 Back
BackMicrobial Mechanisms of Pathogenicity: Study Notes
Study Guide - Smart Notes

Microbial Mechanisms of Pathogenicity
Introduction
This chapter explores the strategies used by microorganisms to cause disease in hosts, focusing on the mechanisms of pathogenicity, portals of entry and exit, and the molecular and cellular interactions that determine the outcome of infection.
Pathogenicity and Virulence
Definitions and Concepts
Pathogenicity: The ability of a microorganism to cause disease.
Virulence: The degree of pathogenicity; a measure of the severity of disease caused by a microbe.
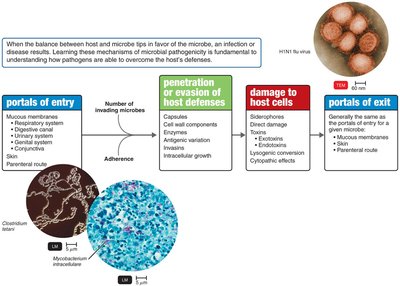
Portals of Entry
Major Portals of Entry
Mucous membranes: Includes the respiratory tract (most common), digestive canal, genitourinary system, and conjunctiva. Pathogens enter via inhalation, ingestion, or sexual contact.
Skin: Generally impenetrable unless compromised; entry may occur through hair follicles or sweat gland ducts.
Parenteral route: Direct deposition into tissues beneath the skin or mucous membranes, such as through punctures, injections, bites, cuts, or surgery.
Most pathogens have a preferred portal of entry that is critical for their ability to cause disease.
Numbers of Invading Microbes
ID50 and LD50
ID50: Infectious dose for 50% of a sample population; measures the virulence of a microbe.
LD50: Lethal dose for 50% of a sample population; measures the potency of a toxin.
Example: Bacillus anthracis has different ID50 values depending on the portal of entry (skin: 10–50 endospores; inhalation: 10,000–20,000; ingestion: 250,000–1,000,000).
Botulinum toxin is extremely potent, with an LD50 of 0.03 ng/kg.
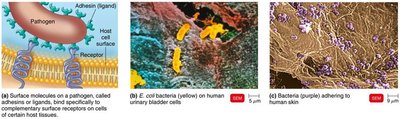
Adherence to Host Cells
Mechanisms of Adherence
Pathogens attach to host tissues via adherence (adhesion).
Adhesins (ligands) on the pathogen bind to specific receptors on host cells.
Examples:
Glycocalyx of Streptococcus mutans enables adherence to teeth.
Fimbriae of Actinomyces adhere to the glycocalyx of S. mutans.
Viral spikes (e.g., SARS-CoV-2) bind to ACE2 receptors on host cells.
Penetration or Evasion of Host Defenses
Capsules and Cell Wall Components
Capsules: Glycocalyx layers that impair phagocytosis (e.g., Streptococcus pneumoniae, Bacillus anthracis).
M protein: Resists phagocytosis (Streptococcus pyogenes).
Opa protein: Facilitates attachment to host cells (Neisseria gonorrhoeae).
Mycolic acid: Waxy lipid in cell wall resists digestion (Mycobacterium tuberculosis).
Enzymes as Virulence Factors
Coagulases: Coagulate fibrinogen to form clots.
Kinases: Digest fibrin clots.
Hyaluronidase: Digests hyaluronic acid, aiding tissue penetration.
Collagenase: Breaks down collagen.
IgA proteases: Destroy IgA antibodies.
Antigenic Variation
Pathogens alter their surface antigens to evade immune detection (e.g., influenza virus, Neisseria gonorrhoeae, Trypanosoma brucei gambiense).
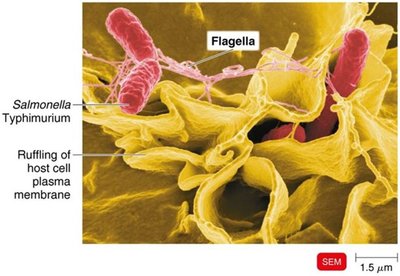
Penetration into Host Cells
Invasins: Bacterial surface proteins that rearrange host actin filaments, causing membrane ruffling and bacterial engulfment (e.g., Shigella, Listeria).
Bacteria may use actin to move between cells.
Some survive inside phagocytes by escaping the phagosome, preventing lysosomal fusion, or tolerating low pH.
Biofilms
Biofilms resist antibiotics and disinfectants, are involved in 65% of infections, and shield bacteria from phagocytosis via extracellular polymeric substances (EPS).
How Bacterial Pathogens Damage Host Cells
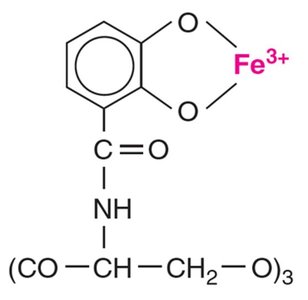
Using the Host’s Nutrients: Siderophores
Pathogens secrete siderophores to bind iron more tightly than host proteins, facilitating iron uptake essential for bacterial growth.
Direct Damage
Pathogens disrupt host cell function, use host nutrients, produce waste products, and may cause cell rupture by multiplying inside cells.
Production of Toxins
Toxins: Poisonous substances produced by microorganisms, causing fever, cardiovascular issues, diarrhea, and shock.
Toxigenicity: Ability to produce toxins.
Toxemia: Presence of toxins in the blood.
Intoxications: Disease caused by toxins without microbial growth.
Exotoxins
Proteins secreted by bacteria, usually highly specific and potent.
