 Back
BackLectures 16, 17
Study Guide - Smart Notes

Energy Conservation in Microbes
Microbial Metabolic Diversity
Microorganisms exhibit remarkable metabolic diversity, enabling them to thrive in a wide range of environments. This diversity is reflected in their ability to utilize various energy sources and metabolic pathways for growth and survival.
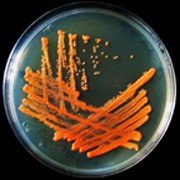
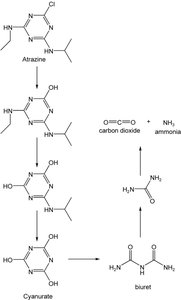
Metabolic Diversity: Microbes can degrade complex compounds such as atrazine, a herbicide, through specialized enzymatic pathways.
Example: Rhodococcus species are known for their ability to degrade atrazine, contributing to environmental bioremediation.

Microbial Iron and Manganese Oxidation
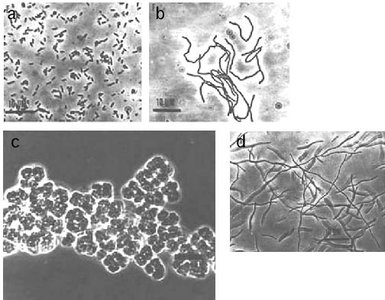
Some microbes oxidize iron (Fe) and manganese (Mn), playing a crucial role in biogeochemical cycling and environmental processes.
Iron Oxidation Reaction:
Formation of Insoluble Precipitate:
Example: Leptothrix discophora is a bacterium involved in iron oxidation in aquatic environments.

Methanogenesis in Archaea
Methanogenic archaea produce methane through the reduction of carbon dioxide, a process significant in anaerobic environments.
Methanogenesis Reaction:
Application: Methanogenesis is important in biogas production and carbon cycling.

