 Back
BackMicrobial Metabolism and Energy Generation: Pathways, Fermentation, and Electron Transport
Study Guide - Smart Notes

Microbial Metabolism and Energy Generation
Energy Carriers and Their Roles in Cellular Reactions
Microbial cells utilize specialized molecules known as energy carriers to transfer and store energy during metabolic reactions. These carriers play a central role in coupling catabolic and anabolic processes.
ATP (Adenosine Triphosphate): The primary energy currency of the cell, used to drive endergonic reactions.
NADH and NADPH: Electron carriers that shuttle electrons during redox reactions, essential for cellular respiration and biosynthesis.
FADH2: Another electron carrier involved in the electron transport chain.
Example: During glycolysis, NAD+ is reduced to NADH as glucose is oxidized.
Enzyme Catalysis
Enzymes are biological catalysts that accelerate chemical reactions by lowering the activation energy required. They are highly specific for their substrates and are not consumed in the reaction.
Enzymes bind substrates at their active site, forming an enzyme-substrate complex.
They facilitate the conversion of substrates to products through mechanisms such as induced fit and transition state stabilization.
Example: Lactate dehydrogenase catalyzes the conversion of pyruvate to lactic acid during fermentation.
Glucose Catabolic Pathways
Glycolysis (Embden-Meyerhof-Parnas Pathway)
Glycolysis is the most common pathway for glucose catabolism, occurring in the cytoplasm and not requiring oxygen. It breaks down one glucose molecule into two pyruvate molecules, generating ATP and NADH.
Location: Cytoplasm
Oxygen Requirement: Anaerobic (does not require oxygen)
Products: 2 pyruvate, 2 ATP, 2 NADH
Equation:
Entner-Doudoroff Pathway
This alternative glycolytic pathway is found mainly in prokaryotes and uses unique enzymes to convert glucose to pyruvate.
Key Enzymes: 6-phosphogluconate dehydratase, 2-keto-3-deoxyphosphogluconate aldolase
Net Yield per Glucose: 1 ATP, 1 NADH, 1 NADPH
Common in: Pseudomonas, Rhizobium, Azotobacter, Agrobacterium, and other Gram-negative genera
Significance: Used by aerobes due to lower ATP yield per glucose
Pentose Phosphate Pathway (PPP)
The PPP is a metabolic pathway parallel to glycolysis, providing reducing power and biosynthetic precursors.
Oxidative Phase: Glucose-6-phosphate is converted to ribulose-5-phosphate, generating 2 NADPH.
Non-oxidative Phase: Synthesis of 5-carbon sugars for nucleotide and amino acid biosynthesis.
Fermentation
Definition and Applications
Fermentation is an anaerobic process where organic molecules regenerate NAD+ from NADH, allowing glycolysis to continue in the absence of oxygen. It is used by many bacteria, archaea, and some eukaryotes.
Applications: Food production (yogurt, cheese, alcohol), industrial biotechnology
Lactic Acid Fermentation
Occurs in animals (muscle cells) and certain bacteria (e.g., yogurt production). Pyruvate is reduced to lactic acid by lactate dehydrogenase.
Equation:
Product: 2 molecules of lactic acid
Alcohol Fermentation
Common in yeast and some bacteria. Pyruvate is converted to ethanol and CO2.
Equation:
Products: 2 ethanol, 2 CO2
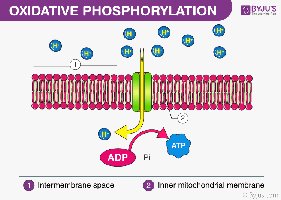
Oxidative Phosphorylation
Overview and Cellular Location
Oxidative phosphorylation is the process of ATP formation as electrons are transferred from NADH and FADH2 to oxygen via the electron transport chain (ETC). This process occurs in the inner mitochondrial membrane (in eukaryotes) or the plasma membrane (in prokaryotes).
Final Step: Cellular respiration
Location: Inner mitochondrial membrane
Mechanism: Electrons move through the ETC, releasing energy to pump protons and generate a proton gradient.
Bioremediation
Definition and Microbial Role
Bioremediation is the use of microorganisms to degrade or detoxify environmental contaminants. Microbes metabolize pollutants, converting them into less harmful substances.
Examples: Oil spill cleanup, degradation of pesticides, heavy metal detoxification
Proton Motive Force (PMF) and Chemiosmotic Theory
PMF: Generation and Function
The proton motive force (PMF) is the electrochemical gradient of protons across a membrane, generated by the electron transport chain. It drives ATP synthesis, transport processes, and flagellar rotation.
Generated by: Electron transport chain pumping protons across the membrane
Function: Powers ATP synthase, active transport, and motility
Chemiosmotic Theory
This theory explains how the energy from electron transfer is used to pump protons, creating PMF, which is then used to synthesize ATP.
Electron flow through the ETC releases energy, pumping protons across the membrane.
Proton gradient stores potential energy, used by ATP synthase to generate ATP.
Diverse Energy Sources in Microbes
Chemotrophy and Phototrophy
Chemotrophy: Energy from chemical compounds via electron transport systems (ETS).
Chemoorganotrophs: Electrons from organic molecules.
Chemolithotrophs: Electrons from inorganic molecules.
Phototrophy: Energy from light; includes photoautotrophy (CO2 fixation) and photoheterotrophy (organic carbon use).
Organotrophy and Respiration
Organotrophs: Use organic molecules as electron donors.
Aerobic Respiration: O2 as terminal electron acceptor.
Anaerobic Respiration: Alternative acceptors (e.g., NO3-).
Electron Transport System (ETS)
Components and Arrangement
The bacterial ETS consists of three main components:
1. Initial Substrate Oxidoreductase (Dehydrogenase): Accepts electrons from donors (e.g., NADH).
2. Mobile Electron Carrier (Quinone Pool): Transfers electrons between complexes.
3. Terminal Oxidase: Transfers electrons to the terminal electron acceptor (e.g., O2).
Example Reaction:
ATP Synthase: Structure and Function
ATP synthase is a membrane-bound enzyme complex that synthesizes ATP using the energy stored in the proton motive force. Protons flow through ATP synthase, driving the phosphorylation of ADP to ATP.
Structure: Composed of F0 (membrane channel) and F1 (catalytic) subunits.
Function: Converts electrochemical energy into chemical energy (ATP).
Other Electron Acceptors and Donors
Microbes can use a variety of electron donors (e.g., H2, Fe2+, organic acids) and acceptors (e.g., nitrate, sulfate, carbon dioxide) in respiration, allowing them to thrive in diverse environments.
Summary Table: Major Glucose Catabolic Pathways
Pathway | Main Location | ATP Yield | NADH/NADPH Yield | Key Products |
|---|---|---|---|---|
Glycolysis (EMP) | Cytoplasm | 2 ATP | 2 NADH | 2 Pyruvate |
Entner-Doudoroff | Cytoplasm (Prokaryotes) | 1 ATP | 1 NADH, 1 NADPH | 2 Pyruvate |
Pentose Phosphate | Cytoplasm | Variable | 2 NADPH | Ribulose-5-phosphate, CO2 |
