 Back
Back4: Microbial Growth and Its Control – Study Notes
Study Guide - Smart Notes

Microbial Growth and Its Control
4.1 Feeding the Microbe: Cell Nutrition
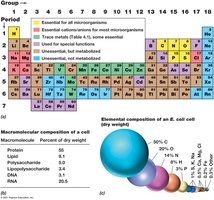
Microbial growth depends on the availability of essential nutrients, which are classified as macronutrients and micronutrients. The chemical composition of microbial cells reflects the dominance of a few key elements and macromolecules.
Macronutrients: Required in large amounts (e.g., C, O, N, H, P, S, K, Mg, Ca, Fe, Na).
Micronutrients: Needed in trace amounts, including trace metals and growth factors (e.g., vitamins, amino acids, purines, pyrimidines).
Elemental composition: C, O, N, H, P, and S make up ~96% of the dry weight of a bacterial cell. Proteins and RNA are the most abundant macromolecules.
Carbon sources: Heterotrophs require organic carbon; autotrophs use CO2 as a carbon source.
Nitrogen sources: Ammonia (NH3), nitrate (NO3−), nitrogen gas (N2), or organic compounds.
Other macronutrients: Phosphorus (nucleic acids, phospholipids), sulfur (amino acids, vitamins), potassium (enzyme activity), magnesium (stabilizes ribosomes, membranes), calcium and sodium (required by some microbes).
Micronutrients: Iron is essential for respiration; trace metals act as enzyme cofactors.
Growth factors: Organic micronutrients, mainly vitamins, required for enzyme function.
4.2 Growth Media and Laboratory Culture
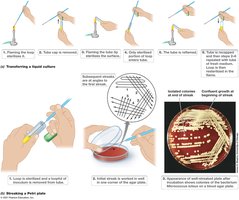
Microorganisms are cultivated in the laboratory using nutrient-rich solutions called culture media. The choice of medium depends on the nutritional requirements of the organism.
Defined media: Exact chemical composition is known.
Complex media: Composed of digests of microbial, animal, or plant products (e.g., yeast extract, meat extract).
Selective media: Inhibit growth of some microbes while allowing others to grow.
Differential media: Contain indicators to distinguish between different metabolic reactions.
Solid media: Prepared by adding agar; used to isolate colonies and assess colony morphology.
Aseptic technique: Essential for transferring cultures without contamination.


4.3 Microscopic Counts of Microbial Cell Numbers
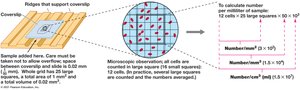
Direct microscopic counting allows for the enumeration of microbial cells in a sample, using specialized counting chambers or stains for visualization.
Total cell count: Cells are counted directly under a microscope using a counting chamber (e.g., Petroff–Hausser chamber).
Staining: Used to differentiate live and dead cells or to identify phylogenetic groups.
Limitations: Cannot distinguish between live and dead cells without special stains; small cells may be missed.
4.4 Viable Counting of Microbial Cell Numbers
Viable (plate) counts estimate the number of living, reproducing cells in a sample by counting colonies formed on agar plates.
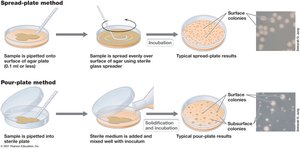
Spread-plate method: Sample is spread on the surface of an agar plate.
Pour-plate method: Sample is mixed with molten agar and poured into a plate.
Serial dilutions: Used to obtain countable numbers of colonies (30–300 per plate).
Colony-forming units (CFU): Accounts for clumps of cells that form a single colony.
Applications: Used in food, water, and clinical microbiology for sensitive detection of viable cells.
Great plate count anomaly: Direct counts often reveal more cells than can be cultured, due to differing growth requirements.

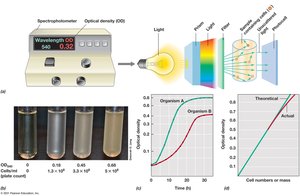
4.5 Turbidimetric Measures of Microbial Cell Numbers
Turbidity measurements estimate cell density in a suspension by measuring light scattering using a spectrophotometer.
Optical density (OD): Proportional to cell number within certain limits; measured at specific wavelengths (e.g., OD540).
Standard curve: Needed to relate OD to actual cell numbers.
Advantages: Rapid, non-destructive, allows repeated measurements.
Limitations: Not suitable for clumped cells or biofilms.
Two Dynamics of Microbial Growth
4.6 Binary Fission and the Microbial Growth Cycle
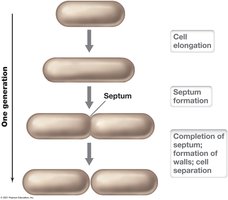
Bacterial growth typically occurs by binary fission, resulting in the doubling of cell numbers at regular intervals. The growth cycle in batch culture is characterized by distinct phases.
Binary fission: Cell divides after enlarging to twice its original size; each daughter cell receives a chromosome and cell constituents.
Generation time (g): Time required for the population to double (e.g., Escherichia coli = 20 min).
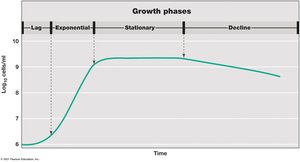
Growth phases in batch culture:
Lag phase: Adaptation to new conditions; biosynthesis of enzymes.
Exponential phase: Cells divide at constant, maximum rate.
Stationary phase: Growth rate slows due to nutrient depletion or waste accumulation; metabolism continues at reduced rate.
Death phase: Cell death exceeds cell division; total cell number declines.
4.7 Quantitative Aspects of Microbial Growth
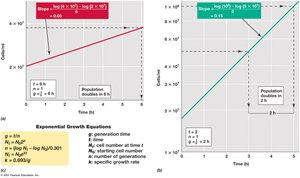
Microbial growth can be described mathematically, allowing for the calculation of generation time and specific growth rate.
Exponential growth: Cell numbers double at regular intervals; plotted as a straight line on a semilogarithmic graph.
Mathematical relationship:
Specific growth rate (k): Expresses the rate of growth at any instant.
Consequences: Exponential growth leads to rapid increases in cell numbers.

