 Back
BackMicrobial Metabolism: Electron Flow and Energy Production
Study Guide - Smart Notes

Metabolism: Overview and Key Concepts
Definition and Types of Metabolism
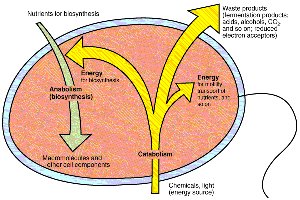
Metabolism is the sum of all chemical reactions occurring within a cell, divided into two main categories: catabolism and anabolism. Catabolism involves the breakdown of molecules to release energy, while anabolism uses energy to synthesize cellular components.
Catabolism: Energy-producing reactions; breakdown of organic or inorganic compounds or light energy to generate ATP and reducing power.
Anabolism: Biosynthetic, energy-consuming reactions; use of ATP and reducing power to build macromolecules and cell structures.
Redox Reactions in Microbial Metabolism
Oxidation-Reduction (Redox) Reactions
Redox reactions are fundamental to energy production in cells. Oxidation is the loss of electrons, while reduction is the gain of electrons. These reactions are always coupled, with electrons transferred from a donor (which is oxidized) to an acceptor (which is reduced).
Electron donor: Substance that loses electrons (is oxidized).
Electron acceptor: Substance that gains electrons (is reduced).
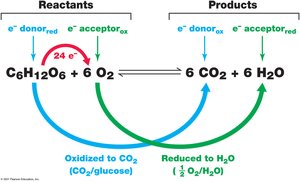
Example: In aerobic respiration, glucose is oxidized to CO2 and O2 is reduced to H2O.
Electron Carriers and NAD+/NADH
Role of Electron Carriers
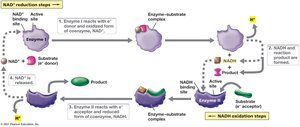
Electron carriers facilitate the transfer of electrons from donors to acceptors. They can be membrane-bound (e.g., cytochromes) or freely diffusible coenzymes (e.g., NAD+/NADH).
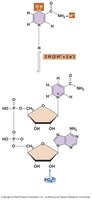
NAD+ (Nicotinamide adenine dinucleotide): Accepts electrons and protons to become NADH.
NADH: Donates electrons in metabolic pathways, especially in respiration.
NAD+/NADH Cycling
NAD+ and NADH cycle between oxidized and reduced forms, shuttling electrons during metabolic reactions. This cycling is essential for maintaining redox balance in the cell.
Energy Storage and ATP Generation
Short-Term and Long-Term Energy Storage
Cells store energy in various forms for immediate or future use:
Short-term: ATP (adenosine triphosphate) and derivatives of coenzyme A (thioester bonds).
Long-term: Glycogen, poly-β-hydroxybutyrate, and elemental sulfur.
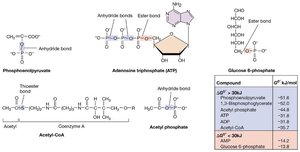
ATP Synthesis Mechanisms
ATP is the primary energy currency of the cell, generated by three main mechanisms:
Substrate-level phosphorylation: Direct transfer of a phosphate group from a high-energy substrate to ADP.
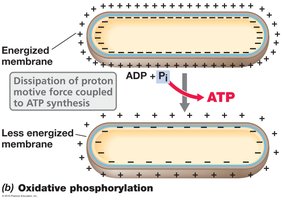
Oxidative phosphorylation: ATP synthesis driven by the dissipation of the proton motive force across a membrane, catalyzed by ATP synthase.
Photophosphorylation: Light-driven ATP synthesis in phototrophs, similar to oxidative phosphorylation but powered by light energy.
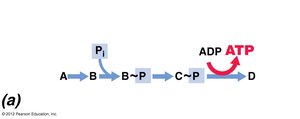
Glycolysis and Fermentation
Glycolysis: Pathway and Key Steps
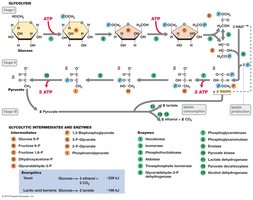
Glycolysis is the central pathway for glucose catabolism, converting glucose to pyruvate and generating ATP and NADH. It occurs in the cytoplasm and is used by many microorganisms.
Net yield per glucose: 2 ATP, 2 NADH, 2 pyruvate.
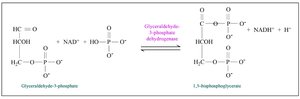
Key steps: Substrate-level phosphorylation (steps 7 and 10), NAD+ reduction (step 6).
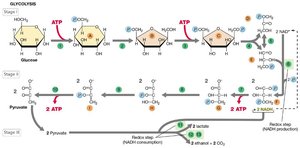
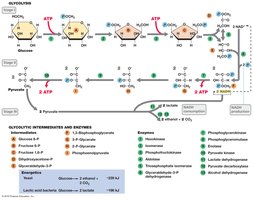
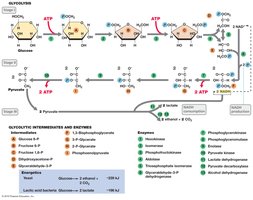
Fermentation
Fermentation is an anaerobic process where organic compounds serve as both electron donors and acceptors. It regenerates NAD+ and produces ATP via substrate-level phosphorylation, yielding various end products such as acids, alcohols, and gases.
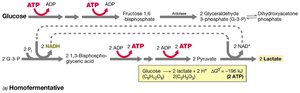
Homolactic fermentation: Produces lactic acid (e.g., Streptococcus).
Alcohol fermentation: Produces ethanol and CO2 (e.g., yeast).
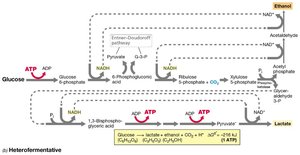
Heterolactic fermentation: Produces lactic acid, ethanol, and CO2 (e.g., Leuconostoc).
Common Fermentation Pathways
Microorganisms utilize diverse fermentation pathways, each with characteristic substrates, products, and energy yields.
Type | Reaction | Energy Yield (kJ/mol) | Organisms |
|---|---|---|---|
Alcoholic | Hexose → 2 ethanol + 2 CO2 | -239 | Yeast, Zymomonas |
Homolactic | Hexose → 2 lactate | -196 | Streptococcus, Lactococcus |
Heterolactic | Hexose → lactate + ethanol + CO2 + H2 | -119 | Leuconostoc, some Lactobacillus |
Mixed acid | Hexose → lactate + acetate + succinate + formate + ethanol + CO2 + H2 | -202 | Escherichia, Salmonella, Shigella |
Propionic acid | Hexose → propionate + acetate + CO2 + H2O | -254 | Propionibacterium |
Butyric acid | Hexose → butyrate + acetate + CO2 + H2 | -293 | Clostridium butyricum |
Butanol | Hexose → butanol + acetone + CO2 + H2 | -254 | Clostridium acetobutylicum |

