 Back
BackMicrobial Metabolism: Foundations and Pathways
Study Guide - Smart Notes

Microbial Metabolism
Introduction to Metabolism
Metabolism encompasses all chemical reactions occurring within a microorganism, enabling it to grow, reproduce, and respond to its environment. These reactions are organized into metabolic pathways, which are determined by enzymes encoded by genes.
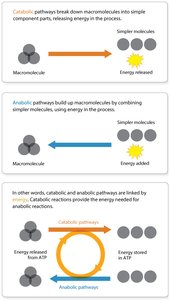
Catabolism: Breaks down complex molecules into simpler ones, releasing energy.
Anabolism: Uses energy to build complex molecules from simpler ones.
Metabolic Pathways: Can be linear, branched, or circular, and are regulated by specific enzymes.
Energy in Chemical Reactions
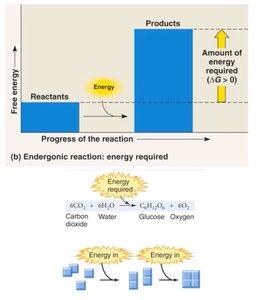
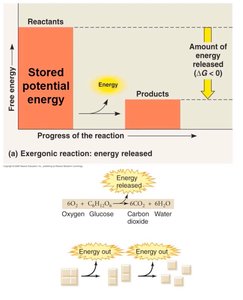
Chemical reactions in cells involve energy changes. Two main types are endergonic (energy-absorbing) and exergonic (energy-releasing) reactions.
Endergonic Reactions: Require energy input; products have more energy than reactants. Example: Photosynthesis.
Exergonic Reactions: Release energy; products have less energy than reactants. Example: Cellular respiration.
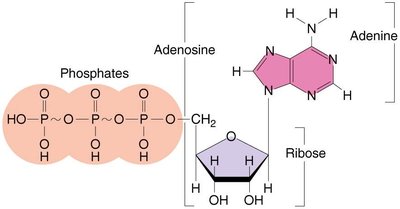
Adenosine Triphosphate (ATP)
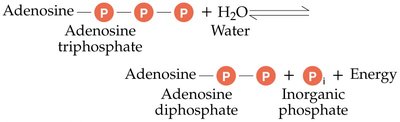
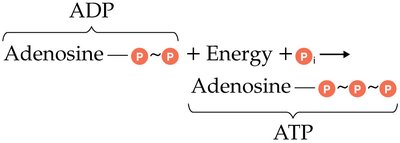
ATP is the primary energy currency in cells. Its structure consists of adenosine (adenine + ribose) and three phosphate groups. Energy is stored in the bonds between phosphate groups and released upon hydrolysis.
ATP Hydrolysis: ATP + H2O → ADP + Pi + Energy
ATP Synthesis: ADP + Pi + Energy → ATP
Role of ATP in Metabolism
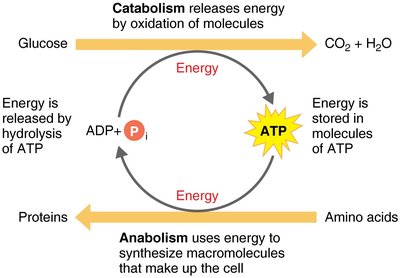
ATP links catabolic and anabolic reactions. Catabolism releases energy, which is stored in ATP and then used to drive anabolic processes.
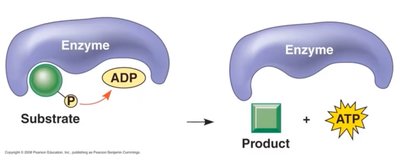
Catabolic Reactions: Generate ATP by breaking down molecules.
Anabolic Reactions: Consume ATP to build cellular components.
Enzymes and Their Function
Enzyme Structure and Function
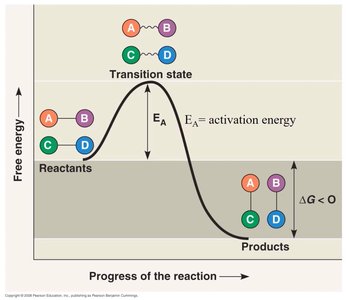
Enzymes are biological catalysts, typically globular proteins, that speed up chemical reactions by lowering activation energy. Each enzyme is specific to a substrate and reaction.
Active Site: Region where substrate binds and reaction occurs.
Enzyme Specificity: Each enzyme catalyzes only one type of reaction.
