50% of affected hosts. Lower LD50 means higher toxicity.
Example: Botulinum toxin has a much lower LD50 than Salmonella toxin, making it more potent.
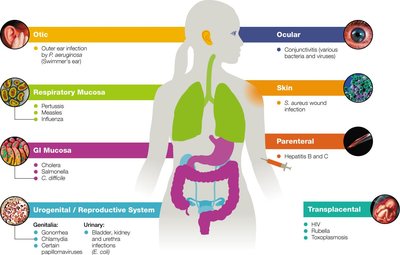
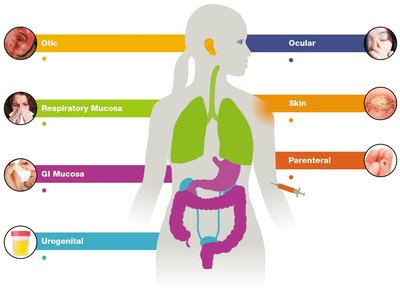
Portals of Entry
Pathogens enter the host through specific portals, which influence the infectious dose and disease manifestation.
Skin (cutaneous): Entry via cuts, abrasions, or direct penetration.
Respiratory tract: Most common portal; pathogens enter via inhalation of droplets or dust.
Gastrointestinal tract: Entry via ingestion of contaminated food or water.
Genitourinary tract: Entry via mucosal surfaces or direct invasion.
Parenteral route: Entry via injections, bites, or wounds.
Transplacental: Vertical transmission from mother to fetus.
Toxins and Their Effects
Endotoxins vs. Exotoxins
Toxins are microbial products that damage host tissues or disrupt normal physiology.
Property | Endotoxins | Exotoxins |
|---|---|---|
Composition | Lipid (Lipid A of LPS) | Protein |
Source | Gram-negative bacteria | Gram-positive and Gram-negative bacteria |
Release | Upon cell death/division | Actively secreted |
Fever | Yes | Sometimes |
Neutralization | No | Yes (some) |
Vaccines | No | Yes (some) |
Toxicity | Lower (high LD50) | Higher (low LD50) |
Endotoxins: Lipid A component of LPS in Gram-negative bacteria; released upon cell death, can cause septic shock.
Exotoxins: Secreted proteins with diverse targets and mechanisms; classified as neurotoxins, enterotoxins, etc.
Types of Exotoxins
Type I: Bind to host cell membranes (e.g., superantigens).
Type II: Disrupt host cell membranes (e.g., hemolysins).
Type III: Enter host cells and disrupt intracellular functions (e.g., diphtheria toxin).
Fungal Toxins
Ergot toxin: Produced by Claviceps purpurea, causes hallucinations.
Aflatoxin: Produced by Aspergillus flavus, contaminates peanuts and is carcinogenic.
Mycotoxins: Neurotoxins from mushrooms such as Amanita phalloides (death cap).


Steps of Infection
Five Steps to Infection
Entry into the host
Adherence to host tissues
Invasion and nutrient acquisition
Evasion of host immune defenses
Transmission to a new host
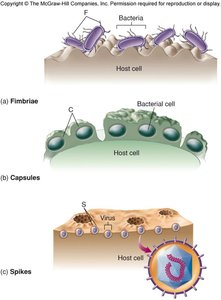
Adhesion Factors
Adhesins are molecules that enable pathogens to attach to host cells. These include fimbriae, pili, capsules, and specific membrane proteins.
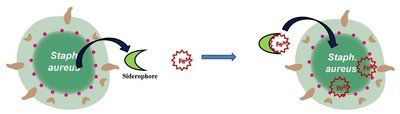
Invasion and Nutrient Acquisition
Pathogens invade tissues using enzymes (invasins) and acquire nutrients through mechanisms such as siderophore production and extracellular enzyme secretion.
Invasin | Mechanism | Examples |
|---|---|---|
Flagella | Motility, adhesion | E. coli, Vibrio vulnificus |
Collagenases | Break down collagen | Clostridium perfringens |
Neuraminidases | Break down neuraminic acid | Vibrio cholerae, Influenza virus |
Coagulases | Promote blood clotting | Staphylococcus aureus |
Kinases | Break down blood clots | Streptococcus pyogenes |
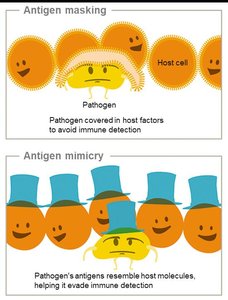
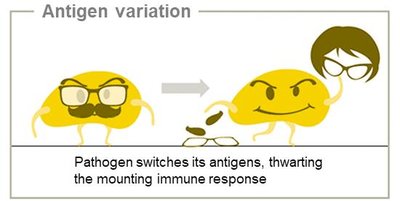
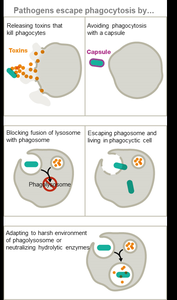
Immune Evasion Strategies
Mechanisms for Escaping Host Immune Defenses
Hiding: Intracellular lifestyle, antigen masking, mimicry, and variation, latency.
Undermining: Suppressing immune function, avoiding phagocytosis, neutralizing immune factors.
Transmission and Exit
Portals of Exit
Pathogens exit the host via specific routes, often the same as their entry portals. Common portals include feces, urine, blood, respiratory droplets, and bodily fluids.
Biosafety and Infection Control
Biosafety Levels (BSL)
Biosafety levels are standardized protocols for handling infectious agents in laboratory and healthcare settings, ranging from BSL-1 (lowest risk) to BSL-4 (highest risk).
Level | PPE Required | Facility Considerations |
|---|---|---|
BSL-1 | None | Hand-washing sinks, open bench work |
BSL-2 | Lab coat, gloves, eye protection | Biohazard signage, restricted access, autoclave |
BSL-3 | Protective covering, gloves, respirators | Biological safety cabinets, controlled access, special airflow |
BSL-4 | Full-body suit, air supply | Specialized facility, highly restricted access |

Infection Control Practices
Standard precautions: Universal measures to prevent transmission of bloodborne and other pathogens (e.g., hand hygiene, gloves, barrier clothing, disinfection).
Transmission precautions: Additional measures for known or suspected infectious agents, including contact, droplet, and airborne precautions.
Biofilms and Quorum Sensing
Biofilms are structured communities of microbes attached to surfaces, protected by an extracellular matrix. They are common sources of persistent infections, especially on medical devices and tissues.
Examples: Catheters, prosthetic joints, dental plaque, kidney stones.
Clinical significance: Biofilms are resistant to antibiotics and immune responses, complicating treatment.
Summary Table: Key Concepts
Concept | Definition/Example |
|---|---|
Pathogenicity | Ability to cause disease |
Virulence | Severity of disease |
Virulence factors | Toxins, adhesins, enzymes, capsules |
ID50 | Infectious dose for 50% of hosts |
LD50 | Lethal dose for 50% of hosts |
Biofilm | Microbial community on surfaces |
BSL | Biosafety level for handling pathogens |