Respiration: Aerobic and Anaerobic
Overview of Respiration
Respiration is a process where electrons from an electron donor are transferred through an electron transport chain to a terminal electron acceptor, generating a proton motive force used to synthesize ATP via oxidative phosphorylation. Aerobic respiration uses O2 as the terminal acceptor, while anaerobic respiration uses other acceptors such as nitrate or sulfate.
Aerobic respiration: Complete oxidation of glucose to CO2; yields up to 38 ATP per glucose.
Anaerobic respiration: Uses alternative electron acceptors; yields less ATP than aerobic respiration.

Electron Transport Chain and ATP Synthase
Electron Carriers in the Electron Transport Chain
The electron transport chain (ETC) consists of a series of protein and non-protein electron carriers, including NADH dehydrogenases, flavoproteins, iron-sulfur proteins, cytochromes, and quinones. Electrons flow through these carriers, releasing energy used to pump protons across the membrane, creating a proton motive force.
NADH dehydrogenase: Accepts electrons from NADH.
Flavoproteins: Contain FMN or FAD as prosthetic groups.
Iron-sulfur proteins: Transfer electrons via iron atoms.
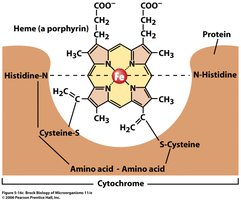
Cytochromes: Contain heme groups with iron.
Quinones: Lipid-soluble electron carriers.
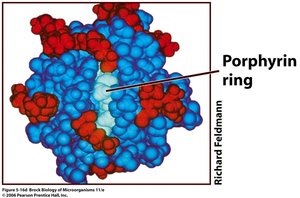
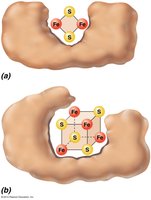
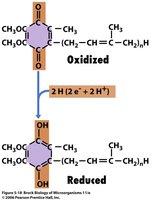
ATP Synthase and Chemiosmosis
ATP synthase (also called ATPase) is a membrane-bound enzyme complex that synthesizes ATP from ADP and inorganic phosphate, powered by the flow of protons down their electrochemical gradient (proton motive force). This process is known as chemiosmosis.
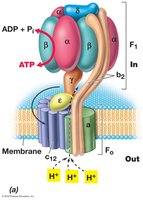
Summary Table: Fermentation vs. Respiration
Process | ATP Yield per Glucose | Electron Acceptor | Key Features |
|---|---|---|---|
Fermentation | 2 | Organic compound (endogenous) | Substrate-level phosphorylation only; incomplete oxidation |
Respiration (Aerobic) | ~38 | O2 | Substrate-level and oxidative phosphorylation; complete oxidation |
Respiration (Anaerobic) | Varies (<38) | Nitrate, sulfate, etc. | Substrate-level and oxidative phosphorylation; incomplete oxidation |
Additional info: These notes cover core concepts from Chapter 3 (Microbial Metabolism) and related sections on electron flow, energy production, and fermentation, as outlined in standard microbiology curricula.
