 Back
BackMicrobial Biosynthesis and Metabolism: Pathways and Building Blocks
Study Guide - Smart Notes

Biosynthesis and Metabolism in Microorganisms
Overview of Microbial Metabolism
Microbial metabolism encompasses all biochemical reactions that occur within microorganisms, including both energy-generating (catabolic) and biosynthetic (anabolic) processes. These reactions are essential for growth, maintenance, and reproduction.
Catabolism: The breakdown of substrates to generate energy, typically in the form of ATP and proton motive force.
Anabolism (Biosynthesis): The use of energy to build macromolecules and cellular constituents from monomers.
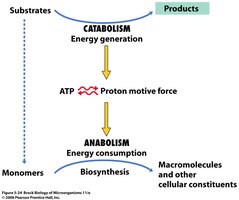
Example: ATP produced during catabolism is used to drive biosynthetic reactions in anabolism.
Acetogenesis in Acetobacterium woodii
Reactions of Acetogenesis from H2 and CO2
Acetogenesis is a process by which certain bacteria, such as Acetobacterium woodii, convert hydrogen and carbon dioxide into acetate. This process is important in anaerobic environments and contributes to the global carbon cycle.
Key Steps: Involves the reduction of CO2 to acetate, coupled with ATP synthesis.
Energy Conservation: Utilizes sodium ion gradients and substrate-level phosphorylation for ATP generation.
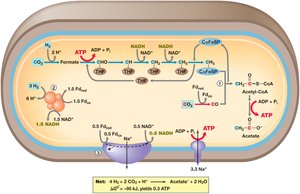
Example: The overall reaction: 4 H2 + 2 CO2 → Acetate + 2 H2O.
The Calvin-Benson Cycle
CO2 Fixation and Sugar Biosynthesis
The Calvin-Benson cycle is the primary pathway for carbon fixation in autotrophic microorganisms and plants. It converts CO2 into organic molecules using ATP and NADPH generated from light-dependent reactions.
Phases: Carboxylation, reduction, and regeneration of ribulose-1,5-bisphosphate.
Products: Fructose-6-phosphate, which can be used for biosynthesis of other sugars.
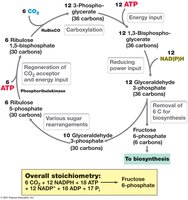
Equation:
Biological Nitrogen Fixation
Nitrogenase and the Reduction of N2 to Ammonia
Nitrogen fixation is the process by which atmospheric nitrogen (N2) is converted to ammonia (NH3) by the enzyme nitrogenase. This process is essential for incorporating nitrogen into biological molecules.
Enzyme: Nitrogenase complex, which requires ATP and reducing power.
Energy Requirement: 16 ATP molecules are hydrolyzed for each N2 reduced.
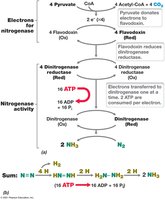
Equation:
Biosynthesis of Sugars and Polysaccharides
Hexose and Pentose Formation
Microorganisms synthesize hexoses (e.g., glucose) and pentoses (e.g., ribose) for use in energy storage and nucleic acid synthesis. These sugars can be obtained from the environment or synthesized via gluconeogenesis and the pentose phosphate pathway.
Hexoses: Formed by reversal of glycolysis or from citric acid cycle intermediates.
Pentoses: Produced by decarboxylation of hexoses via the pentose phosphate pathway.
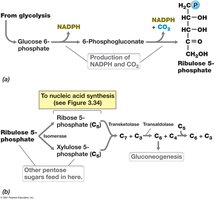
Example: Ribose-5-phosphate is a precursor for nucleotide biosynthesis.
Polysaccharide Biosynthesis
Polysaccharides such as glycogen, starch, and peptidoglycan subunits are synthesized from activated glucose derivatives, including UDP-glucose (UDPG) and ADP-glucose (ADPG).
Activation: Glucose is activated by attachment to nucleotides (UDP or ADP).
Polymerization: Activated glucose units are added to growing polysaccharide chains.

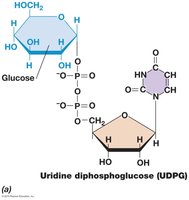
Equation:
Biosynthesis of Amino Acids
Sources of Carbon Skeletons and Amino Groups
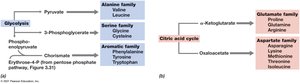
Amino acids are synthesized from intermediates of glycolysis and the citric acid cycle. The amino group is typically derived from inorganic nitrogen sources such as ammonia.
Families: Amino acids are grouped based on their biosynthetic precursors (e.g., pyruvate, oxaloacetate, α-ketoglutarate).
Transamination: Transfer of amino groups from glutamate or glutamine to other carbon skeletons.
Ammonia Incorporation in Bacteria
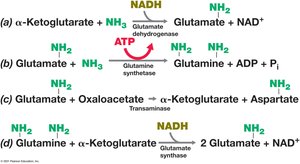
Bacteria assimilate ammonia via two main pathways: the glutamate dehydrogenase pathway and the glutamine synthetase–glutamate synthase pathway.
Glutamate Dehydrogenase: Incorporates ammonia into α-ketoglutarate to form glutamate.
Glutamine Synthetase: Converts glutamate and ammonia to glutamine (requires ATP).
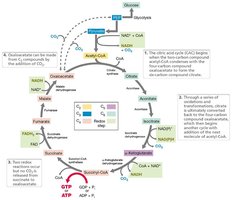
Citric Acid Cycle in Biosynthesis
The citric acid cycle provides key intermediates for the biosynthesis of amino acids and other cellular components.
Intermediates: Oxaloacetate and α-ketoglutarate serve as precursors for aspartate and glutamate families, respectively.
Biosynthesis of Nucleotides
Purine and Pyrimidine Synthesis
Nucleotides are synthesized from carbon and nitrogen sources, including amino acids, CO2, and folic acid derivatives. Inosinic acid is the precursor for purines (adenine and guanine), while uridylate is the precursor for pyrimidines (cytosine, thymine, and uracil).
Purine Ring: Assembled from amino acids (glycine, aspartate, glutamine), CO2, and formyl groups.
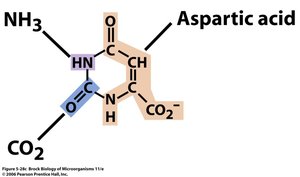
Pyrimidine Ring: Formed from aspartate and carbamoyl phosphate.
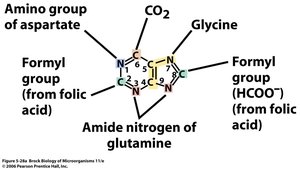
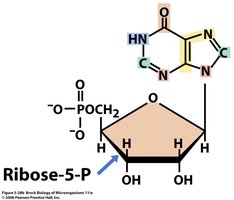
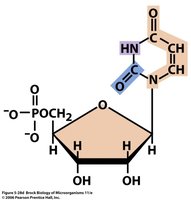
Example: Ribose-5-phosphate is a key precursor for both purine and pyrimidine nucleotide synthesis.
Biosynthesis of Fatty Acids and Lipids
Fatty Acid Synthesis
Fatty acids are synthesized by the sequential addition of two-carbon units (acetyl groups) carried by acyl carrier protein (ACP). The process is repeated until the desired chain length is achieved.
Key Enzymes: Fatty acid synthase complex, which uses NADPH as a reducing agent.
End Products: Long-chain fatty acids such as palmitate (C16).
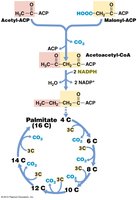
Example: Fatty acids are incorporated into membrane lipids, which may also contain glycerol, phosphate, and sugars.
