 Back
BackMicrobial Metabolism and Energetics: An Overview
Study Guide - Smart Notes

Microbial Metabolism: Overview
Definition and Types of Metabolism
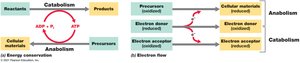
Metabolism encompasses all biochemical reactions that sustain life, divided into two main categories: catabolism and anabolism.
Catabolism: The breakdown of molecules to obtain energy and reducing power. These reactions are typically exergonic, releasing free energy.
Anabolism: The synthesis of cellular materials from simpler precursors, requiring energy input (endergonic reactions).
Free energy (ΔG) is the energy available to do work. Exergonic reactions (ΔG < 0) release energy, while endergonic reactions (ΔG > 0) require energy input, often in the form of ATP.
Bioenergetics and Redox Reactions
Oxidation-Reduction (Redox) Reactions
Redox reactions are central to energy production in cells. They involve the transfer of electrons from an electron donor (oxidized) to an electron acceptor (reduced).
Oxidation: Loss of electrons; the molecule becomes more oxidized.
Reduction: Gain of electrons; the molecule becomes more reduced.
The reduction potential (E0') measures a substance's tendency to accept electrons, expressed in volts (V). Electrons flow from donors with more negative E0' to acceptors with more positive E0'.

Example: Glucose oxidation with O2 as the electron acceptor yields more free energy than with NO3- as the acceptor.
Electron Carriers
Electron transfer in cells often involves carriers such as NAD+ and FAD. These molecules shuttle electrons between metabolic pathways, facilitating redox reactions.
Enzymes and Catalysis
Enzyme Structure and Function
Enzymes are biological catalysts, usually proteins, that accelerate chemical reactions by lowering the activation energy. They are highly specific for their substrates and are not consumed in the reaction.
Contain active sites for substrate binding.
Form temporary transition-state complexes with substrates.
Some enzymes require cofactors:
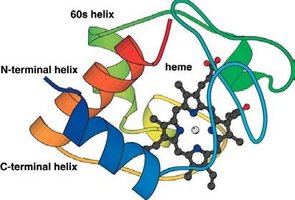
Prosthetic groups: Tightly bound, e.g., heme in cytochromes.
Coenzymes: Loosely bound, e.g., NAD+.
Enzyme Classifications
Enzymes are classified by the type of reaction they catalyze:
Class | Function | Example |
|---|---|---|
Oxidoreductases | Redox reactions | Lactate dehydrogenase |
Transferases | Transfer functional groups | Hexokinase |
Hydrolases | Hydrolysis reactions | Invertase |
Lyases | Cleavage without hydrolysis | Aldolase |
Isomerases | Isomerization | Phosphoglucose isomerase |
Ligases | Bond formation with ATP hydrolysis | DNA ligase |
Examples of Enzyme-Catalyzed Reactions
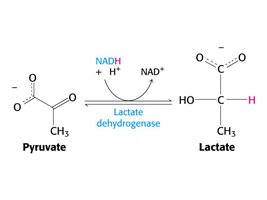
Oxidoreductase: Lactate dehydrogenase catalyzes the reduction of pyruvate to lactate, regenerating NAD+ from NADH.
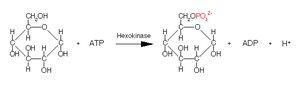
Transferase: Hexokinase transfers a phosphate group from ATP to glucose, forming glucose-6-phosphate.
Hydrolase: Invertase hydrolyzes sucrose into glucose and fructose.
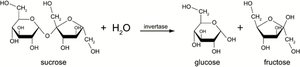
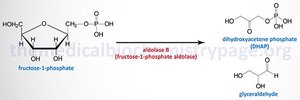
Lyase: Aldolase splits fructose-1-phosphate into dihydroxyacetone phosphate and glyceraldehyde.
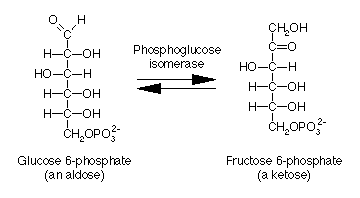
Isomerase: Phosphoglucose isomerase converts glucose-6-phosphate to fructose-6-phosphate.
