 Back
BackMicrobial Electron Flow and Metabolism: Phototrophy, Respiration, and Bioelectricity
Study Guide - Smart Notes

Metabolism II: Electron Flow
Introduction to Electron Flow in Microbial Metabolism
Electron flow is a fundamental process in microbial metabolism, enabling cells to harness energy for growth and maintenance. Microorganisms utilize various sources of electrons, including sunlight and chemical compounds, to drive cellular processes such as photosynthesis and respiration.
Electron flow refers to the movement of electrons through metabolic pathways, powering ATP synthesis and other cellular functions.
Microbial cells can capture energy from the sun (phototrophy) or from chemical compounds (chemotrophy).

Phototrophy and Photosynthesis
Phototrophy: Harnessing Light Energy
Phototrophy is the process by which microorganisms use light energy to excite electrons, which are then used to power cell growth and biosynthesis. While most ecosystems rely on chlorophyll-based photosynthesis, many prokaryotes utilize simpler, ancient forms of phototrophy.
Phototrophy is the harnessing of photoexcited electrons to power cell growth.
Chlorophyll-based photosynthesis is common, but prokaryotes may use bacteriorhodopsin or proteorhodopsin.
Bacteriorhodopsin is a light-driven proton pump found in halophilic archaea.
Photosystems: Types and Functions
The steps of photolysis and electron transport occur in three distinct systems in bacteria:
Anaerobic photosystem I (PS I)
Anaerobic photosystem II (PS II)
Oxygenic Z pathway
Photosystems I and II share common ancestry and are specialized for different electron donors and acceptors.
Photosystem I (PS I)
Found in chlorobia (green sulfur bacteria).
Separates electrons from hydrogen in H2S, organic donors, or reduced iron (Fe2+).
Electrons are transferred to NAD+ or NADP+, forming NADH or NADPH for CO2 fixation and biosynthesis.
Generates a proton gradient for ATP synthesis.
Photosystem II (PS II)
Found in alphaproteobacteria (purple nonsulfur bacteria).
Separates electrons from bacteriochlorophyll itself.
Electrons are transferred to an electron transport system (ETS) and returned to bacteriochlorophyll (cyclic photophosphorylation).
Generates ATP but does not directly produce NADH or NADPH.
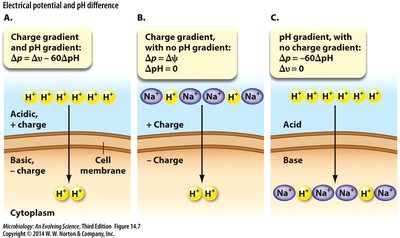
The Proton Motive Force (PMF)
Generation and Components of PMF
The transfer of protons (H+) across a membrane by a proton pump creates an electrochemical gradient known as the proton motive force (PMF). PMF drives ATP synthesis via ATP synthase and powers other cellular processes.
PMF consists of two components:
Electrical potential (Δψ): Separation of charge across the membrane.
pH difference (ΔpH): Log ratio of external to internal H+ concentration.
The relationship is given by:
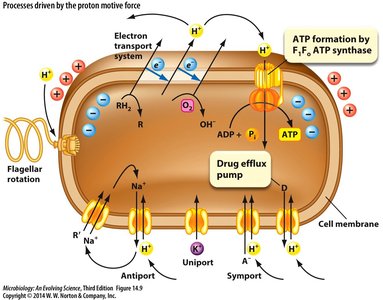
Functions Driven by PMF
Besides ATP synthesis, PMF drives several essential cell functions:
Rotation of flagella
Uptake of nutrients
Efflux of toxic drugs



