 Back
BackMicrobial Metabolism: An Overview of Energy and Enzyme Function in Microbiology
Study Guide - Smart Notes

Microbial Metabolism
Introduction to Metabolism
Metabolism encompasses all the chemical reactions that occur within a cell, enabling the buildup and breakdown of nutrients. These reactions provide energy and create substances essential for sustaining life. Microbial metabolism is not only central to cell survival but also has significant implications for disease, food spoilage, and beneficial industrial processes.
Catabolism: The breakdown of complex molecules into simpler ones, releasing energy.
Anabolism: The synthesis of complex molecules from simpler ones, requiring energy input.
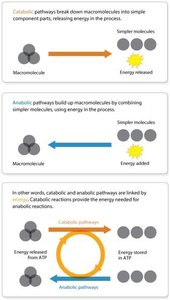
Catabolic and Anabolic Pathways
Catabolic and anabolic reactions are interconnected through the molecule ATP, which acts as an energy currency in the cell. Catabolic reactions release energy, some of which is stored in ATP, while anabolic reactions consume ATP to build macromolecules.
Catabolic Pathways: Exergonic (energy-releasing) processes, such as the breakdown of glucose during cellular respiration.
Anabolic Pathways: Endergonic (energy-consuming) processes, such as protein synthesis.
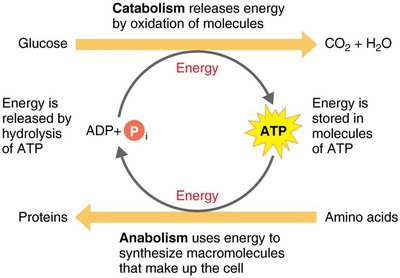
The Role of ATP in Metabolism
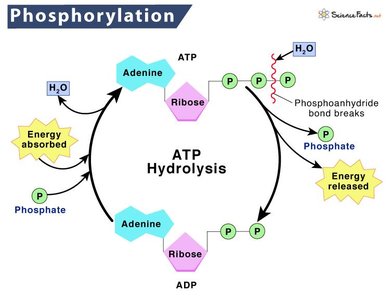
ATP (adenosine triphosphate) is the primary energy carrier in cells. It couples catabolic and anabolic reactions by storing energy from catabolic reactions and providing it for anabolic processes. The hydrolysis of ATP releases energy that can be used for cellular work, including mechanical, transport, and chemical activities.
ATP Hydrolysis:
Phosphorylation: The process of adding a phosphate group to a molecule, often making it more reactive.

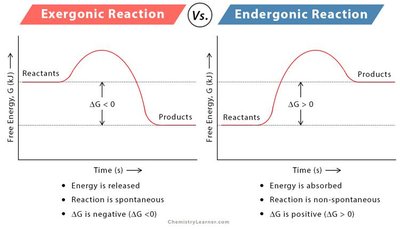
Enzymes and Metabolic Pathways
Enzymes: Biological Catalysts
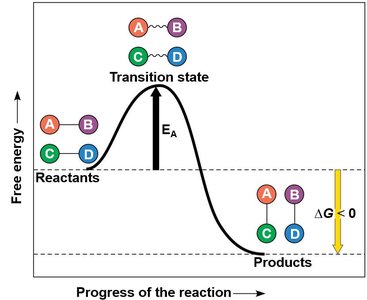
Enzymes are proteins (or RNA molecules, in the case of ribozymes) that catalyze biochemical reactions, increasing their rate without being consumed. Each enzyme is specific to a particular substrate and lowers the activation energy required for the reaction.
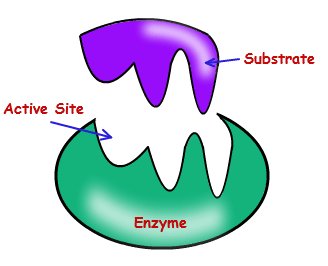
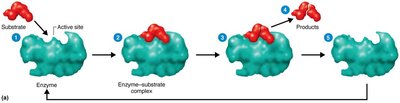
Active Site: The region on the enzyme where the substrate binds.
Enzyme-Substrate Complex: The intermediate formed when an enzyme binds its substrate.
Turnover Number: The number of substrate molecules converted to product per second by an enzyme.
