 Back
BackLectures 18 and 19
Study Guide - Smart Notes

Catabolism in Microorganisms
Introduction to Catabolism
Catabolism refers to the breakdown or degradation of complex molecules into simpler ones, releasing energy that microbes use for survival. This process is central to microbial metabolism and is essential for energy conservation and cellular function.
Energy Conservation: Microbes must generate and store reducing power and conserve energy in the form of ATP, metabolites, and ion motive force (imf).
Metabolic Capabilities: The ability to utilize various substrates for energy is a key survival trait.
Learning Goals
Apply chemical knowledge to understand microbial survival strategies.
Analyze metabolic pathways using chemical reaction patterns.
Understand how prokaryotes conserve energy.
Fermentation: Definition and Mechanism
What is Fermentation?
Fermentation is a metabolic process where energy is derived from substrate-level phosphorylation (SLP) without the use of terminal electron acceptors. It is a less efficient energy conservation strategy compared to respiration.
Energy Source: ATP is generated directly from chemical reactions (SLP).
No Terminal Electron Acceptors: Unlike respiration, fermentation does not use oxygen or other external electron acceptors.
Lower Energy Yield: Fermentation yields less energy than respiration.
Typical Substrates: Sugars, amino acids, purines, etc.
Typical Products: Organic acids (acetate, lactate), alcohols (ethanol, butanediol), and gases (H2, CO2).

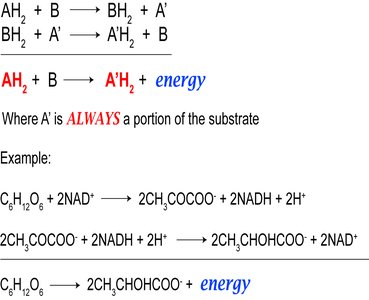
General Fermentation Reaction
The general pattern of fermentation involves the transfer of electrons from a reduced substrate to an oxidized substrate, generating energy:
Redox Balance: The substrate is both oxidized and reduced, ensuring redox balance.
Example: Glycolysis followed by fermentation of pyruvate to lactate or ethanol.
Substrate-Level Phosphorylation (SLP)
Mechanism of SLP
SLP is the direct synthesis of ATP from ADP and inorganic phosphate during a single enzymatic reaction in catabolic pathways. It is the primary means of ATP generation in fermentation.
Energy-Rich Intermediates: Compounds such as phosphoenolpyruvate, acetyl phosphate, and succinyl-CoA are involved.
Dehydrogenation and Lyase Reactions: These reactions generate high-energy intermediates that drive ATP synthesis.
Key Reactions Coupled to SLP
Substrate | Product(s) | Energy-Rich Intermediate |
|---|---|---|
Pyruvate + 2H2O | Acetate + HCO3- + H2 + H+ | Acetyl-CoA / Acetyl-P |
Acetaldehyde + H2O | Acetate + H2 + H+ | Acetyl-CoA / Acetyl-P |
GAP + H2O | PG + H2 + H+ | Biphosphoglycerate |
α-Ketoglutarate + 2H2O | Succinate2- + HCO3- + H2 + H+ | Succinyl-CoA |
HSO3- + H2O | SO42- + H2 + H+ | Adenylyl sulfate |
Abbreviations: GAP = glyceraldehyde phosphate; PG = phosphoglycerate

Important Reaction Patterns in Catabolism
Key Chemical Transformations
Oxidation of Alcohols to Carbon Dioxide: Alcohol → Aldehyde → Acid → CO2
Oxidation of Saturated Bonds: -CH2-CH2- → -CH=CH- → -CO-CH2-
Oxidative Decarboxylation of α-Keto Acids: R-CO-COOH → R-COOH + CO2
These patterns are central to the degradation of carbohydrates, fats, and hydrocarbons in microbial metabolism.
Strategy for Degradation
Steps in Microbial Catabolism
Activate the substrate (often by phosphorylation or CoA attachment).
Rearrange the carbon skeleton to facilitate further breakdown.
Oxidize the activated substrate to extract electrons (reducing power).
Couple reactions to SLP to generate ATP.
Balance redox by reducing another substrate or excreting reduced products.
Excrete waste products (organic acids, alcohols, gases).
Fermentation Balance and Oxidation Numbers
Purpose of Oxidation Numbers
Oxidation numbers are assigned to molecules to account for the conservation of reducing equivalents between the substrate and the products of fermentation. This helps ensure that all products are identified and the redox balance is maintained.
Equation: C + O + H = 0
Rules: O: -2; H: +1; H2: -2
Examples:
CO2: Oxidation number of C = +4
CH4: Oxidation number of C = -4
CH3CH2OH: C = -2



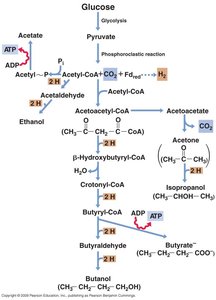
Solvent Production Pathways
Fermentative Pathways and Products
Microbes can convert glucose into a variety of solvents and acids through fermentation. The pathway chosen depends on the organism and environmental conditions.
Key Products: Acetone, butanol, ethanol, butyrate, and others.
ATP Generation: Some steps are coupled to ATP synthesis via SLP.
Butanediol Fermentation Example
Product Distribution and Redox Balance
Butanediol fermentation produces a mixture of acids, alcohols, and gases. The oxidation number analysis ensures that the sum of oxidized and reduced products matches the substrate input, confirming the identification of all products.
Product | mol formed | Oxidation # | Total |
|---|---|---|---|
Formic acid | 17 | +2 | +34 |
Acetic acid | 0.5 | 0 | 0 |
Lactic acid | 2.9 | 0 | 0 |
Ethanol | 69.5 | -4 | -278 |
Butanediol | 66.5 | -6 | -399 |
CO2 | 172 | +4 | +688 |
H2 | 35.5 | -2 | -71 |
Oxidized products | +722 | ||
Reduced products | -748 | ||
Total | -26 | ||
If the oxidized/reduced ratio is close to 1, all products have been identified.

Catabolism of Fats
Fatty Acid Degradation
Fats are catabolized via similar reaction patterns as carbohydrates. Each cycle of fatty acid degradation yields acetyl-CoA, which enters the TCA cycle to generate reducing power for further energy conservation.
Acetyl-CoA: Central intermediate in both catabolism and anabolism.
TCA Cycle: Generates abundant reducing power (NADH, FADH2).
Summary Table: Common High-Energy Bonds
Compound | ΔG' (kcal/mol) |
|---|---|
Phosphoenolpyruvate | -51.6 |
1,3-Bisphosphoglycerate | -49.3 |
ATP | -30.5 |
Acetyl phosphate | -43.3 |
Acetyl-CoA | -31.4 |
ADP | -11.8 |
AMP | -14.2 |
Glucose 6-phosphate | -13.8 |

Conclusion
Microbial catabolism is a complex but highly organized set of chemical reactions that allow cells to extract energy from a wide variety of substrates. Fermentation and substrate-level phosphorylation are central to energy conservation in anaerobic conditions, while respiration provides a more efficient alternative when terminal electron acceptors are available. Understanding the patterns and balances in these pathways is essential for microbiology students.
