 Back
Backlec 25:Inflammation and Innate Immunity: The Body’s First Line of Defense
Study Guide - Smart Notes

Inflammation and Innate Immunity
Overview of Innate Immunity
Innate immunity is the body’s immediate, non-specific defense mechanism against pathogens. Unlike adaptive immunity, which develops memory and specificity, innate immunity is always present and ready to respond to general signs of infection or tissue damage. Inflammation is a hallmark response, aiming to contain and eliminate threats rapidly.
Innate immunity acts within minutes to hours of infection.
It recognizes broad patterns associated with pathogens or cell damage, not specific antigens.
Inflammation is triggered to recruit immune cells and promote healing.
Barrier Defenses
Physical Mechanical Barriers
Physical barriers are the first line of defense, preventing pathogen entry into the body. The skin and mucous membranes are the primary structures involved.
Skin (Epidermis): Composed of multiple layers of epithelial cells, forming a tough, protective shield.
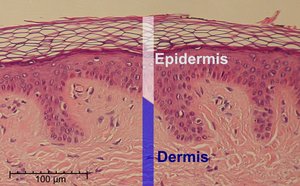
Keratinization: The outermost skin layer consists of dead, keratin-filled cells, creating a dry, inhospitable environment for microbes.
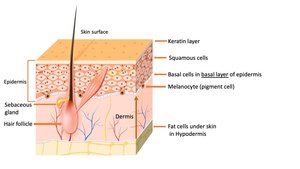
Desquamation: Continuous shedding of skin cells removes attached microbes from the surface.
Mucous Membranes: Line the respiratory, digestive, and urogenital tracts, providing a sticky barrier that traps pathogens.
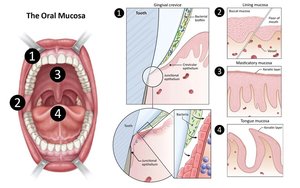
Mucus Trapping: Mucus captures microbes and debris, preventing their penetration into tissues.
Mucociliary Escalator: In the respiratory tract, ciliated cells move mucus and trapped pathogens upward to be expelled or swallowed.
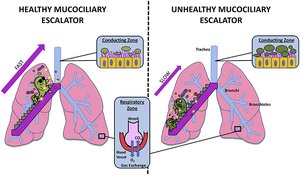
Flushing Mechanisms: Tears, saliva, urine, and sweat physically wash away microbes from body surfaces.

Chemical Barriers
Chemical defenses act rapidly to destroy or inhibit microbes that breach physical barriers.
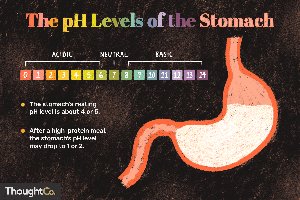
Acidic Environments:
Stomach acid: Gastric juice with a pH of 1–2 kills most ingested pathogens.
Skin secretions: Sebum contains fatty acids, lowering skin pH to 4–5, which inhibits bacterial growth.

Enzymes & Antimicrobial Proteins:
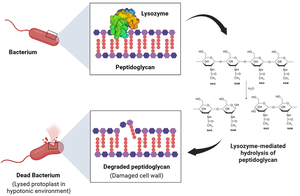
Lysozyme: Found in tears, saliva, and mucus; breaks down bacterial cell walls, leading to cell lysis.
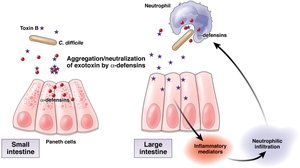
Antimicrobial Peptides (AMPs): Molecules like defensins disrupt microbial membranes, acting as natural antibiotics.
Lactoferrin: Binds iron in secretions, depriving bacteria of this essential nutrient and inhibiting their growth.

Biological Barriers (The Microbiome)
The human microbiome consists of trillions of commensal microbes that protect against pathogens through several mechanisms.
Competitive Exclusion: Commensal microbes occupy niches and consume resources, preventing pathogen colonization.

Bacteriocins: Some commensal bacteria produce toxins that kill or inhibit pathogenic species.
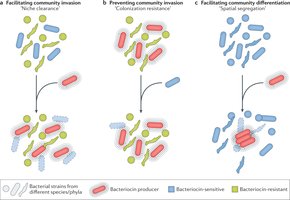
Immune Training: The microbiome helps maintain immune readiness and responsiveness.
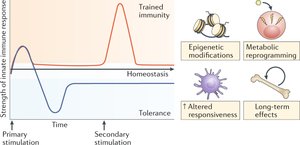
Pattern Recognition and Immune Activation
Pattern Recognition Receptors (PRRs)
PRRs are specialized proteins that detect common molecular patterns associated with pathogens (PAMPs) or cell damage (DAMPs). They are strategically located to sense danger both outside and inside cells.
Transmembrane PRRs: Located on cell surfaces (e.g., TLRs 1, 2, 4, 5, 6).
Endosomal PRRs: Found in intracellular vesicles (e.g., TLRs 3, 7, 8, 9).
Cytoplasmic PRRs: Detect cytosolic threats (e.g., NLRs, RIG-I-like receptors).