Enzyme Components
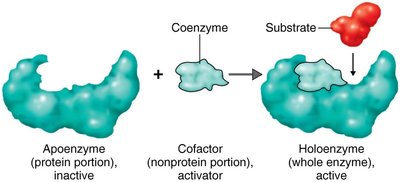
Enzymes may require additional non-protein components to be active:
Apoenzyme: The protein portion of an enzyme (inactive alone).
Cofactor: A non-protein component (can be a metal ion or organic molecule).
Coenzyme: An organic cofactor, often derived from vitamins (e.g., NAD+, FAD).
Holoenzyme: The complete, active enzyme with its cofactor.
Factors Influencing Enzyme Activity
Enzyme activity is affected by several factors:
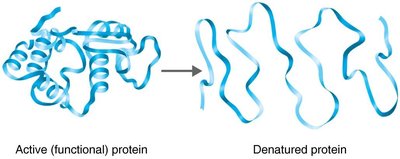
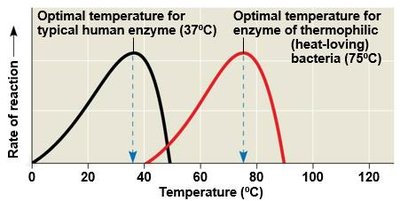
Temperature: Each enzyme has an optimal temperature; high temperatures can denature enzymes.
pH: Each enzyme has an optimal pH range.
Substrate Concentration: Enzyme activity increases with substrate concentration until saturation is reached.
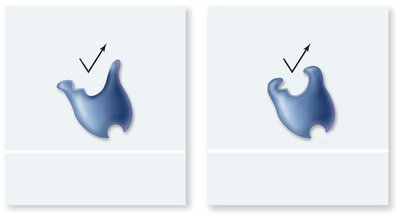
Inhibitors: Substances that decrease enzyme activity, either competitively (binding the active site) or noncompetitively (binding elsewhere and altering enzyme shape).

Enzyme Inhibition and Regulation
Enzyme activity can be regulated by inhibitors:
Competitive Inhibitors: Compete with the substrate for the active site.
Noncompetitive Inhibitors: Bind to an allosteric site, changing the enzyme's shape and function.
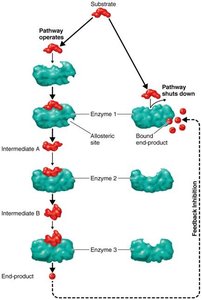
Feedback Inhibition: The end product of a metabolic pathway inhibits an enzyme involved earlier in the pathway, preventing overproduction.
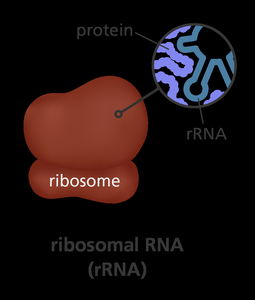
Ribozymes
Ribozymes are RNA molecules with catalytic activity, primarily involved in RNA processing and protein synthesis within ribosomes.
Energy Production and Redox Reactions
Oxidation-Reduction (Redox) Reactions
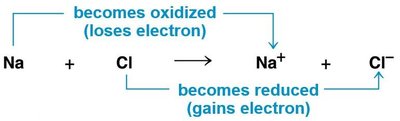
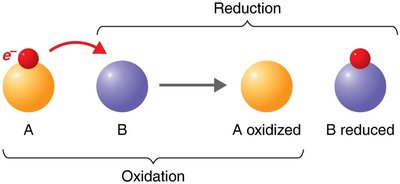
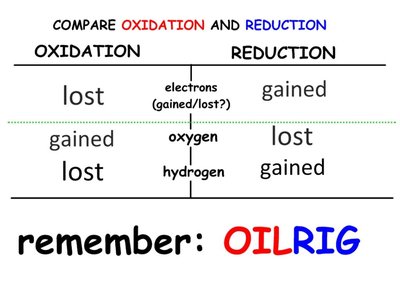
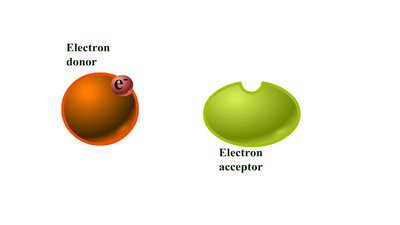
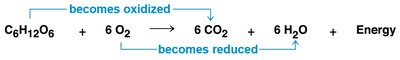
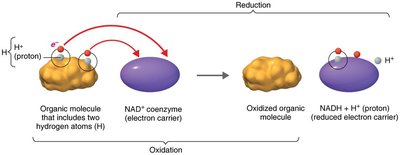
Redox reactions involve the transfer of electrons between molecules, releasing energy used to synthesize ATP. In oxidation, a molecule loses electrons (and often hydrogen), while in reduction, a molecule gains electrons (and often hydrogen).
