 Back
BackMicrobial Metabolism: Classification, Energy Pathways, and Enzyme Regulation
Study Guide - Smart Notes

Microbial Metabolism
Overview of Microbial Metabolism
Microbial metabolism encompasses all the chemical processes that microorganisms use to obtain energy and nutrients for growth and reproduction. These processes are fundamental to microbial life and underpin many ecological and industrial functions.
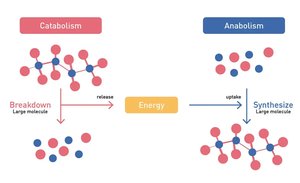
Catabolism: The breakdown of large molecules into smaller ones, releasing energy.
Anabolism: The synthesis of large molecules from smaller ones, requiring energy input.
Classification by Nutritional Mode
Microbes are classified based on their sources of carbon and energy, which allows them to thrive in diverse and extreme environments.
Autotrophs: Use carbon dioxide (CO2) as their carbon source to build cellular material.
Heterotrophs: Obtain carbon from organic compounds such as sugars, fats, or proteins.

Phototrophs: Use sunlight as their energy source.
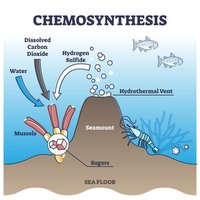
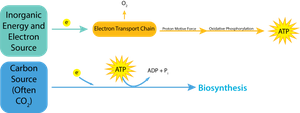
Chemotrophs: Derive energy from the oxidation of chemical compounds.
Chemolithotrophs: Utilize inorganic molecules (e.g., ammonia, hydrogen sulfide, iron) for energy.
Specialized Energy Pathways
Microbes generate ATP through various pathways, depending on the availability of oxygen.
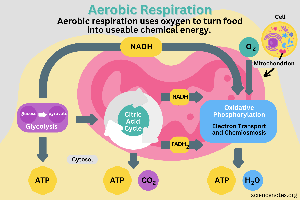
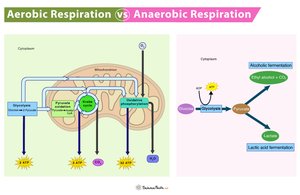
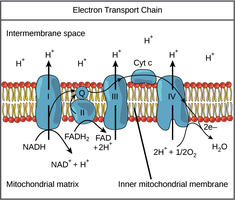
Aerobic Respiration: Uses oxygen as the terminal electron acceptor, producing the most ATP and being highly efficient.
Anaerobic Respiration: Utilizes alternative electron acceptors such as nitrate or sulfate in the absence of oxygen.
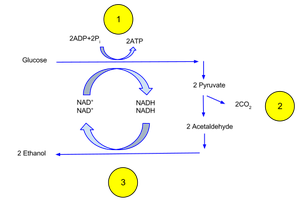
Fermentation: Occurs without oxygen; organic molecules (e.g., pyruvate) accept electrons, resulting in byproducts like alcohol or lactic acid.
Unique Microbial Capabilities
Microbes possess metabolic abilities that are essential for Earth's biogeochemical cycles.
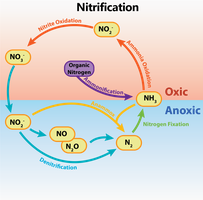
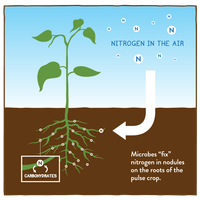
Nitrogen Fixation: Conversion of atmospheric nitrogen (N2) into ammonia, usable by plants.
Methanogenesis: Production of methane gas by certain Archaea.
Extreme Digestion: Breakdown of tough materials like cellulose, plastics, or toxic chemicals, important for bioremediation.
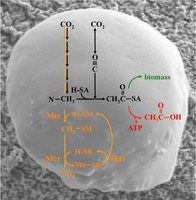