4.8 Continuous Culture
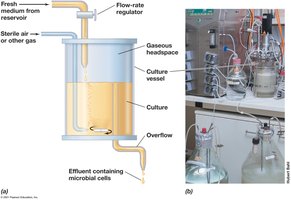
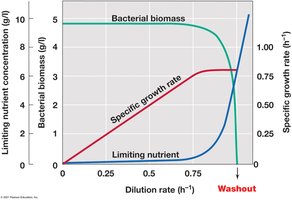
Continuous culture systems, such as the chemostat, allow for the maintenance of microbial populations in exponential growth for extended periods.
Chemostat: Fresh medium is continuously added, and spent medium is removed at the same rate, maintaining steady-state conditions.
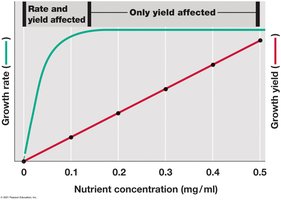
Control: Both growth rate and population density can be independently controlled by adjusting dilution rate and limiting nutrient concentration.
Applications: Used to study microbial physiology, ecology, and evolution.
4.9 Biofilm Growth
Microbes can grow as free-floating (planktonic) cells or as surface-attached communities called biofilms, which have distinct properties and implications for health and industry.
Biofilm: Cells embedded in a self-produced polysaccharide matrix attached to a surface.
Stages of biofilm formation: Attachment, colonization, development, dispersal.
Importance: Biofilms are involved in medical device infections, dental plaque, and industrial fouling.


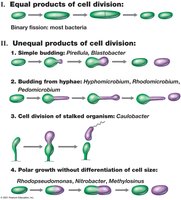
4.10 Alternatives to Binary Fission
Some bacteria divide by mechanisms other than binary fission, such as budding or hyphal growth, leading to diverse cell morphologies and reproductive strategies.
Budding: Unequal cell division, sometimes with formation of stalks or appendages.
Hyphal growth: Filamentous bacteria (e.g., Streptomyces) grow by extending hyphae; mycelia are networks of hyphae.
Multiple fission: Hyphal filaments form many septa simultaneously, producing multiple daughter cells.
Three Environmental Effects on Growth: Temperature
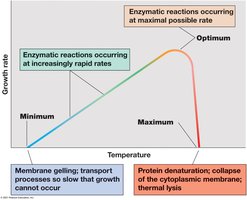
4.11 Temperature Classes of Microorganisms
Microorganisms are classified based on their temperature growth optima, which reflect adaptations to their environments.
Cardinal temperatures: Minimum, optimum, and maximum temperatures for growth; range is typically less than 40°C for a given species.
Temperature classes:
Psychrophiles: Optimum ≤ 15°C; found in cold environments.
Mesophiles: Optimum 20–45°C; most common, includes human pathogens.
Thermophiles: Optimum 45–80°C; found in hot environments.
Hyperthermophiles: Optimum >80°C; found in extremely hot habitats.
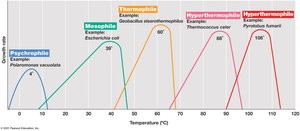
4.12 Microbial Life in the Cold
Psychrophilic and psychrotolerant microorganisms have evolved molecular adaptations to thrive in cold environments.
Psychrophiles: True cold-loving microbes; optimal growth at ≤ 15°C, maximum < 20°C.
Psychrotolerant: Can grow at 0°C but have higher optima (20–40°C).
Adaptations: Enzymes with more α-helices, more polar amino acids, flexible membranes with unsaturated fatty acids, production of cryoprotectants and exopolysaccharides.

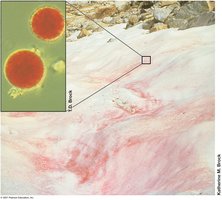
4.13 Microbial Life at High Temperatures
Thermophiles and hyperthermophiles possess unique adaptations for survival at high temperatures, including heat-stable proteins and membranes.
Thermophiles: Optimum 45–80°C; hyperthermophiles: >80°C.
Adaptations: Heat-stable enzymes (e.g., Taq polymerase), increased ionic bonding, hydrophobic protein interiors, specialized membrane lipids (e.g., lipid monolayers in Archaea).
Applications: Thermostable enzymes are valuable in biotechnology.