OIL RIG: Oxidation Is Loss, Reduction Is Gain (of electrons).
ATP Generation Mechanisms
Substrate-Level Phosphorylation: Direct transfer of a phosphate group to ADP from a phosphorylated intermediate.
Oxidative Phosphorylation: ATP is generated as electrons are transferred through the electron transport chain to a final electron acceptor (usually O2), coupled with chemiosmosis.
Photophosphorylation: Light energy is used to generate ATP in photosynthetic organisms.
Carbohydrate Catabolism
Overview of Carbohydrate Catabolism
Carbohydrate catabolism is the process by which cells break down carbohydrates to release energy. The main stages are glycolysis, oxidation of pyruvate, the Krebs cycle, and the electron transport chain.
Glycolysis: Glucose is broken down into pyruvate, producing ATP and NADH.
Pentose Phosphate Pathway: Generates NADPH and pentoses for biosynthesis.
Entner-Doudoroff Pathway: Alternative pathway in some bacteria, producing NADPH and ATP.
Krebs Cycle (Citric Acid Cycle)
The Krebs cycle completes the oxidation of organic molecules, generating ATP, NADH, and FADH2. Each turn of the cycle processes one acetyl-CoA, and two turns are required per glucose molecule.
Products per turn: 1 ATP, 3 NADH, 1 FADH2, CO2
Electron Transport Chain and Chemiosmosis
The electron transport chain (ETC) is a series of carrier molecules that transfer electrons, releasing energy used to pump protons and generate ATP via chemiosmosis. The final electron acceptor is oxygen in aerobic respiration, or another inorganic molecule in anaerobic respiration.
Theoretical ATP yield: 38 ATP per glucose in prokaryotes, 36 ATP in eukaryotes (actual yield is lower).
Fermentation
Fermentation is an anaerobic process that allows glycolysis to continue by regenerating NAD+. It produces small amounts of ATP and various end products, such as lactic acid or ethanol.
Lactic Acid Fermentation: Pyruvate is reduced to lactic acid.
Alcohol Fermentation: Pyruvate is converted to ethanol and CO2.
Lipid and Protein Catabolism
Catabolism of Lipids and Proteins
Lipids and proteins can also be catabolized for energy. Lipids are broken down into glycerol and fatty acids, while proteins are broken down into amino acids. These intermediates enter glycolysis or the Krebs cycle at various points.
Biochemical Tests and Bacterial Identification
Biochemical Tests
Biochemical tests are used in microbiology to identify bacteria based on their metabolic capabilities, such as fermentation of sugars or the presence of specific enzymes (e.g., oxidase test).
Photosynthesis and Metabolic Diversity
Photosynthesis
Photosynthesis is the process by which light energy is converted into chemical energy. It includes light-dependent reactions (producing ATP and NADPH) and light-independent reactions (Calvin-Benson cycle, fixing CO2 into sugars).
Metabolic Diversity Among Organisms
Phototrophs: Use light as an energy source.
Photoautotrophs: Use light and CO2 as a carbon source.
Photoheterotrophs: Use light for energy but require organic compounds for carbon.
Chemoautotrophs: Obtain energy from inorganic chemicals and use CO2 as a carbon source.
Chemoheterotrophs: Obtain both energy and carbon from organic compounds.
Integration of Metabolism
Amphibolic Pathways
Amphibolic pathways are metabolic pathways that function in both anabolism and catabolism. The Krebs cycle is a prime example, as it provides intermediates for both energy production and biosynthesis.
