 Back
BackMicrobial Biosynthesis: Autotrophy, Nitrogen Fixation, and Macromolecule Formation
Study Guide - Smart Notes

Microbial Metabolism: Synthesis from Inorganic Sources
Overview of Microbial Biosynthesis
Microorganisms possess remarkable metabolic capabilities, allowing them to synthesize essential biomolecules from simple inorganic compounds. This process is fundamental to global nutrient cycling and underpins the ecological success of bacteria and archaea. Key biosynthetic pathways include autotrophy (CO2 fixation), nitrogen fixation, and the synthesis of carbohydrates, nucleotides, amino acids, and lipids.
Autotrophy: CO2 Fixation
Definition and Types of Autotrophy
Autotrophy refers to the ability of organisms to use inorganic carbon (CO2) as their sole carbon source. This process is carried out by both phototrophs (using light energy) and chemotrophs (using chemical energy), and is often associated with lithotrophic metabolism.
Photoautotrophs: Use light energy to fix CO2 (e.g., plants, cyanobacteria).
Chemoautotrophs: Use chemical energy from inorganic compounds to fix CO2 (e.g., nitrifying bacteria).
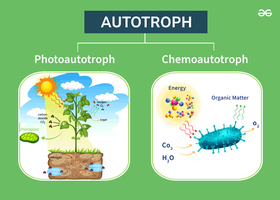
Importance: Autotrophic bacteria, phytoplankton, and algae play a vital role in global carbon cycling.
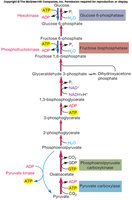
Calvin Cycle and Gluconeogenesis
The Calvin Cycle is the primary pathway for CO2 fixation in autotrophs. CO2 is incorporated into ribulose 1,5-bisphosphate, forming 3-phosphoglycerate (3-PG), which is then used to build carbohydrates via gluconeogenesis.
Carbon fixation: Consumes ATP and NADPH generated during photosynthesis.
Gluconeogenesis: Converts 3-PG into glucose and other sugars.
Nitrogen Fixation
Atmospheric Nitrogen and Biological Availability
The atmosphere contains ~78% nitrogen gas (N2), but most organisms cannot use N2 directly. Nitrogen fixation is the process by which certain bacteria convert N2 into ammonia (NH3), making nitrogen biologically available.
Enzyme: Nitrogenase complex mediates the reduction of N2 to NH3.
Energy cost: 16 ATP per N2 molecule fixed.
Electron donors: Electrons are supplied by catabolic metabolism (e.g., pyruvate, acetyl-CoA).
Carbohydrate Biosynthesis
Gluconeogenesis
Gluconeogenesis is the synthesis of glucose from non-carbohydrate precursors, essential for cells growing on non-sugar carbon sources. It is essentially the reverse of glycolysis, with three key steps requiring energy input from ATP or GTP.
Key steps: Pyruvate to PEP, FBP to F6P, G6P to glucose.
Importance: Provides glucose for cell wall synthesis (e.g., NAG and NAM for peptidoglycan).
Pentose Phosphate Pathway
The pentose phosphate pathway generates ribose-5-phosphate for nucleotide synthesis, erythrose-4-phosphate for aromatic amino acids, and NADPH for biosynthetic reactions.
Major outcomes: Ribulose 5-phosphate (nucleotides), erythrose-4-phosphate (amino acids), NADPH (biosynthesis).
Nucleotide and Amino Acid Biosynthesis
Nucleotide Synthesis
Nucleotides are synthesized from ribose (via the pentose phosphate pathway) and nitrogenous bases derived from amino acids. Purines are synthesized as inosinic acid, while pyrimidines are synthesized as uridylate.
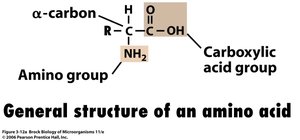
Amino Acid Synthesis
Amino acids are formed from carbon skeletons generated during glycolysis and the citric acid cycle. The amino group is often incorporated from free ammonia through reductive transamination and ammonia assimilation.
General structure: Contains an α-carbon, amino group, carboxylic acid group, and variable R group.
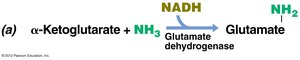
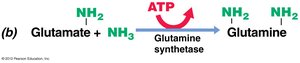
Ammonia assimilation: NH3 is incorporated into glutamate.
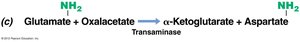
Transamination: Amino group from glutamate or glutamine is transferred to other compounds to form other amino acids.
Lipid Biosynthesis
Formation of Lipids
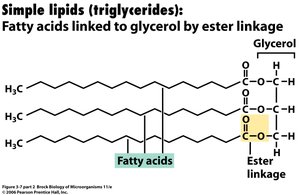
Lipids are synthesized from glycerol (produced from DHAP in glycolysis) and fatty acids (synthesized two carbons at a time with acyl carrier proteins). Fatty acids are attached to glycerol to form triglycerides.
Synthesis of Fatty Acids
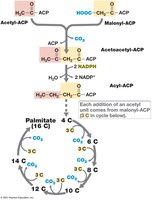
Fatty acid synthesis involves the condensation of acetyl-ACP and malonyl-ACP, followed by reduction steps requiring NADPH. Each cycle adds two carbons to the growing fatty acid chain.
Key reactions: Acetyl-ACP + Malonyl-ACP → Acetoacetyl-ACP; Acetoacetyl-ACP + 2NADPH → Acyl-ACP + 2NADP+
Energy cost: Fatty acid synthesis is energetically expensive due to high NADPH requirements.
Lithotrophy and Electron Sources
Definition and Types of Lithotrophy
Lithotrophy refers to the use of inorganic compounds as electron donors for energy generation. Lithotrophs can be chemolithotrophs (energy from chemicals) or photolithotrophs (energy from light).
Organotrophs: Use organic compounds as electron donors.
Lithotrophs: Use inorganic compounds as electron donors.
Metabolic Diversity
Both chemolithotrophy and chemoorganotrophy can involve aerobic or anaerobic respiration and rely on oxidative phosphorylation for ATP production. However, chemolithotrophs typically do not use fermentation and are often associated with autotrophic metabolism.
Photolithotrophy
Photolithotrophs use light energy to drive electron transport and ATP synthesis. There are two major forms:
Anoxygenic: Water is not the electron donor (found only in Bacteria).
Oxygenic: Water is the electron donor (e.g., cyanobacteria).
Electrons are derived from inorganic donors (e.g., H2S), and reducing power is generated by reverse electron transport.
Anaerobic Respiration
Diversity of Respiratory Pathways
Cellular respiration in bacteria and archaea is highly modular, allowing for the use of various inorganic compounds as terminal electron acceptors in anaerobic environments. This metabolic flexibility has contributed to the evolutionary success and ecological diversity of bacteria.
Key point: Respiration is not limited to oxygen; many inorganic compounds can serve as electron acceptors.
Summary of Key Concepts
Microbes can synthesize essential biomolecules from inorganic sources via autotrophy and nitrogen fixation.
Gluconeogenesis and the pentose phosphate pathway are central to carbohydrate and nucleotide biosynthesis.
Amino acids and lipids are synthesized from metabolic intermediates, with energy and reducing power supplied by catabolic and photosynthetic processes.
Lithotrophy and anaerobic respiration highlight the metabolic diversity and adaptability of bacteria and archaea.