Four Environmental Effects on Growth: pH, Osmolarity, and Oxygen
4.14 Effects of pH on Microbial Growth
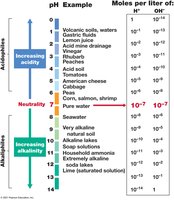
Microorganisms are classified by their pH growth optima, and must maintain cytoplasmic pH near neutrality for stability.
Neutrophiles: Optimum pH 5.5–7.9.
Acidophiles: Optimum pH < 5.5; require protons for membrane stability.
Alkaliphiles: Optimum pH ≥ 8; found in soda lakes, alkaline soils.
Buffers: Used in media to maintain constant pH.
4.15 Osmolarity and Microbial Growth
Water availability, expressed as water activity (aw), is critical for microbial growth. Microbes adapt to varying osmotic conditions by accumulating compatible solutes.
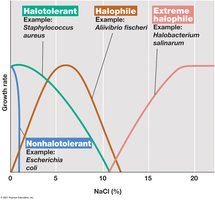
Halophiles: Require NaCl for growth; extreme halophiles need very high salt concentrations.
Halotolerant: Tolerate some salt but grow best without it.
Osmophiles: Thrive in high-sugar environments; xerophiles in dry environments.
Compatible solutes: Small molecules (e.g., glycine betaine, KCl, sugars) accumulated to maintain water balance without inhibiting cellular processes.
4.16 Oxygen and Microbial Growth
Microorganisms vary in their oxygen requirements and tolerance, which is reflected in their growth patterns and enzymatic defenses against toxic oxygen species.
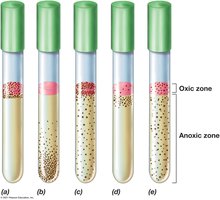
Aerobes: Require oxygen for growth.
Microaerophiles: Grow best at low oxygen concentrations.
Facultative organisms: Can grow with or without oxygen.
Anaerobes: Do not require oxygen; obligate anaerobes are killed by oxygen, aerotolerant anaerobes tolerate but do not use oxygen.
Culture techniques: Use reducing agents, specialized media, and anaerobic chambers to grow anaerobes.
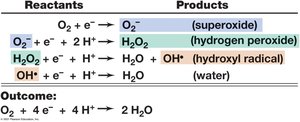
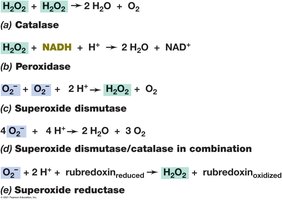
Toxic oxygen species: Superoxide anion (O2−), hydrogen peroxide (H2O2), hydroxyl radical; destroyed by enzymes such as catalase, peroxidase, and superoxide dismutase.
Five Controlling Microbial Growth
4.17 General Principles and Microbial Growth Control by Heat
Microbial growth can be controlled by physical and chemical methods, with heat sterilization being the most common approach.
Decontamination: Making objects safe to handle.
Disinfection: Targets pathogens, not all microbes.
Heat sterilization: Kills microbes exponentially faster at higher temperatures; moist heat is more effective than dry heat.
Decimal reduction time (D): Time required to reduce viability by 90% at a given temperature.
Autoclave: Uses steam under pressure (121°C) to sterilize; kills endospores.
Pasteurization: Reduces microbial load in heat-sensitive liquids; does not sterilize but kills pathogens.


4.18 Other Physical Control Methods: Radiation and Filtration
Radiation and filtration are alternative physical methods for controlling microbial growth, especially for heat-sensitive materials.
Ultraviolet (UV) radiation: Damages DNA; used for surface decontamination.
Ionizing radiation: Produces ions and reactive molecules; used for sterilizing medical supplies and food.
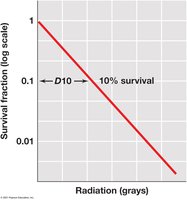
D10 value: Dose required to reduce population by 90%.
Filtration: Removes microbes from liquids and gases using depth or membrane filters; HEPA filters remove particles from air.

4.19 Chemical Control of Microbial Growth
Chemical agents are used to kill or inhibit microorganisms on surfaces, in liquids, or on living tissues.
Antimicrobial agents: Classified as -cidal (kill) or -static (inhibit growth).
Bacteriostatic: Inhibit growth without killing; bactericidal: kill cells; bacteriolytic: kill by lysis.
Minimum inhibitory concentration (MIC): Lowest concentration that inhibits growth.
Disk diffusion assay: Used to assess antimicrobial susceptibility; zone of inhibition indicates effectiveness.
Types of agents: Sterilants (destroy all microbes), disinfectants (kill most, not endospores), sanitizers (reduce numbers), antiseptics (safe for living tissues).