Pathogen-Associated Molecular Patterns (PAMPs)
PAMPs are conserved microbial structures recognized by PRRs, enabling rapid immune activation.
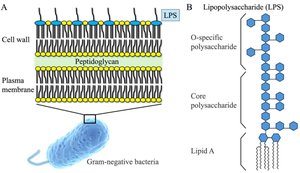
Lipopolysaccharide (LPS): Found in Gram-negative bacteria; detected by TLR4.
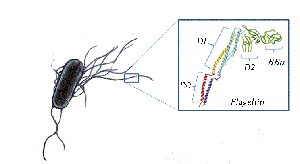
Flagellin: Protein component of bacterial flagella; detected by TLR5.
Peptidoglycan: Major component of bacterial cell walls.
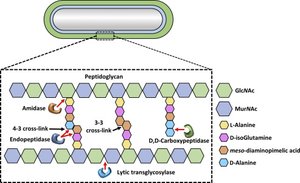
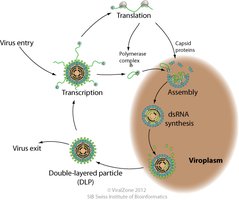
Double-stranded RNA (dsRNA): Indicates viral infection; not found in healthy human cells.
Damage-Associated Molecular Patterns (DAMPs)
DAMPs are host molecules that signal tissue injury or cellular stress when detected in abnormal locations.
Extracellular ATP: Signals cell rupture and tissue damage.
High Mobility Group Box 1 (HMGB1): Nuclear protein that triggers inflammation when released extracellularly.
Cytosolic DNA: Presence in the cytoplasm indicates infection or nuclear damage.
The Complement System
The complement system is a group of plasma proteins that enhance innate immunity by directly killing microbes or marking them for destruction (opsonization).
Forms membrane attack complexes that lyse bacteria.
Promotes phagocytosis and inflammation.
Inflammatory Response
Phases of Inflammation
Inflammation is a coordinated response to infection or injury, characterized by redness, heat, swelling, pain, and loss of function.
Detection & Signaling: Sentinel cells (mast cells, macrophages) detect PAMPs/DAMPs and release cytokines and histamines.
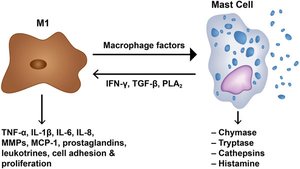
Vasodilation: Blood vessels widen and become more permeable, increasing blood flow and immune cell delivery.
Recruitment (Diapedesis): White blood cells migrate through vessel walls to the site of infection or injury.
Phagocytosis: Neutrophils and macrophages engulf and destroy pathogens.
Resolution & Healing: Inflammation subsides and tissue repair begins.
Types of Inflammation
Acute Inflammation: Short-term, resolves with healing.
Chronic Inflammation: Long-term, can damage healthy tissue and contribute to diseases (e.g., Alzheimer’s, heart disease).
Summary Table: Key Innate Immune Barriers and Mechanisms
Barrier/Mechanism | Main Function | Example |
|---|---|---|
Physical | Block entry | Skin, mucous membranes |
Chemical | Destroy/inhibit microbes | Stomach acid, lysozyme, defensins |
Biological | Compete with pathogens | Microbiome, bacteriocins |
PRRs | Detect PAMPs/DAMPs | TLRs, NLRs |
Complement | Lyse/tag microbes | Membrane attack complex |
Practice Questions
A patient with impaired keratinization but intact microbiome is MOST likely to first experience: B. Increased bacterial colonization on the skin surface
Which of the following BEST represents a DAMP-mediated inflammatory trigger? C. Extracellular ATP detection after tissue injury
A defect in ciliary movement would MOST directly result in: B. Impaired clearance of trapped pathogens
Which of the following scenarios would most strongly activate endosomal TLRs rather than cytoplasmic PRRs? B. Phagocytosis of a virus followed by detection of its nucleic acids in vesicles
A mutation prevents recognition of cytosolic DNA but leaves all other PRRs intact. Which infection is MOST likely to evade detection initially? B. DNA virus replicating in cytoplasm
Why does innate immunity rely on PRRs instead of highly specific receptors like adaptive immunity? B. PRRs allow immediate response without prior exposure
If vasodilation occurs but diapedesis fails, which outcome is MOST likely? C. Increased blood flow without effective immune cell infiltration
Why are PAMPs ideal targets for innate immune recognition? C. They are conserved and essential for microbial survival
Which mechanism BEST explains how microbiota prevent pathogen colonization? B. Competitive exclusion and bacteriocin production