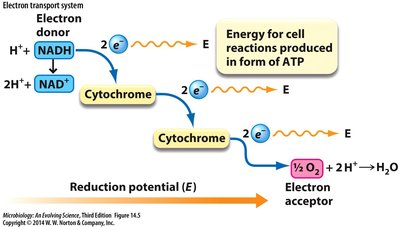
Respiratory Electron Transport System (ETS) and ATP Synthase
Overview of ETS
The respiratory ETS transfers electrons from NADH and FADH2 to O2, producing H2O. Microbes can use alternative electron donors and acceptors. ETS proteins, such as cytochromes, mediate electron transfer via cofactors (metal ions, conjugated rings).
Three main components:
Initial substrate oxidoreductase (dehydrogenase)
Mobile electron carrier (e.g., quinone)
Terminal oxidase
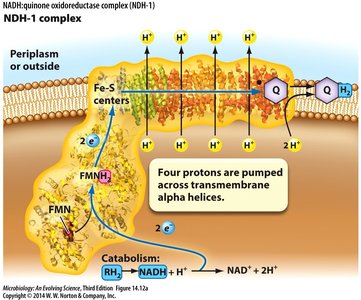
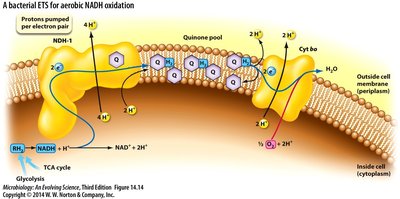
ETS Steps and Proton Pumping
Substrate dehydrogenase receives electrons from NADH or H2.
Electrons are transferred to quinone, coupled with pumping 4H+ across the membrane.
Terminal oxidase complex receives electrons from quinol (QH2), pumps 2H+.
Electrons are transferred to terminal electron acceptor (e.g., O2), forming H2O:
Escherichia coli ETS can pump up to 8H+ per NADH, 6H+ per FADH2.
F1Fo ATP Synthase
Highly conserved protein complex with two parts:
Fo: Embedded in membrane, pumps protons.
F1: Protrudes in cytoplasm, generates ATP.
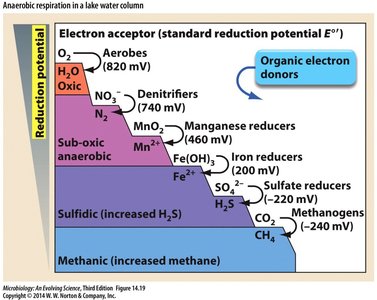
Anaerobic Respiration in Organotrophs
Alternative Electron Acceptors
Obligate aerobes use O2 as terminal electron acceptor.
Prokaryotes can use metals, oxidized ions of nitrogen and sulfur as alternative acceptors.
Anaerobic respiration occurs in oxygen-scarce environments (wetlands, digestive tract).
Reduction of Nitrogen Compounds
Nitrate is reduced in steps:
Each species typically performs one or two steps in the series.
Electron Acceptors in Anaerobic Environments
Different electron acceptors are used as each is depleted, with reduced forms appearing.
Microbial species specialize in different transformations.
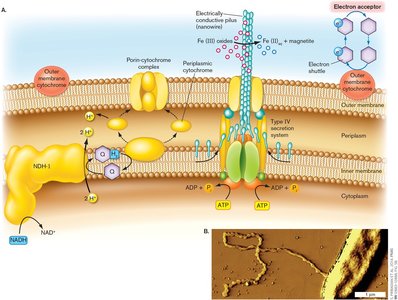
Nanowires, Electron Shuttles, and Fuel Cells
Electron Transfer to Insoluble Acceptors
Metal-reducing bacteria form electrogenic biofilms to transfer electrons to insoluble or distant acceptors, such as Fe3+. These biofilms can be harnessed to generate electricity in fuel cells.
Soluble acceptors (O2, NO3-) diffuse to cells, but metals require specialized mechanisms.
Electrically conductive pili (nanowires) facilitate electron transfer.
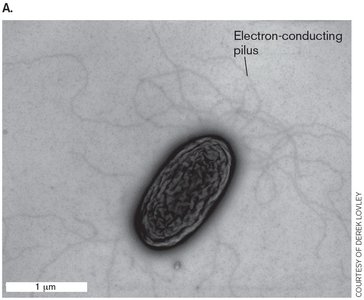
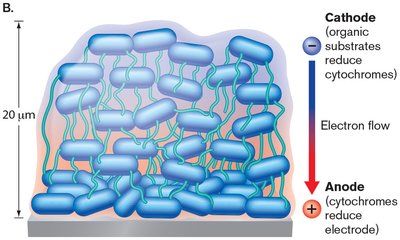
Extracellular Cytochromes, Shuttles, and Cell Extensions
Bacteria such as Geobacter use cytochromes and outer membrane proteins to connect conductive pili to the inner membrane ETS.
Extracellular electron shuttles transfer electrons to distant acceptors.
Species like Shewanella use cytoplasmic extensions to reduce metals in sediments.
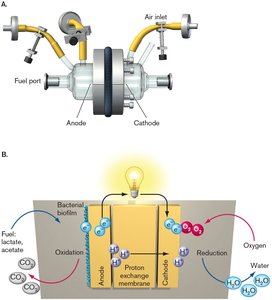
Fuel Cells: Bacterial Electric Power
Bacterial electrical currents can be harnessed to power devices. In electrolytic fuel cells, bacteria form a biofilm on the anode, oxidize organic substances, and transfer electrons to the anode, generating a current.
Fuel can be organic waste or sewage.
Electrons pass through a circuit to a cathode, generating electricity.
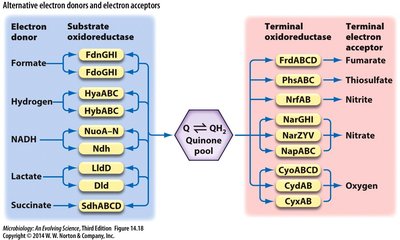
Summary Table: Alternative Electron Donors and Acceptors
Electron Donor | Substrate Oxidoreductase | Terminal Oxidoreductase | Terminal Electron Acceptor |
|---|---|---|---|
Formate | FdnGHI | FrdABCD | Fumarate |
Hydrogen | HybABC | PhsABCD | Thiosulfate |
NADH | NuoA-N, Ndh | NrfAB | Nitrite |
Lactate | LldD | NarZYV, NapABCD | Nitrate |
Succinate | SdhABCD | CyoABCD, CyxAB | Oxygen |
Additional info: Table summarizes key electron donors and acceptors in microbial respiration. |
Key Equations
Proton potential:
Water formation:
Nitrate reduction:
Conclusion
Microbial electron flow is central to energy generation, biosynthesis, and environmental adaptation. Understanding phototrophy, respiration, and bioelectricity provides insight into microbial ecology, biotechnology, and applied microbiology. Additional info: Academic context was added to clarify mechanisms, terminology, and applications for exam preparation.