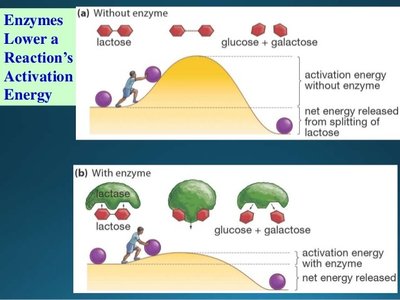
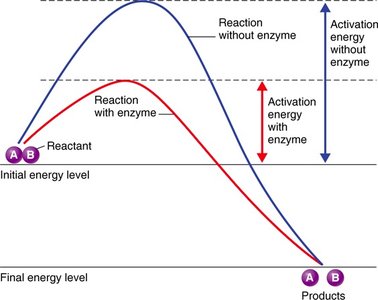
Mechanism of Enzymatic Action
Enzymes function by forming an enzyme-substrate complex, transforming the substrate into products, and releasing the products while remaining unchanged.
Turnover Number: Number of substrate molecules converted per second (typically 1–10,000).
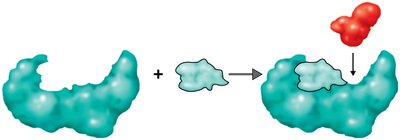
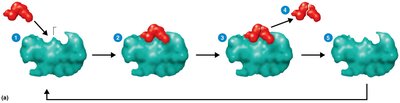
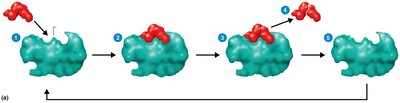
Enzyme Components
Enzymes may require non-protein helpers called cofactors or coenzymes to function.
Apoenzyme: Protein portion, inactive alone.
Cofactor: Non-protein component (inorganic or organic).
Holoenzyme: Apoenzyme plus cofactor, fully active.
Important Coenzymes: NAD+, NADP+, FAD, Coenzyme A.
Factors Influencing Enzyme Activity
Enzyme activity is affected by temperature, pH, substrate concentration, and inhibitors.
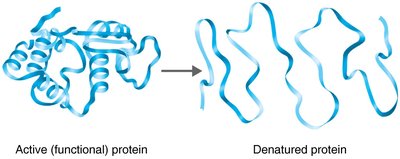
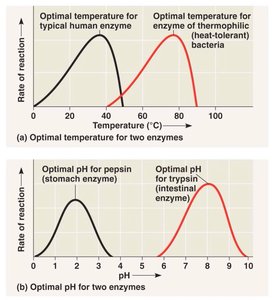
Temperature: Each enzyme has an optimal temperature; extreme heat can denature enzymes.
pH: Each enzyme has an optimal pH; deviations can reduce activity.
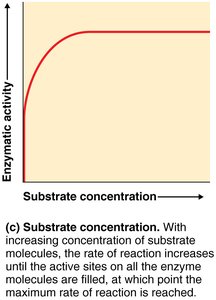
Substrate Concentration: Activity increases with substrate concentration until saturation.
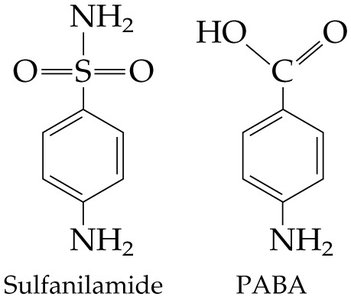
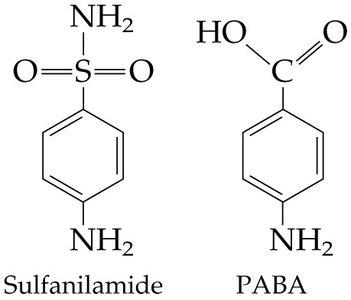
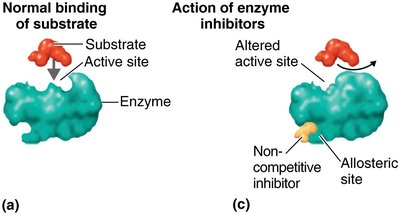
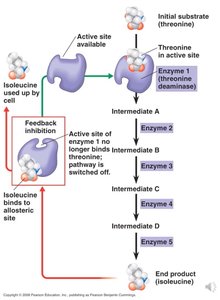
Inhibitors: Can be competitive (bind active site) or noncompetitive (bind allosteric site).
Types of Enzymes
Endoenzymes: Act inside the cell; essential for metabolism.
