 Back
BackMacromolecules: Structure, Function, and Relevance in Microbiology
Study Guide - Smart Notes

Macromolecules in Microbiology
Introduction to Macromolecules
Macromolecules are large, complex molecules essential for life, including carbohydrates, proteins, nucleic acids, and lipids. In microbiology, understanding their structure and function is fundamental for studying cellular processes and microbial physiology.
Polymer: A long molecule composed of repeating subunits called monomers.
Monomer: The basic building block of a polymer.
Three major classes of macromolecules—carbohydrates, proteins, and nucleic acids—are polymers.

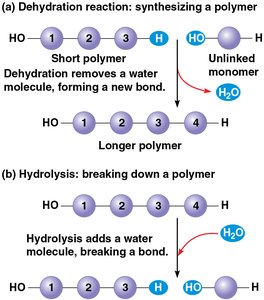
Synthesis and Breakdown of Polymers
Polymers are formed and broken down by specific chemical reactions, which are catalyzed by enzymes in biological systems.
Dehydration Reaction: Joins two monomers by removing a water molecule, forming a covalent bond.
Hydrolysis: Breaks a polymer into monomers by adding a water molecule, breaking the covalent bond.
These reactions are essential for the metabolism and recycling of macromolecules in cells.
Types of Biological Macromolecules
Carbohydrates
Carbohydrates are organic molecules consisting of carbon, hydrogen, and oxygen. They serve as energy sources and structural components in cells.
Monosaccharides: Simple sugars (e.g., glucose, fructose) that are the monomers of carbohydrates.
Disaccharides: Composed of two monosaccharides joined by a glycosidic bond (e.g., sucrose).
Polysaccharides: Long chains of monosaccharides (e.g., starch, cellulose, glycogen) with structural or storage roles.
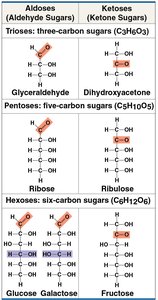
Monosaccharide Classification
Monosaccharides are classified by the number of carbon atoms and the location of the carbonyl group.
Aldoses: Contain an aldehyde group.
Ketoses: Contain a ketone group.
Common examples: Glucose (aldose), Fructose (ketose).
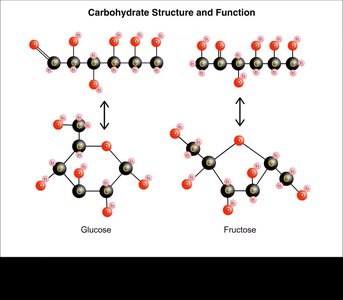
Carbohydrate Structure and Function
Monosaccharides can exist in linear or ring forms, with the ring form being more stable in aqueous solutions. Polysaccharides differ in their glycosidic linkages, affecting their structure and function.
Starch: Storage polysaccharide in plants, composed of glucose monomers (α configuration).
Glycogen: Storage polysaccharide in animals, highly branched (α configuration).
Cellulose: Structural polysaccharide in plants, composed of glucose monomers (β configuration).
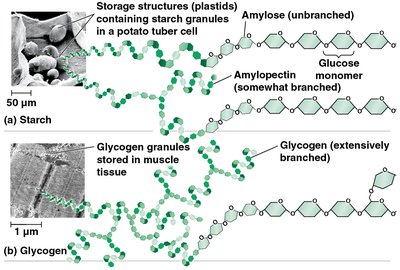
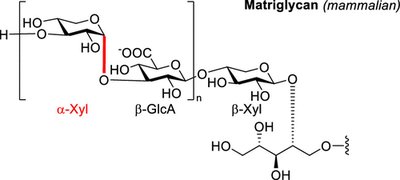
Specialized Polysaccharides
Some polysaccharides, such as matriglycan, play specialized roles in cellular structure and disease.
Matriglycan: A complex sugar involved in muscle cell membrane structure and associated with Duchenne Muscular Dystrophy.
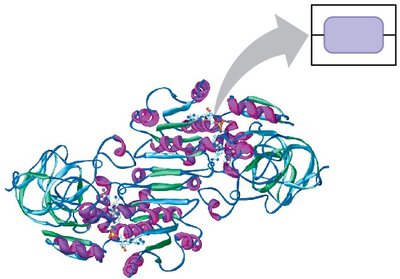
Proteins
Proteins are polymers of amino acids and perform a wide range of functions, including catalysis, transport, and structural support.
Amino Acid: The monomer unit of proteins, containing an amino group, carboxyl group, and a unique side chain.
Polypeptide: A chain of amino acids linked by peptide bonds.
Protein structure is determined by the sequence of amino acids and their interactions.
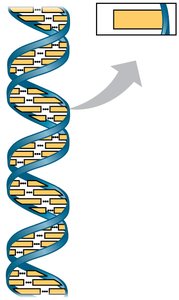
Nucleic Acids
Nucleic acids, including DNA and RNA, are polymers of nucleotides and are responsible for storing and transmitting genetic information.
Nucleotide: The monomer of nucleic acids, consisting of a nitrogenous base, a pentose sugar, and one or more phosphate groups.
DNA: Double-stranded molecule with complementary base pairing (A-T, G-C).
RNA: Single-stranded molecule with uracil (U) replacing thymine (T).
Nucleoside: A nucleotide without the phosphate group.

Types of RNA
mRNA (messenger RNA): Carries genetic information from DNA to ribosomes for protein synthesis.
tRNA (transfer RNA): Brings amino acids to the ribosome during translation.
rRNA (ribosomal RNA): Forms the core of ribosome structure and catalyzes protein synthesis.
shRNA and siRNA: Used in gene regulation and scientific research for gene knockdown.
Lipids
Lipids are diverse molecules that are not polymers. They are hydrophobic and serve as energy sources, components of cell membranes, and hormones.
Phospholipids: Major component of cell membranes, forming bilayers.
Triglycerides: Storage form of energy in cells.
Steroids: Act as hormones and signaling molecules.
Key Chemical Principles in Macromolecule Biology
Chemical Bonds in Macromolecules
Macromolecules are held together by covalent bonds formed during polymerization reactions. Hydrogen bonds and other non-covalent interactions stabilize their three-dimensional structures.
Glycosidic bond: Joins monosaccharides in polysaccharides.
Peptide bond: Joins amino acids in proteins.
Phosphodiester bond: Joins nucleotides in nucleic acids.
Summary Table: Macromolecule Classes
Macromolecule | Monomer | Bond Type | Main Functions |
|---|---|---|---|
Carbohydrate | Monosaccharide | Glycosidic | Energy, structure |
Protein | Amino acid | Peptide | Catalysis, transport, structure |
Nucleic Acid | Nucleotide | Phosphodiester | Genetic information, gene expression |
Lipid | Fatty acid (not a true polymer) | Varied | Energy, membranes, hormones |
Conclusion
Understanding the structure and function of macromolecules is essential for microbiology, as these molecules underpin cellular processes, genetic inheritance, and metabolic pathways. Mastery of these concepts provides a foundation for advanced study in microbial physiology, genetics, and biotechnology.
