 Back
BackHost–Microbe Interactions and Pathogenesis: Study Notes
Study Guide - Smart Notes

Host–Microbe Interactions
Basics of Host–Microbe Interactions
Host–microbe interactions are dynamic relationships that can foster health or lead to disease. The human body is colonized by normal microbiota (flora) on the skin, digestive, genital, urinary, and respiratory systems. These microbes often have mutualistic relationships with the host, providing benefits such as vitamin synthesis, competition with pathogens, and immune system maturation.
Mutualism: Both host and microbe benefit from the relationship.
Dysbiosis: Disruption of the normal microbiota balance, which can compromise health and allow opportunistic pathogens to flourish.
Pathogens: Microbes with adaptations that allow them to cause disease in the host.
Host Factors and Microbiota
Host factors such as immune status, age, and genetics influence whether a microbe is harmless or pathogenic. For example, Group B Streptococcus (GBS) is a commensal in many women but can cause severe disease in newborns due to their underdeveloped immune systems.
Commensalism: One organism benefits while the other is unaffected.
Screening for GBS in pregnant women helps prevent neonatal infections.
Opportunistic Pathogens
Normal microbiota can become opportunistic pathogens if the host's immune defenses are compromised or if microbes translocate to normally sterile sites. For example, Escherichia coli is harmless in the gut but can cause life-threatening infection if it enters the abdominal cavity.
Opportunistic pathogens: Cause disease when host defenses are weakened or when they access new body sites.
Dysbiosis: Antibiotic use can disrupt microbiota, allowing organisms like Candida albicans to cause infections.
Tropism
Tropism refers to the preference of a pathogen for a specific host or tissue. This specificity influences which organisms and tissues a pathogen can infect. Tropism can evolve, leading to the emergence of new pathogens.
Example: Shigella flexneri infects human and primate intestinal tracts.
Host factors such as age, gender, and health status influence disease development.
Virulence and Pathogenicity
Definitions and Concepts
Pathogenicity is the ability of a microbe to cause disease, while virulence describes the degree of disease caused. Virulence factors are mechanisms that help pathogens overcome host defenses, adhere to cells, invade tissues, and acquire nutrients.
Virulence factors: Include adhesins, toxins, immune evasion strategies, invasins, and nutrient acquisition systems.
Virulence evolves in response to host and environmental pressures, such as antibiotic use.

Host Factors and Virulence
Host characteristics (immune fitness, microbiota balance) influence the impact of virulence factors. For example, the 1918 influenza pandemic was more virulent in young adults, while SARS-CoV-2 is often asymptomatic in children.
Transmission and Virulence
Virulence factors are often linked to transmission strategies. Pathogens that rely on direct transmission (e.g., STDs) tend to avoid causing severe symptoms that would incapacitate the host. The basic reproduction number () and effective reproduction number () are used to measure pathogen transmissibility.
: Average number of secondary infections from one case in a fully susceptible population.
: Number of secondary infections at a given time, accounting for immunity in the population.
Virulence as an Evolving Property
Virulence changes in response to host and environmental factors. Pathogens may become attenuated (weakened) when grown in laboratory conditions, losing virulence factors but remaining infectious. Attenuated strains are sometimes used in vaccines.
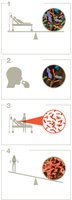
Dosage and Health Outcomes
The infectious dose-50 () is the number of cells or virions needed to infect 50% of exposed hosts. The lethal dose-50 () is the amount of toxin required to kill 50% of affected hosts. These values vary by species, immune status, and route of exposure.
Lower indicates higher infectivity.
Pathogenicity and virulence are distinct properties.
Toxins as Virulence Factors
Types of Toxins
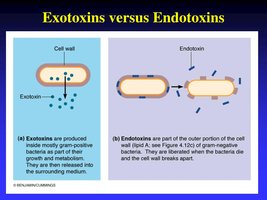
Toxins are molecules that cause adverse effects in the host, such as tissue damage and immune suppression. Toxigenic microbes produce toxins, and toxemia refers to toxins in the bloodstream. There are two major classes: endotoxins and exotoxins.
Endotoxins
Endotoxins are components of the outer membrane of Gram-negative bacteria, specifically the lipid A portion of lipopolysaccharide (LPS). They are released when bacteria die and can cause fever, inflammation, and septic shock.
Endotoxins are not found in Gram-positive bacteria.
Endotoxemia can result from localized or systemic infections, or surgical complications.
Endotoxins are not readily neutralized and there are no vaccines against them.
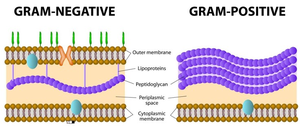
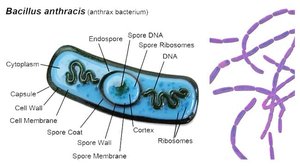
Exotoxins
Exotoxins are toxic proteins secreted by both Gram-positive and Gram-negative bacteria. They target specific cell types and are classified by their mode of action:
Type I: Membrane-acting extracellular toxins (e.g., superantigens) that bind to cell surface receptors and alter cell signaling.
Type II: Membrane-damaging toxins that disrupt plasma membranes, causing cell lysis.
Type III: Intracellular toxins (often AB toxins) that enter cells and disrupt cellular processes.
Steps to Infection
Five Steps to Infection
To establish infection, a pathogen must:
Enter the host
Adhere to host tissues
Invade tissues and obtain nutrients
Replicate while evading immune defenses
Transmit to a new host
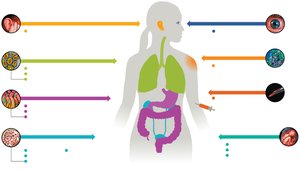
Portals of Entry
Pathogens enter the host through various portals, most commonly mucous membranes (respiratory, digestive, urogenital tracts). Other routes include skin, ocular, otic, parenteral (bypassing skin), and transplacental transmission.
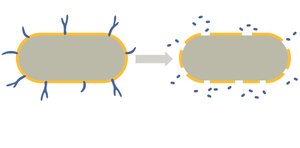
Adhesion to Host Tissues
After entry, pathogens must adhere to host tissues. Adhesins are virulence factors that mediate attachment, including cell wall components, capsules, fimbriae, and pili. Specificity for host cell receptors determines tissue tropism.
Biofilms and Quorum Sensing
Bacteria can form biofilms on natural and artificial surfaces, which enhances survival and virulence. Quorum sensing is a communication method that regulates gene expression in response to cell density, promoting biofilm formation and increased pathogenicity.
Invasion and Nutrient Acquisition
Pathogens invade tissues to obtain nutrients, causing cytopathic effects. They may remain on the surface, invade deeper tissues, or reside intracellularly. Invasins are structures or enzymes that facilitate tissue invasion, often by breaking down host barriers or inducing host uptake.
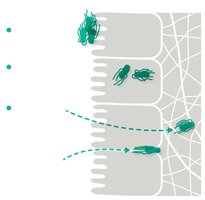
Motility: Flagella and other structures aid in spreading within the host.
Additional info: These notes are based on Chapter 10 of "Microbiology: Basic and Clinical Principles" and are suitable for college-level microbiology students studying host–microbe interactions and pathogenesis.