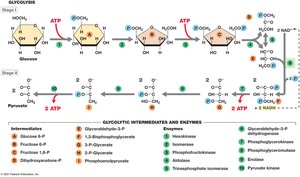
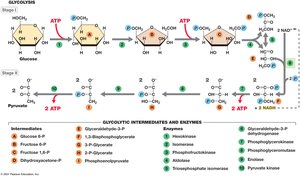
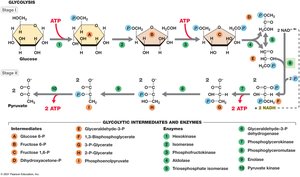
Glycolysis (Embden–Meyerhof–Parnas Pathway)
Overview and Stages
Glycolysis is the primary pathway for glucose catabolism in most organisms. It is an anaerobic process that converts glucose to pyruvate, generating ATP and NADH.
Stage I: Energy Investment – 2 ATP are consumed to phosphorylate glucose and its intermediates.
Stage II: Energy Conservation – 4 ATP are produced (net gain of 2 ATP), and 2 NAD+ are reduced to 2 NADH.
Fermentation
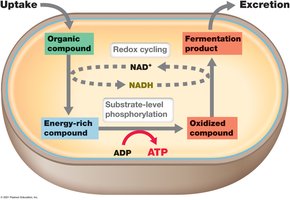
Fate of Pyruvate and NAD+ Regeneration
After glycolysis, pyruvate can undergo fermentation or respiration. Fermentation occurs in the absence of oxygen and allows cells to regenerate NAD+ from NADH, enabling glycolysis to continue.
Homolactic fermentation: Pyruvate is reduced to lactic acid (e.g., in lactic acid bacteria).
Alcoholic fermentation: Pyruvate is converted to ethanol and CO2 (e.g., in yeast).
Mixed acid fermentation: Produces a variety of acids and gases (e.g., in enteric bacteria).
Fermentation yields few ATP compared to respiration but is essential for anaerobic energy production.
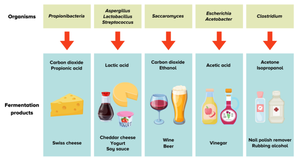
Useful Products from Microbial Fermentations
Microbial fermentations are exploited for the production of foods and beverages such as cheese, yogurt, wine, beer, vinegar, and solvents.
Respiration
Overview of Respiration
Respiration is a process that completely oxidizes pyruvate to CO2, generating more ATP than fermentation. It involves:
Glycolysis
The Citric Acid Cycle (Krebs or TCA cycle)
Electron Transport Chain (ETC)
Oxidative Phosphorylation
Citric Acid Cycle
The citric acid cycle generates reduced coenzymes (NADH, FADH2), ATP, and biosynthetic intermediates. In prokaryotes, it occurs in the cytoplasm; in eukaryotes, in the mitochondria.
Electron Transport Chain and Proton Motive Force
The ETC consists of a series of electron carriers embedded in the cytoplasmic membrane (in prokaryotes). Electrons from NADH and FADH2 are transferred through the chain to a terminal electron acceptor (often O2), generating a proton motive force (PMF) across the membrane.
PMF drives ATP synthesis via ATP synthase (oxidative phosphorylation).
ETC regenerates oxidized coenzymes (NAD+, FAD).
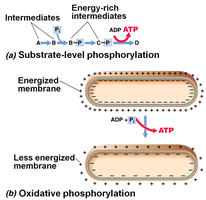
ATP Synthesis: Substrate-Level vs. Oxidative Phosphorylation
Type | Mechanism | Location |
|---|---|---|
Substrate-level phosphorylation | Direct transfer of phosphate to ADP from a phosphorylated intermediate | Glycolysis, Citric Acid Cycle |
Oxidative phosphorylation | ATP synthesis driven by PMF generated by ETC | Cell membrane (prokaryotes), mitochondria (eukaryotes) |
Energetics: Fermentation vs. Respiration
Fermentation: Low ATP yield (2 ATP per glucose), incomplete oxidation of glucose.
Respiration: High ATP yield (up to 38 ATP per glucose in prokaryotes), complete oxidation of glucose to CO2.
Summary
Microbial metabolism includes catabolic and anabolic reactions, with energy and electron flow at its core.
Enzymes catalyze metabolic reactions, often requiring cofactors.
Glycolysis and fermentation are central to anaerobic energy production, while respiration enables maximal ATP yield via the citric acid cycle and electron transport chain.
