 Back
BackHost–Microbe Interactions and Pathogenesis: Principles and Mechanisms
Study Guide - Smart Notes

Host–Microbe Interactions and Pathogenesis
Introduction
This chapter explores the complex relationships between hosts and microbes, focusing on the mechanisms by which microbes establish infection, evade host defenses, and cause disease. Key concepts include the role of the normal microbiota, virulence factors, steps of infection, and strategies for transmission control.
Microbiota
Normal Microbiota and Their Roles
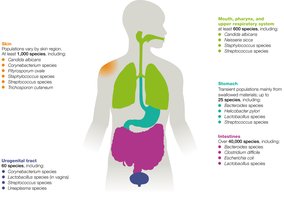
Normal microbiota colonize the skin, digestive, genital, urinary, and respiratory systems.
They manufacture vitamins, compete with pathogens, and promote immune system maturation.
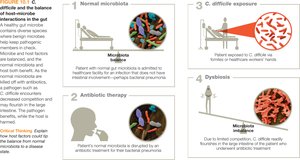
Dysbiosis: Disruption of the microbiota, which can compromise health and lead to disease.
Microbiota and Disease
Microbiota composition varies between individuals; a harmless species in one host may be pathogenic in another.
Example: Group B streptococci (GBS) are normal in ~30% of women but can cause severe disease in newborns.
The immune system maintains a balanced response to resident microbes.
Opportunistic pathogens: Normally harmless microbes that cause disease under certain conditions (e.g., E. coli outside the gut, yeast infections in immunocompromised hosts).
Virulence
Pathogenicity and Virulence
Pathogenicity: The ability of a microbe to cause disease.
Virulence: The degree or extent of disease caused by a pathogen.
Tropism: Pathogen preference for specific hosts or tissues, which can change over time.
Host factors (age, health, habits) influence disease development.
Virulence Factors
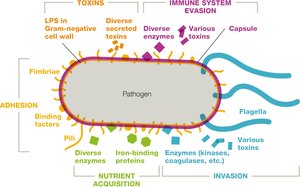
Virulence factors help pathogens overcome host defenses and cause damage by:
Directly damaging host cells
Provoking harmful immune responses
Examples: Toxins, enzymes, adhesins, immune evasion mechanisms.
Pathogens balance virulence to persist in populations; highly lethal pathogens may cause isolated outbreaks.
Virulence factors are often linked to transmission strategies (e.g., STIs are often minimally symptomatic).
Pathogens may lose virulence factors (attenuation) when grown in culture, which is used in vaccine development.
Measuring Infectivity and Toxicity
Infectious dose-50 (ID50): Number of cells/virions needed to infect 50% of hosts; lower ID50 means higher infectivity.
Lethal dose-50 (LD50): Amount of toxin needed to kill 50% of hosts; lower LD50 means higher toxicity.