Can be neutralized by antitoxins or inactivated as toxoids for vaccines.
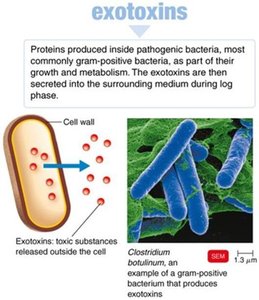
Types of Exotoxins
A-B toxins: Consist of an active (A) and binding (B) component (e.g., diphtheria toxin).
Genotoxins: Damage DNA, potentially leading to cancer.
Membrane-disrupting toxins: Lyse host cells by disrupting membranes (e.g., leukocidins, hemolysins, streptolysins).
Superantigens: Trigger excessive immune responses, causing fever, shock, and death.
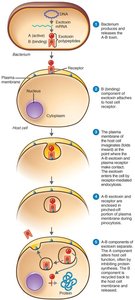
Diseases Caused by Exotoxins
Disease | Bacterium | Type of Exotoxin | Mechanism |
|---|---|---|---|
Botulism | Clostridium botulinum | A-B | Neurotoxin; prevents nerve impulse transmission; flaccid paralysis. |
Tetanus | C. tetani | A-B | Neurotoxin; blocks muscle relaxation; uncontrollable contractions. |
Diphtheria | Corynebacterium diphtheriae | A-B | Cytotoxin; inhibits protein synthesis. |
Cholera | V. cholerae | A-B | Enterotoxin; causes diarrhea. |
Anthrax | Bacillus anthracis | A-B | Shock and immune suppression. |
Food poisoning | S. aureus | Superantigen | Enterotoxin; diarrhea. |
Toxic shock syndrome | S. aureus | Superantigen | Shock, decreased blood volume. |
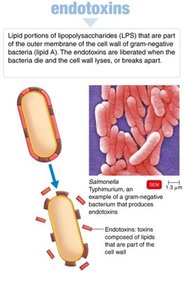
Endotoxins
Lipid A portion of lipopolysaccharides (LPS) in the outer membrane of gram-negative bacteria.
Released during bacterial death or cell wall lysis.
Cause fever, shock, disseminated intravascular coagulation, and may weaken the blood-brain barrier.
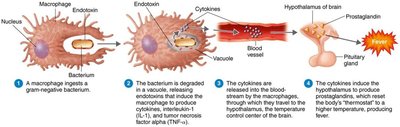
Limulus Amebocyte Lysate (LAL) Assay
Used to detect endotoxins; based on the clotting reaction of horseshoe crab blood in the presence of endotoxin.
Comparison of Exotoxins and Endotoxins
Property | Exotoxins | Endotoxins |
|---|---|---|
Bacterial Source | Gram-positive and gram-negative | Gram-negative |
Chemistry | Proteins (A-B structure) | Lipid A of LPS |
Heat Stability | Unstable (destroyed at 60–80°C) | Stable (withstands 121°C) |
Toxicity | High | Low |
Fever-Producing | No | Yes |
Immunology | Can be neutralized by antitoxin; toxoids for vaccines | Not easily neutralized; no effective toxoids |
Lethal Dose | Small | Large |
Representative Diseases | Tetanus, botulism, diphtheria | Typhoid fever, meningococcal meningitis |
Plasmids, Lysogeny, and Pathogenicity
Plasmids: May carry genes for toxins, antibiotic resistance, and virulence factors.
Lysogenic conversion: Incorporation of a prophage can confer new properties, such as toxin production (e.g., diphtheria toxin).
Pathogenic Properties of Viruses
Mechanisms of Viral Pathogenicity
Viruses evade host defenses by intracellular location, using host cell surface molecules, attacking immune components, methylating RNA, and antigenic variation.
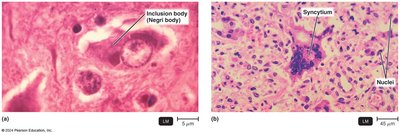
Cytopathic Effects (CPE)
Visible effects of viral infection on host cells; can be cytocidal (cell death) or noncytocidal (cell damage).
Examples:
Disruption of cell junctions
Cytokine storm induction
Inclusion body formation (e.g., Negri bodies in rabies)
Syncytium formation (fusion of cells)
Antigenic changes, chromosomal damage, loss of contact inhibition (cancer)
Interferon production
Interferons
Alpha and beta interferons are produced by infected cells to protect neighboring cells by inhibiting viral protein synthesis and inducing apoptosis.
Some viruses can evade interferon responses.
Pathogenic Properties of Fungi, Protozoa, Helminths, and Algae
Fungi
Produce toxic metabolic products, provoke allergic responses, and secrete proteases that modify host membranes.
Capsules (e.g., Cryptococcus neoformans) prevent phagocytosis.
Mycotoxins (e.g., aflatoxin, ergot alkaloids) can be carcinogenic or neurotoxic.
Protozoa
Cause symptoms by their presence and waste products.
Evade defenses by digesting cells, growing in phagocytes, and antigenic variation (e.g., Trypanosoma).
Helminths
Use host tissues for growth, produce large masses, and release waste products that cause symptoms.
Algae
Some produce neurotoxins (e.g., saxitoxin from Alexandrium), causing paralytic shellfish poisoning.
Portals of Exit
Major Portals of Exit
Respiratory tract: Coughing and sneezing
Gastrointestinal tract: Feces and saliva
Genitourinary tract: Urine and genital secretions
Skin
Blood: Via arthropod bites or contaminated needles
Portals of exit are generally the same as the portals of entry for a given microbe.