Energy Carriers in Microbial Cells
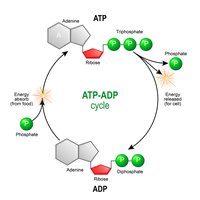
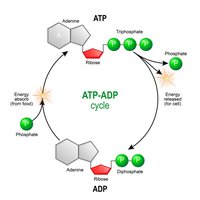
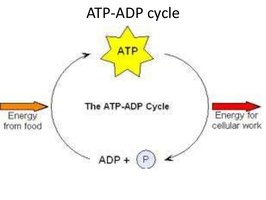
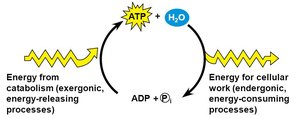
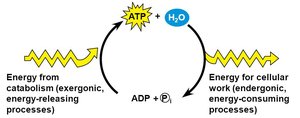
ATP: The Energy Currency
Adenosine Triphosphate (ATP) is the primary energy carrier in cells. Energy is stored in the high-energy phosphate bonds and released when ATP is hydrolyzed to ADP.
Charging ATP: Energy from food breakdown is used to add a phosphate to ADP, forming ATP.
Using ATP: Removal of the third phosphate releases energy for cellular work.
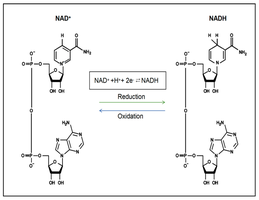
NADH and FADH2: Electron Carriers
NADH and FADH2 carry high-energy electrons (reducing power) from food molecules to the electron transport chain (ETC), where their energy is used to generate ATP.
Charging: NAD+ accepts electrons and a proton to become NADH; FAD becomes FADH2 similarly.
Using the Energy: These carriers deliver electrons to the ETC, creating a proton gradient for ATP synthesis.
Recycling Mechanisms: Coupled Cycles
ATP/ADP Cycle
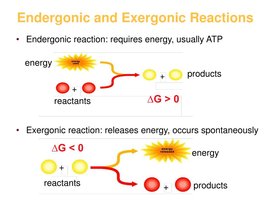
Cells couple energy-releasing (exergonic) reactions with energy-requiring (endergonic) reactions, recycling ATP and ADP continuously.
Exergonic Reactions: Release energy, used to phosphorylate ADP to ATP.
Endergonic Reactions: Require energy, obtained by hydrolyzing ATP to ADP.
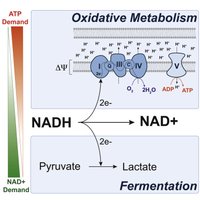
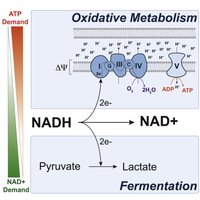
NAD+/NADH Cycle (Redox Balance)
For metabolism to proceed, NAD+ must be regenerated.
In Respiration: NADH donates electrons to the ETC, regenerating NAD+.
In Fermentation: Electrons from NADH are transferred to organic molecules (e.g., pyruvate), regenerating NAD+ without a functional ETC.
Enzymes in Metabolic Reactions
Role of Enzymes
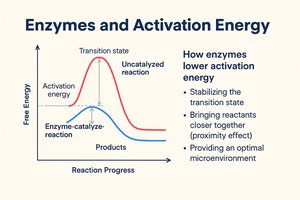
Enzymes are biological catalysts that accelerate chemical reactions by lowering activation energy, enabling life-sustaining reactions to occur at normal cell temperatures.
Activation Energy (Ea): The minimum energy required to start a reaction.
Enzyme Solution: Enzymes bind substrates, position them correctly, and strain bonds to lower Ea.
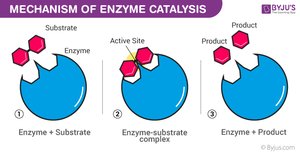
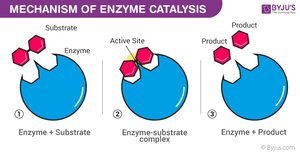
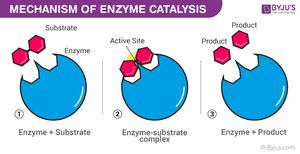
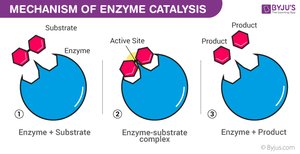
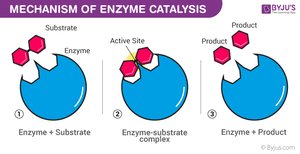
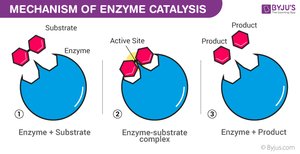
Enzyme Catalytic Cycle
Enzymes are highly specific and reusable, facilitating one particular reaction on one substrate.
Substrate Binding: Substrate fits into the enzyme's active site (lock-and-key or induced fit model).
Enzyme–Substrate Complex: Enzyme holds substrate, strains bonds.
Catalysis: Reaction occurs, substrate is converted to product.
Product Release: Product leaves the active site.
Recycling: Enzyme is unchanged and ready for another substrate.
Coupling Reactions
Enzymes often couple energy-requiring reactions with energy-releasing ones, such as ATP hydrolysis, to drive biosynthetic processes efficiently.
ATP Breakdown: Releases energy, which is captured and used by enzymes to drive difficult reactions.
Regulation of Metabolic Pathways
Microbes regulate enzyme activity to optimize energy use and respond to environmental changes.
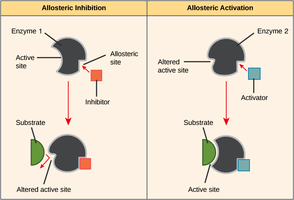
Allosteric Regulation: Molecules bind to sites other than the active site, altering enzyme activity (activation or inhibition).
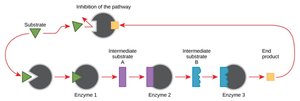
Feedback Inhibition: The end product of a pathway inhibits the first enzyme, preventing overproduction.
Environmental Sensitivity: Enzymes are adapted to specific temperatures and pH, depending on the microbe's habitat.
Summary Table: Microbial Metabolism Classification
Classification | Carbon Source | Energy Source | Example |
|---|---|---|---|
Autotroph | CO2 | Light (Phototroph) or Inorganic Chemicals (Chemolithotroph) | Cyanobacteria, Nitrifying bacteria |
Heterotroph | Organic compounds | Chemicals (Chemoheterotroph) | Most bacteria, fungi |
Phototroph | Varies | Light | Algae, photosynthetic bacteria |
Chemotroph | Varies | Chemicals | Sulfur bacteria, iron bacteria |
Key Equations
ATP Formation:
ATP Hydrolysis:
NAD+ Reduction:
Example Application
Bioremediation: Microbes are used to clean up environmental pollutants due to their ability to metabolize toxic compounds.
Industrial Fermentation: Microbial fermentation is harnessed to produce alcohol, antibiotics, and other valuable products.
Additional info: Expanded explanations and context were added to ensure completeness and academic quality.
