 Back
BackHost–Microbe Interactions and Virulence Factors: Structured Study Notes
Study Guide - Smart Notes

Host–Microbe Interactions and Virulence Factors
Normal Biota and Microbial Antagonism
Normal biota, also known as resident flora, indigenous biota, or normal flora, refers to the diverse collection of microbes that colonize various regions of the human body, including the skin, digestive, genital, urinary, and respiratory systems. These microbes include bacteria, fungi, protozoans, and viruses, and their interactions with the host are often beneficial.
Benefits of normal biota:
Manufacture essential vitamins for the host
Compete with potential pathogens, preventing their overgrowth
Promote immune system maturation
Influence the development of organs
Microbial antagonism: The antagonistic effect of beneficial microbes against intruder microorganisms, helping to protect the host from infection.
Dysbiosis: Disruption of the normal microbiota balance, which can lead to disease (e.g., antibiotic use allowing Clostridioides difficile or Candida albicans to flourish).
Pathogens and Types of Pathogenic Relationships
A pathogen is a microbe whose relationship with its host is parasitic, resulting in infection and disease. Pathogens are classified as:
True pathogens: Infect healthy individuals.
Opportunistic pathogens: Cause disease when host defenses are compromised or when they colonize an unnatural site.
Tropism refers to the preference of a pathogen for a specific host or tissue. Disease outcome depends on host factors such as age, genetics, health status, and immune fitness.
Pathogenicity, Virulence, and Virulence Factors
Pathogenicity is the ability of a microbe to cause disease, while virulence describes the degree of disease caused. Virulence factors are substances or structures that enable microbes to establish infection and cause damage (e.g., adhesins, toxins).
Attenuated pathogens: Weakened pathogens that have lost virulence factors, often used in vaccines.
Infectious Dose and Lethal Dose
To establish disease, a pathogen must infect the host:
Infectious dose (ID50): Number of cells/virions needed to infect 50% of exposed hosts.
Lethal dose (LD50): Amount of toxin needed to kill 50% of affected hosts.
These measures vary by species, host immune status, and route of exposure.
Five Steps to Infection
Successful pathogens complete five general tasks to establish infection:
Enter the host via specific portals of entry (skin, mucous membranes, respiratory, gastrointestinal, urogenital, transplacental).
Adhere to host tissues using adhesins such as fimbriae, capsules, suckers, and spikes.
Invade tissues and obtain nutrients by secreting extracellular enzymes and toxins.
Replicate while evading immune defenses through antigen masking, mimicry, variation, latency, and immune suppression.
Transmission to a new host via symptoms that facilitate spread (e.g., coughing, sneezing, diarrhea).
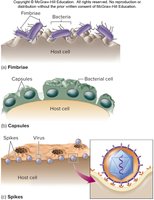
Step 2—Adhere to Host Tissues
Adhesion is crucial for establishing infection. Microbes use specific structures to bind to host cells:
Fimbriae and pili (e.g., Neisseria gonorrhoeae, E. coli)
Capsules (e.g., Streptococcus mutans, Streptococcus pneumoniae)
Spikes (e.g., influenza virus)
Biofilms are sticky communities that enhance microbial resistance and persistence on surfaces such as teeth, catheters, and implants.
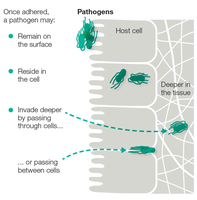
Step 3—Invade Tissues and Obtain Nutrients
Pathogens secrete enzymes and toxins to invade tissues and acquire nutrients:
Coagulase: Produces clots (e.g., Staphylococcus species)
Kinase: Dissolves clots (e.g., Staphylococcus, Streptococcus species)
Hyaluronidase: Breaks down hyaluronic acid in connective tissue (e.g., Streptococcus pyogenes, Clostridium perfringens)
Collagenase: Breaks down collagen (e.g., Clostridium perfringens)
Siderophores: Acquire iron from host proteins
Step 4—Replicate While Warding Off Immune Defenses
Pathogens evade immune responses by various mechanisms:
Antigen masking: Coating with host molecules
Antigenic mimicry: Emulating host molecules
Antigenic variation: Altering surface molecules


Latency: Ability to persist quietly (e.g., herpes viruses)
Intracellular survival: Surviving inside macrophages (e.g., Mycobacterium, Histoplasma)
Immune suppression: Directly targeting immune cells, breaking down antibodies, interfering with signaling
Interference with phagocytosis: Making capsules, escaping phagosomes, blocking fusion, neutralizing enzymes, damaging phagocytes
Step 5—Transmission to a New Host
Transmission is facilitated by symptoms such as rashes, sneezing, coughing, and diarrhea. Pathogens exit via portals similar to their entry points and maintain reservoirs in the environment, organisms, or fomites.
Toxins as Major Virulence Factors
Toxins are molecules that cause tissue damage and suppress immune responses. Toxigenic microbes produce toxins, and toxemia refers to toxins in the bloodstream. There are two main classes:
Endotoxins: Lipid A component of lipopolysaccharide (LPS) in Gram-negative bacteria, released upon cell death.
Exotoxins: Potent, soluble proteins secreted by both Gram-positive and Gram-negative bacteria, classified by their mode of action.
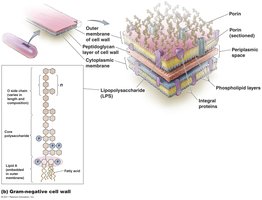
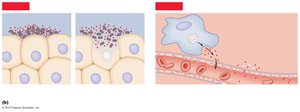
Endotoxins
Gram-negative bacteria have an outer membrane rich in LPS. The lipid A region is toxic and is released when bacteria die, causing fever, inflammation, diarrhea, and potentially septic shock. Endotoxins do not induce antibody production and are not readily neutralized.
Exotoxins
Exotoxins are classified into three families:
Type I: Membrane-acting extracellular toxins (e.g., superantigens, erythrogenic toxins)
Type II: Membrane-disruptive toxins (e.g., hemolysins)
Type III: Intracellular toxins (e.g., AB toxins such as botulinum and tetanospasmin)
Exotoxins stimulate antibody production and have specific cellular targets, such as the nervous system (neurotoxins) or gastrointestinal tract (enterotoxins).
Summary Table: Endotoxins vs. Exotoxins
Feature | Endotoxins | Exotoxins |
|---|---|---|
Source | Gram-negative bacteria (LPS) | Gram-positive and Gram-negative bacteria |
Release | Upon cell death | Secreted during growth |
Immunogenicity | Do not induce antibody production | Stimulate antibody production |
Neutralization | Not readily neutralized | Can be neutralized by antibodies |
Examples | Salmonella typhi, N. meningitidis, E. coli | Botulinum toxin, tetanospasmin, hemolysins |
Additional info: These notes expand on the original content by providing definitions, examples, and a comparative table for toxins, ensuring a comprehensive and self-contained study guide for microbiology students.
