 Back
BackMacromolecules in Microbiology: Structure, Function, and Relevance
Study Guide - Smart Notes

Macromolecules: An Overview
Definition and Major Classes
Macromolecules are large, complex molecules essential for life. Living organisms are primarily composed of four major types of macromolecules: carbohydrates, proteins, nucleic acids, and lipids. Each class plays distinct and critical roles in cellular structure and function.
Carbohydrates: Energy storage and structural components
Proteins: Catalysis, structure, transport, regulation, and more
Nucleic acids: Storage and transmission of genetic information
Lipids: Membrane structure, energy storage, and signaling

Monomers and Polymers
Building Blocks and Assembly
Most macromolecules are polymers, constructed from repeating subunits called monomers. The process of linking monomers to form polymers is called dehydration synthesis (or condensation), while breaking polymers into monomers is called hydrolysis (or decomposition).
Carbohydrates: Monomer = monosaccharide (e.g., glucose); Polymer = polysaccharide (e.g., starch, cellulose, glycogen)
Proteins: Monomer = amino acid; Polymer = polypeptide/protein
Nucleic acids: Monomer = nucleotide; Polymer = DNA or RNA
Lipids: Not true polymers, but built from fatty acids and glycerol (e.g., triglycerides, phospholipids)

Carbohydrates
Structure and Function
Carbohydrates are a major energy source for most living things and are important structural components in cells (e.g., cell wall, capsule). The primary carbohydrate used by human cells for energy during cellular respiration is glucose.
Monosaccharides: Simple sugars (e.g., glucose)
Disaccharides: Two monosaccharides joined together (e.g., sucrose)
Polysaccharides: Long chains of monosaccharides (e.g., starch, cellulose, glycogen)

Concept Check Example
Which of these is a monomer? Glucose (starch, cellulose, and glycogen are polymers)
Polymers and Monomers: Synthesis and Breakdown
Dehydration Synthesis and Hydrolysis
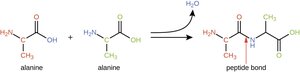
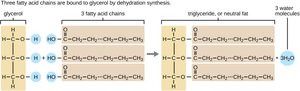
Polymers are formed by dehydration synthesis, where a water molecule is removed to join two monomers. Polymers are broken down by hydrolysis, where water is added to split the bond.
Dehydration synthesis: Builds macromolecules
Hydrolysis: Breaks macromolecules into monomers
Equation for dehydration synthesis:
Equation for hydrolysis:
Proteins
Structure and Function
Proteins are polymers of amino acids and are the most functionally diverse macromolecules. They serve as enzymes, structural components, transporters, regulators, and more. The tetanus toxin is an example of a bacterial protein that disrupts human physiology by blocking inhibitory neurons, leading to rigid paralysis.
Structural proteins: Provide support (e.g., collagen)
Motility proteins: Enable movement (e.g., actin, myosin)
Transport proteins: Move substances across membranes (e.g., hemoglobin, membrane channels)
Enzymes: Catalyze biochemical reactions (e.g., DNA polymerase)
Regulatory proteins: Control cellular processes (e.g., transcription factors)
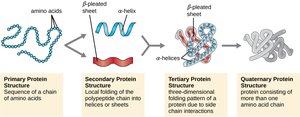
Protein Structure
Proteins are made of amino acids linked by peptide bonds. The general structure of an amino acid includes an amino group, a carboxyl group, and a variable side chain (R group). Proteins fold into specific shapes, which determine their function.
Primary structure: Sequence of amino acids
Secondary structure: Local folding (α-helix, β-sheet)
Tertiary structure: Overall 3D shape
Quaternary structure: Multiple polypeptide chains
Protein Denaturation
Denaturation is the loss of protein structure (and function) due to heat, chemicals, or pH changes. Only proteins in their correct tertiary or quaternary structure are fully functional.
Functional proteins: Tertiary and quaternary structures
Non-functional proteins: Primary or secondary structure only
Nucleic Acids
DNA and RNA: Structure and Function
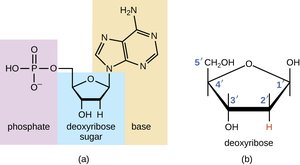
Nucleic acids store and transmit genetic information. DNA contains the instructions for building and controlling cells, while RNA is involved in protein synthesis and, in some viruses, also stores genetic information.
DNA: Double helix, antiparallel strands, base pairing (A-T, G-C), hydrogen bonds
RNA: Single-stranded, contains ribose and uracil (instead of deoxyribose and thymine)
mRNA: Messenger RNA, copy of a gene
rRNA: Ribosomal RNA, part of ribosome structure
tRNA: Transfer RNA, delivers amino acids during translation
Concept Check Example
What is unique to RNA compared to DNA? RNA contains uracil (U) and ribose sugar.
Lipids
Types and Functions
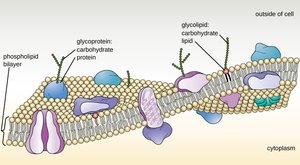
Lipids are hydrophobic molecules with diverse structures and functions. They are not true polymers but are essential for cell membrane structure, energy storage, and signaling.
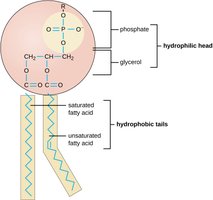
Phospholipids: Main component of cell membranes; structure includes phosphate, glycerol, and fatty acids
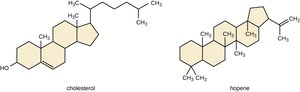
Steroids: Four carbon-based rings; strengthen cell membranes (e.g., cholesterol in eukaryotes, hopanoids in prokaryotes)
Triglycerides: Glycerol and three fatty acids; energy storage
Applications and Microbial Relevance
Examples of Macromolecule Use in Microbes
Carbohydrates: Bacterial cell walls (peptidoglycan), energy storage (glycogen)
Proteins: Enzymes for metabolism, toxins (e.g., tetanus toxin)
Nucleic acids: Plasmids for antibiotic resistance, viral genomes
Lipids: Membrane structure (phospholipids), energy reserves (polyhydroxyalkanoates)
Additional info: Understanding macromolecules is foundational for topics such as microbial metabolism, genetics, pathogenesis, and biotechnology.