Bioenergetics: How Microbes Conserve Energy
Fundamental Concepts of Energy in Cells
Cells require energy to build ordered structures, accelerate reactions, and perform work such as synthesis, transport, and motility. Energy can neither be created nor destroyed, only transformed.
Chemotrophs: Obtain energy from chemicals.
Phototrophs: Harness energy from light.
Entropy: Living systems use energy to maintain order and combat entropy (disorder).
Thermodynamics in Biological Systems
Biological reactions are governed by thermodynamic principles, particularly the concepts of enthalpy, entropy, and free energy.
Free Energy Change (): The energy released that is available to do work.
Equation:
Calculation:
Favorable Reactions: Increase in entropy (negative ), e.g., starch breakdown.
Unfavorable Reactions: Decrease in entropy (positive ), e.g., peptidoglycan synthesis.
Enzymes: Molecular Machines of Cells
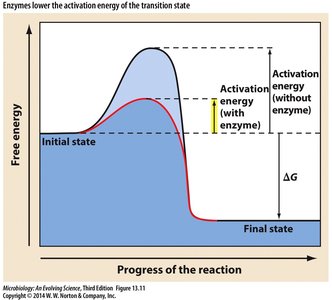
Enzymes are biological catalysts that accelerate reactions by lowering the activation energy required for substrates to reach the transition state.
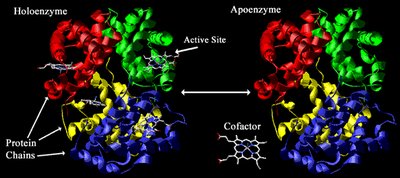
Structure: Enzymes consist of a protein (apoenzyme) and may require a co-catalyst (coenzyme or prosthetic group).
Holoenzyme: The complete, active enzyme with all necessary cofactors.
Activation Energy: Lowered by enzyme binding, facilitating faster reaction rates.
Driving Cellular Reactions
Cells accelerate reactions by manipulating substrate and product concentrations, and by utilizing redox reactions involving electron and proton transfers.
Redox Reactions: Involve the transfer of electrons and protons between molecules.
Substrate Concentration: Increasing substrates or removing products drives reactions forward.
Energy Conservation Mechanisms
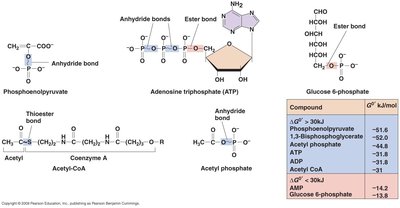
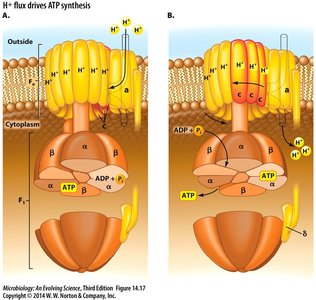
High-Energy Chemicals and ATP Synthesis
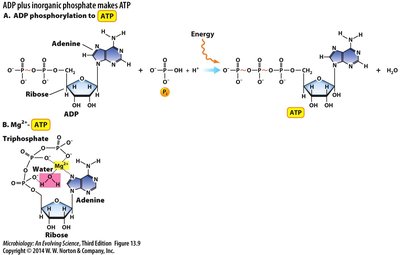
Cells conserve energy in high-energy chemicals, such as ATP, which is synthesized through substrate-level phosphorylation and oxidative phosphorylation.
ATP Formation:
Substrate-Level Phosphorylation: Direct transfer of phosphate to ADP from a high-energy substrate.
Oxidative Phosphorylation: ATP synthesis driven by a proton-motive force (PMF) generated by electron transport.
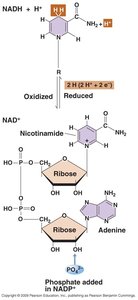
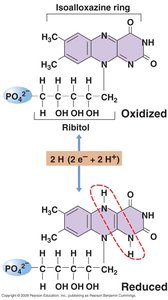
Redox Chemistry and Electron Carriers
Energy conservation in cells relies on redox reactions, where oxidants accept electrons and reductants donate electrons. Electron carriers facilitate these transfers.
Oxidant: Accepts electrons.
Reductant: Donates electrons.
Redox Potential: Indicates the tendency of a molecule to accept or donate electrons.
Redox Tower: Visualizes the energy range and electron transfer tendencies of chemicals in nature.
Types of Catabolism
Microbes utilize different catabolic strategies based on their energy and electron sources.
Photolithoautotrophs: Energy from light, electrons from inorganic sources, carbon from CO2.
Chemoheterotrophs: Energy and electrons from organic compounds, carbon from organics.
Chemolithoautotrophs: Energy and electrons from inorganic chemicals, carbon from CO2.
Electron Transport System (ETS) and Respiration
Oxidative Phosphorylation and ETS
Oxidative phosphorylation is a key process in energy conservation, involving a membrane-bound electron transport system that generates a proton-motive force for ATP synthesis.
Electron Carriers: Include flavins, quinones, iron-sulfur proteins, and cytochromes.
Proton-Motive Force (PMF): Created by proton extrusion across the membrane, driving ATP synthesis.
Terminal Electron Acceptors: Oxygen, nitrate, fumarate, dimethyl sulfoxide, etc.
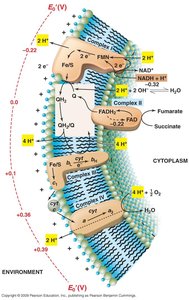
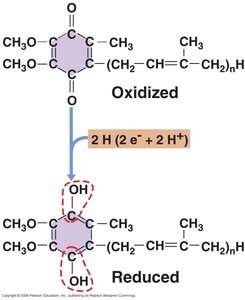
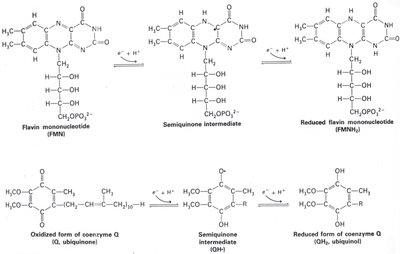
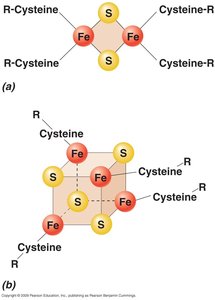
ETS Stepwise Mechanism
The ETS operates through a series of steps, transferring electrons and protons to generate PMF.
Step 1: Protons and electrons transferred to flavoprotein.
Step 2: Electrons move to Fe/S centers and quinone pool; protons are extruded.
Step 3: Electrons transferred to cytochromes and terminal electron acceptor; further proton extrusion.

