 Back
BackMicrobial Metabolism: Energy, Electron, and Carbon Sources; Respiration and Fermentation
Study Guide - Smart Notes

Microbial Metabolism: Energy, Electron, and Carbon Sources
Overview of Microbial Nutritional Types
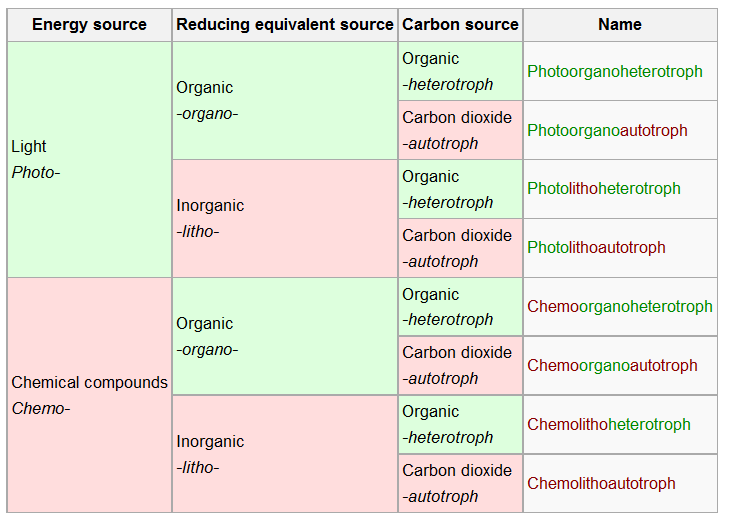
Microorganisms are classified based on their sources of energy, electrons, and carbon. These classifications help in understanding their metabolic diversity and ecological roles.
Energy Source: Light (phototrophs) or chemical compounds (chemotrophs)
Electron Source: Organic (organotrophs) or inorganic (lithotrophs) molecules
Carbon Source: Organic compounds (heterotrophs) or carbon dioxide (autotrophs)
Catabolism and Anabolism
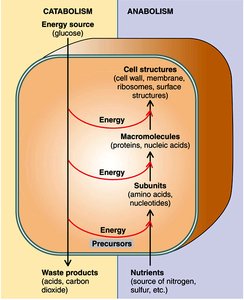
Microbial metabolism consists of two main processes: catabolism (breakdown of molecules to release energy) and anabolism (biosynthesis of cellular components using energy).
Catabolism: Degradation of energy sources (e.g., glucose) to produce ATP, reducing power, and precursor metabolites.
Anabolism: Synthesis of macromolecules (proteins, nucleic acids, etc.) from subunits using energy and precursors from catabolism.
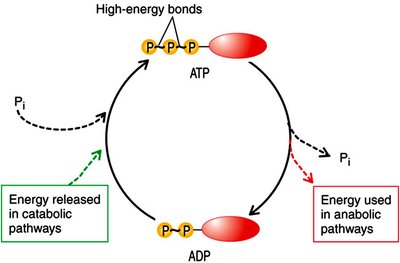
ATP: The Energy Currency of the Cell
ATP Generation and Utilization
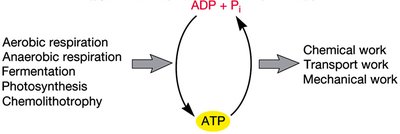
ATP (adenosine triphosphate) is the universal energy currency in cells. It is generated by various catabolic processes and used to drive cellular work, including biosynthesis, transport, and movement.
ATP Synthesis: Produced by substrate-level phosphorylation, oxidative phosphorylation, and photophosphorylation.
ATP Utilization: Powers chemical, transport, and mechanical work in the cell.
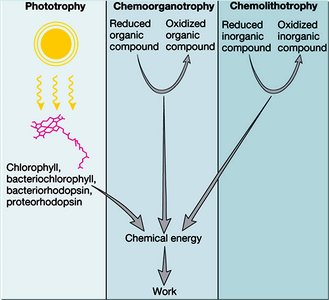
Major Types of Microbial Metabolism
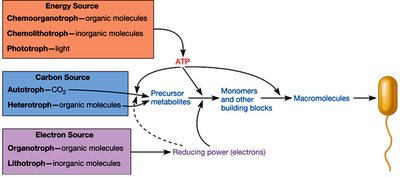
Phototrophy, Chemoorganotrophy, and Chemolithotrophy
Microbes obtain energy through different metabolic strategies:
Phototrophy: Light energy is converted to chemical energy (e.g., photosynthesis).
Chemoorganotrophy: Organic compounds are oxidized for energy and electrons.
Chemolithotrophy: Inorganic compounds are oxidized for energy and electrons.
