 Back
BackMicrobial Biogeochemical Cycling: Carbon and Nitrogen Cycles
Study Guide - Smart Notes

Nutrient and Biogeochemical Cycling
Overview of Biogeochemical Cycles
Biogeochemical cycles describe the movement and transformation of elements through the biosphere, atmosphere, hydrosphere, and lithosphere. Microbes, especially prokaryotes, are essential for these cycles, enabling the recycling of nutrients necessary for life. - Key Point 1: Prokaryotes are responsible for cycling all vital elements on Earth, including carbon and nitrogen. - Key Point 2: Without microbial activity, biogeochemical cycles would cease, disrupting life and ecosystem function. - Example: All organic nitrogen is originally fixed by prokaryotes; over half of organic carbon is of bacterial origin. 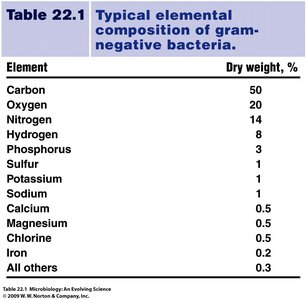
Major Nutrient Reservoirs
Elements cycle through various reservoirs: lithosphere (rocks), hydrosphere (water), biosphere (living organisms), and atmosphere (air). - Key Point 1: Carbon and nitrogen are found in multiple reservoirs and undergo chemical transformations as they move through ecosystems. - Key Point 2: Disruption of these cycles by human activity (e.g., fossil fuel burning, deforestation) can lead to environmental imbalances.
The Carbon Cycle
Microbial Roles in the Carbon Cycle
The carbon cycle involves the fixation of CO2 by photosynthetic and chemolithoautotrophic microbes, and its release through respiration and decay. - Key Point 1: Photosynthesizers (e.g., cyanobacteria) convert atmospheric CO2 into organic matter. - Key Point 2: Microbial decomposition returns carbon to the atmosphere, completing the cycle. - Example: Microbial decay of felled trees in forests releases carbon, while regrowth sequesters it. 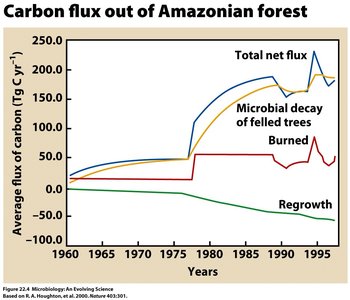
Global Carbon Imbalance
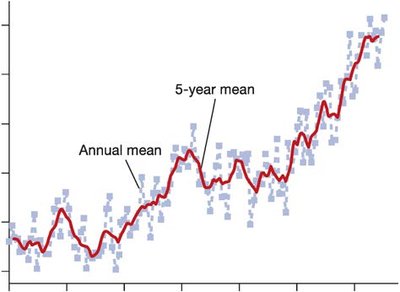
Human activities have altered the carbon cycle, leading to increased atmospheric CO2 and climate change. - Key Point 1: Atmospheric CO2 is determined by the balance between CO2 release (fossil fuel burning, deforestation) and CO2 fixation (photosynthesis). - Key Point 2: Current imbalance: CO2 release = 232 bmt/year; CO2 fixation = 227 bmt/year; net increase = 5 bmt/year. - Example: Rising global concentrations of greenhouse gases (CO2, methane, N2O) correlate with increasing global temperatures. 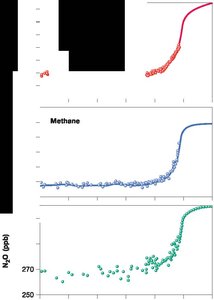
The Nitrogen Cycle
Importance and Limitation of Nitrogen
Nitrogen is an essential, growth-limiting element in ecosystems. It is a major component of proteins, nucleic acids, and other macromolecules. - Key Point 1: Nitrogen must be "fixed" from atmospheric N2 into biologically usable forms (NH3, NO3). - Key Point 2: Nitrogenase enzyme, found in certain prokaryotes, catalyzes nitrogen fixation but is sensitive to O2.
Nitrogen Reservoirs and Key Processes
The nitrogen cycle includes fixation, assimilation, nitrification, and denitrification. - Key Point 1: Nitrogen-fixing bacteria convert N2 to NH3. - Key Point 2: Nitrifying bacteria (e.g., Nitrosomonas, Nitrobacter) convert NH4+ to NO2- and then to NO3-. - Key Point 3: Denitrifying bacteria (e.g., Paracoccus denitrificans) convert NO3- to N2, returning nitrogen to the atmosphere. - Example: Nitrogen-fixing bacteria in root nodules of legumes provide plants with usable nitrogen. 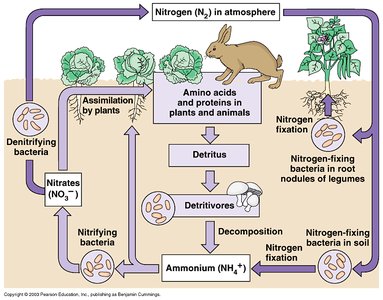
Imbalances in the Nitrogen Cycle
Human activities (fertilizer use, fossil fuel combustion) disrupt the nitrogen cycle, causing environmental issues such as dead zones and greenhouse gas emissions. - Key Point 1: Excess nitrification and denitrification can lead to nutrient pollution and hypoxic zones in aquatic environments. - Key Point 2: Dead zones are areas of low oxygen caused by nutrient overload, threatening marine life. 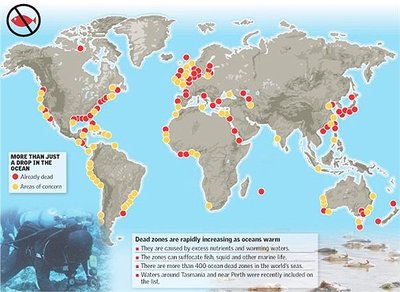
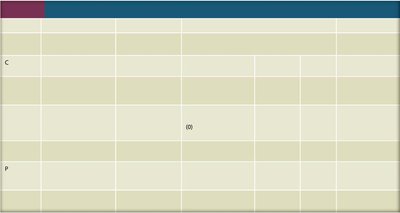
Summary Table: Elemental Composition of Gram-Negative Bacteria
Major Elements in Bacterial Biomass
The table below summarizes the typical elemental composition of gram-negative bacteria, highlighting the importance of carbon, nitrogen, and other elements in microbial life.
Element | Dry weight, % |
|---|---|
Carbon | 50 |
Oxygen | 20 |
Nitrogen | 14 |
Hydrogen | 8 |
Phosphorus | 3 |
Sulfur | 1 |
Potassium | 1 |
Sodium | 1 |
Calcium | 0.5 |
Magnesium | 0.5 |
Chlorine | 0.5 |
Iron | 0.2 |
All others | 0.3 |
Key Equations
Atmospheric CO2 Balance
The net change in atmospheric CO2 is determined by the difference between release and fixation:
Nitrification and Denitrification Reactions
Nitrification: Denitrification:
Conclusion
Microbial biogeochemical cycling is fundamental to ecosystem function and global environmental health. Understanding the carbon and nitrogen cycles, and the microbial processes that drive them, is essential for microbiology students and for addressing human impacts on the biosphere. Additional info: Expanded explanations and context were added to clarify the role of microbes in biogeochemical cycles and the environmental consequences of cycle disruption.