Toxins
Toxins: Molecules that cause adverse effects (e.g., tissue damage, immune suppression).
Toxigenic: Microbes that produce toxins.
Toxemia: Presence of toxins in the bloodstream.
Endotoxins vs. Exotoxins
Endotoxins: Lipid A component of lipopolysaccharide (LPS) in Gram-negative bacteria; released when bacteria die or divide.
Exotoxins: Soluble proteins secreted by both Gram-positive and Gram-negative bacteria; highly toxic and specific in action.
Structure of LPS
LPS consists of O antigen, core polysaccharide, and Lipid A (endotoxin).
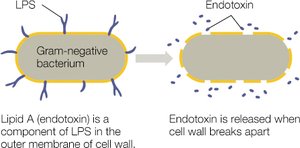
Endotoxemia and Clinical Impact
Endotoxemia: Endotoxin in the bloodstream, often from Gram-negative infections or medical procedures.
Symptoms: Fever, chills, hypotension, tachycardia, inflammation, organ failure (septic shock).
Endotoxins are not easily neutralized and no vaccines exist; contamination prevention is critical in healthcare.
Exotoxin Classification
Exotoxins are classified by their target or action:
Neurotoxins: Affect the nervous system
Enterotoxins: Target the GI tract
Hepatotoxins: Affect the liver
Nephrotoxins: Damage the kidneys
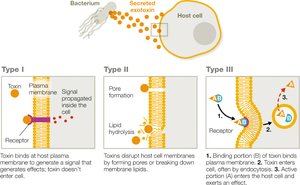
Three main families based on mode of action:
Type I: Membrane-acting extracellular toxins (alter cell signaling)
Type II: Membrane-damaging toxins (form pores or disrupt membranes)
Type III: Intracellular toxins (AB toxins; enter cells and disrupt function)
Steps of Infection
Overview of Infection Process
To establish infection, a pathogen must:
Enter the host
Adhere to host tissues
Invade tissues and obtain nutrients
Replicate while evading immune defenses
Transmit to a new host
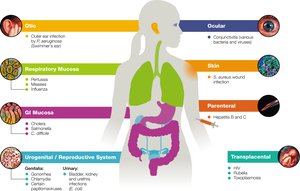
Portal of Entry
Common portals: Mucous membranes (respiratory, GI, urogenital), skin, conjunctiva, parenteral routes (bites, cuts, injections).
Respiratory tract is the most common portal of entry.
Adherence to Host Tissues
Pathogens use adhesins (e.g., fimbriae, pili, capsules) to attach to host cells.
Biofilms play a major role in chronic infections and device-associated infections.
Invasion and Nutrient Acquisition
Pathogens may remain on the surface, invade deeper tissues, or become intracellular.
Invasins: Enzymes that help pathogens invade tissues (e.g., collagenases, coagulases).
Pathogens compete for nutrients, especially iron, using molecules like siderophores.
Extracellular enzymes (lipases, proteases) break down host tissues for nutrients.
Pathogens can cause cytopathic effects: cell death (cytocidal) or cell damage (noncytocidal).
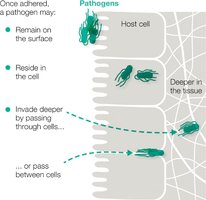
Immune Evasion and Replication
Pathogens evade the immune system by:
Hiding inside host cells (intracellular pathogens)
Latency (e.g., herpesviruses, HIV)
Antigenic masking, mimicry, and variation
Interfering with phagocytosis or immune signaling
Suppressing immune responses (e.g., proteases that degrade antibodies)
Transmission to New Hosts
Portal of exit: Route used by pathogens to leave the host (e.g., feces, urine, blood, saliva, respiratory droplets).
Reservoirs: Environmental sources, animals, humans, or fomites.
Transmission Control
Biosafety Levels (BSL)
BSL-1: Minimal risk, standard precautions (e.g., Bacillus subtilis).
BSL-2: Moderate risk, not airborne, includes most clinical pathogens (e.g., Staphylococcus aureus).
BSL-2+: Dangerous, incurable, not airborne (e.g., HIV).
BSL-3: Serious/lethal, often airborne (e.g., Mycobacterium tuberculosis).
BSL-4: High risk, exotic, no treatment (e.g., Ebola virus).
Infection Control Practices
Universal and standard precautions: Hand hygiene, gloves, barrier clothing, proper waste management, and disinfection.
Transmission precautions: Additional measures for contact, droplet, or airborne pathogens (e.g., isolation rooms, masks, limited transport).
Tables
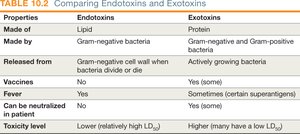
Comparing Endotoxins and Exotoxins
Properties | Endotoxins | Exotoxins |
|---|---|---|
Made of | Lipid | Protein |
Made by | Gram-negative bacteria | Gram-negative and Gram-positive bacteria |
Released from | Cell wall when bacteria divide or die | Actively growing bacteria |
Vaccines | No | Yes (some) |
Fever | Yes | Sometimes (certain superantigens) |
Can be neutralized in patient | No | Yes (some) |
Toxicity level | Lower (relatively high LD50) | Higher (many have a low LD50) |

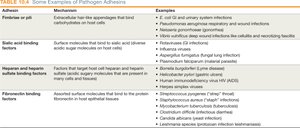
Examples of Pathogen Adhesins
Adhesin | Mechanism | Examples |
|---|---|---|
Fimbriae or pili | Bind carbohydrates on host cells | E. coli (GI/urinary infections) |
Sialic acid binding factors | Bind to sialic acid on host cells | Influenza virus |
Heparan and heparin sulfate binding factors | Bind to heparan sulfate on host cells | Borrelia burgdorferi (Lyme disease) |
Fibronectin binding factors | Bind to fibronectin in host tissues | Staphylococcus aureus |
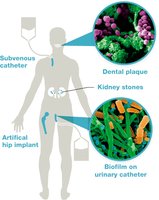
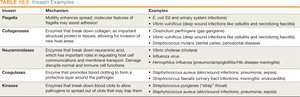
Examples of Invasins
Invasin | Mechanism | Examples |
|---|---|---|
Flagella | Motility, chemotaxis | E. coli, Pseudomonas aeruginosa |
Collagenases | Break down collagen | Clostridium perfringens |
Neuraminidases | Break down sialic acid | Influenza virus |
Coagulases | Promote clotting | Staphylococcus aureus |
Kinases | Break down clots | Streptococcus pyogenes |
Summary
Host–microbe interactions are dynamic and influenced by both microbial and host factors.
Virulence factors enable pathogens to infect, evade defenses, and cause disease.
Understanding these mechanisms is essential for infection control and clinical management.