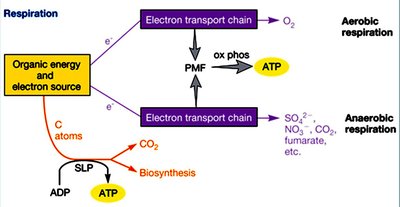
Respiration and Fermentation
Respiration
Respiration involves the oxidation of organic or inorganic molecules, with electrons transferred through an electron transport chain (ETC) to an external electron acceptor, generating a proton motive force (PMF) used for ATP synthesis (oxidative phosphorylation).
Aerobic Respiration: Oxygen is the terminal electron acceptor.
Anaerobic Respiration: Other molecules (e.g., nitrate, sulfate, CO2) serve as terminal electron acceptors.
Substrate-Level Phosphorylation (SLP): ATP is also generated directly in some steps of catabolism.
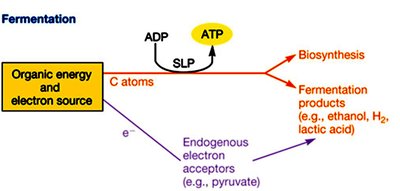
Fermentation
Fermentation is an anaerobic process where organic molecules serve as both electron donors and acceptors. ATP is generated only by substrate-level phosphorylation, and the electron transport chain is not involved.
Endogenous Electron Acceptors: Typically pyruvate or its derivatives.
Products: Organic acids, alcohols, gases (e.g., ethanol, lactic acid, H2).
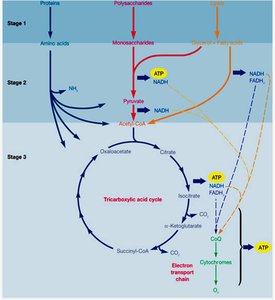
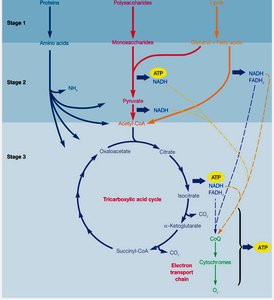
Stages of Catabolism: Glycolysis and the TCA Cycle
Glycolysis
Glycolysis is the central pathway for the breakdown of glucose to pyruvate, generating ATP and NADH. There are three main glycolytic pathways in microbes:
Embden-Meyerhof Pathway: The most common glycolytic pathway, yielding 2 ATP and 2 NADH per glucose.
Pentose Phosphate Pathway: Generates NADPH and pentoses for biosynthesis.
Entner-Doudoroff Pathway: An alternative pathway found in some bacteria.
Key Reaction (Embden-Meyerhof):
Tricarboxylic Acid (TCA) Cycle
The TCA cycle (also called the citric acid or Krebs cycle) completes the oxidation of organic fuel. Pyruvate is converted to acetyl-CoA, which enters the cycle. Each turn of the cycle produces CO2, NADH, FADH2, and GTP (or ATP).
Location: Cytoplasm in prokaryotes, mitochondria in eukaryotes.
Products per Acetyl-CoA: 2 CO2, 3 NADH, 1 FADH2, 1 GTP/ATP.
ATP Yield in Respiration and Fermentation
ATP-Generating Processes in Prokaryotic Chemoorganoheterotrophs
The total ATP yield depends on the metabolic pathway used:
Metabolic Process | Uses an Electron Transport Chain | Terminal Electron Acceptor | ATP by Substrate-Level Phosphorylation | ATP by Oxidative Phosphorylation | Total ATP (Theoretical Maximum) |
|---|---|---|---|---|---|
Aerobic respiration | Yes | O2 | 4 (2 in glycolysis, 2 in TCA cycle) | 34 | 38 |
Anaerobic respiration | Yes | Molecule other than O2 (e.g., NO3-, SO42-) | Varies | Varies (less than aerobic) | <38 |
Fermentation | No | Organic molecule (e.g., pyruvate) | 2 (in glycolysis) | 0 | 2 |

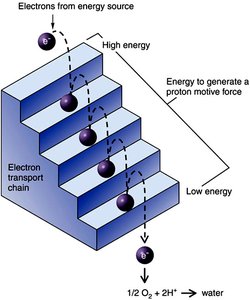
Electron Transport Chain and Oxidative Phosphorylation
Principle of the Electron Transport Chain (ETC)
The ETC is a series of membrane-bound carriers that transfer electrons from NADH and FADH2 to a terminal electron acceptor. The energy released is used to pump protons across the membrane, generating a proton motive force (PMF) that drives ATP synthesis via ATP synthase.
Localization: Plasma membrane (prokaryotes), mitochondrial membrane (eukaryotes)
Electron Flow: From carriers with more negative standard reduction potential (E0') to more positive E0'
Summary Table: Microbial Metabolic Diversity
The diversity of microbial metabolism allows adaptation to a wide range of environments and ecological niches. Understanding these processes is fundamental to microbiology, biotechnology, and environmental science.