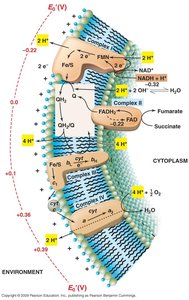
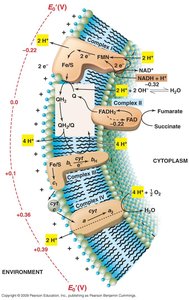
Sulfur and Iron Oxidizers
Microbes can oxidize sulfur and iron compounds, contributing to environmental energy cycles.
Sulfur Oxidizers: Use sulfur compounds as electron donors.
Iron Oxidizers: Use iron compounds as electron donors.
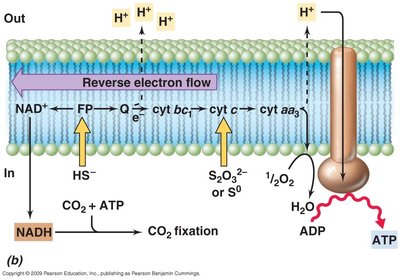
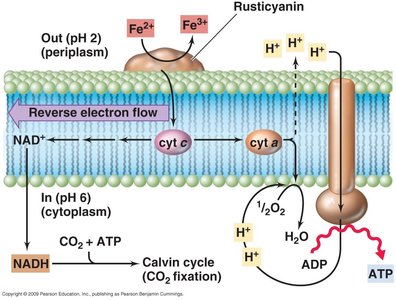
Anaerobic Respiration
Many microbes respire in the absence of oxygen, using alternative electron acceptors and membrane-bound ETS.
Efficiency: Anaerobic respiration is less efficient than aerobic respiration.
Alternative Acceptors: Nitrite (NO2-), nitrate (NO3-), sulfate (SO42-), etc.
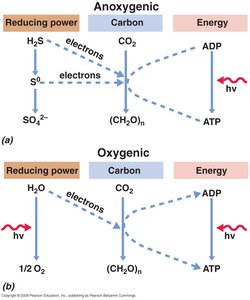
Light-Driven Energy Conservation
Photophosphorylation and Photosynthesis
Microbes can use light to generate ATP and reducing power through photophosphorylation, a photon-driven, PMF-generating process.
ATP Yield: Limited by photon availability and capture efficiency.
Photosynthetic Bacteria: Utilize specialized pigments and structures for light capture.
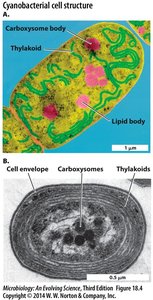
Cyanobacteria and Light Capture
Cyanobacteria possess antennae structures, such as chlorosomes, to efficiently capture light and funnel electrons into the reaction center.
Chlorosome: Super antenna with crystalline arrays of bacteriochlorophyll.
Protein Base Plate: Funnels electrons into the reaction center.
Summary and Key Takeaways
Substrate Activation: Substrates are activated before use.
Reducing Power: Generated through substrate oxidation.
Energy Generation: Occurs after redox reactions and PMF generation.
Non-Redox Reactions: Transform compounds into usable substrates.
Additional info:
Some details about electron carriers and redox potentials were inferred from standard microbiology textbooks to clarify the role of ETS and energy conservation.