Exoenzymes: Secreted outside the cell; act on large substrates or neutralize harmful chemicals.
Constitutive Enzymes: Always present and produced.
Induced (Adaptive) Enzymes: Produced only when substrate is present.
Ribozymes: RNA molecules with catalytic activity, found in eukaryotic cells.
Oxidation-Reduction (Redox) Reactions
Redox Reactions in Metabolism
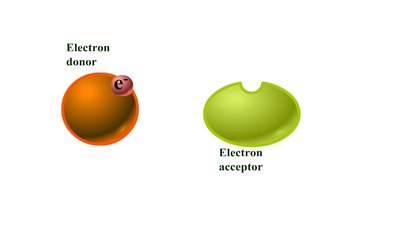
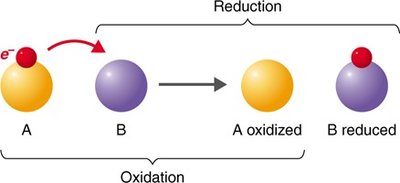
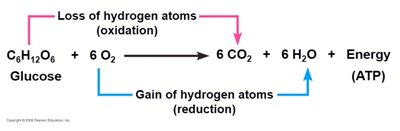
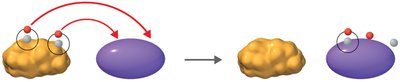
Redox reactions involve the transfer of electrons between molecules. Oxidation is the loss of electrons, while reduction is the gain of electrons. These reactions are essential for energy production in cells.
Oxidation: Loss of electrons (often as hydrogen atoms).
Reduction: Gain of electrons (often as hydrogen atoms).
Coenzymes: NAD+, FAD, and NADP+ act as electron carriers.
ATP Generation Mechanisms
Phosphorylation Methods
Cells generate ATP from ADP by three main mechanisms:
Substrate-Level Phosphorylation: Direct transfer of phosphate to ADP from a phosphorylated intermediate.
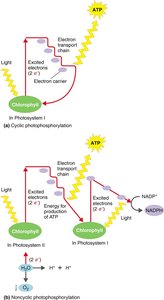
Oxidative Phosphorylation: ATP generated via electron transport chain and chemiosmosis.
Photophosphorylation: ATP generated using light energy in photosynthetic cells.
Modes of Metabolism
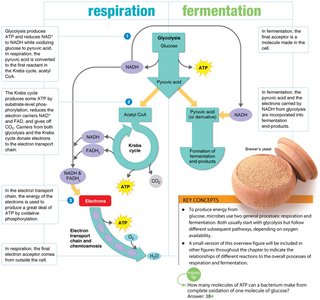
Respiration and Fermentation
Microorganisms obtain energy through cellular respiration or fermentation, depending on the availability of oxygen and the metabolic pathway used.
Cellular Respiration: Complete breakdown of glucose; can be aerobic (O2 as final electron acceptor) or anaerobic (other inorganic molecules as acceptors).
Fermentation: Incomplete breakdown of glucose; anaerobic; organic molecules as final electron acceptors; yields less ATP.
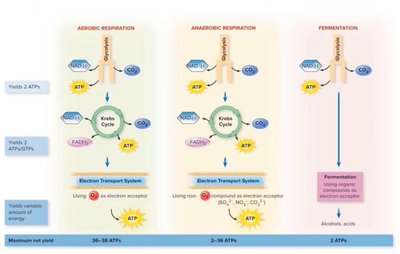
Summary Table: Respiration vs. Fermentation
Pathway | ATP Yield | Oxygen Required | Final Electron Acceptor | End Products |
|---|---|---|---|---|
Aerobic Respiration | 30-32 ATP | Yes | O2 | CO2, H2O |
Anaerobic Respiration | 3-29 ATP | No | NO3-, SO42-, CO32- | NO2-, N2, H2S, CH4 |
Fermentation | 2 ATP | No | Organic molecule | Alcohol, acids |
Key Pathways in Cellular Respiration
Glycolysis
Glycolysis is the first step in glucose metabolism, occurring in the cytosol. It converts glucose to pyruvate, producing ATP and NADH.
Products per glucose: 2 pyruvate, 2 ATP (net), 2 NADH
Citric Acid Cycle (Krebs Cycle)
Occurs in the mitochondrial matrix (eukaryotes) or cytosol (prokaryotes). Acetyl CoA is oxidized, generating CO2, NADH, FADH2, and ATP.
Products per acetyl CoA: 2 CO2, 3 NADH, 1 FADH2, 1 ATP
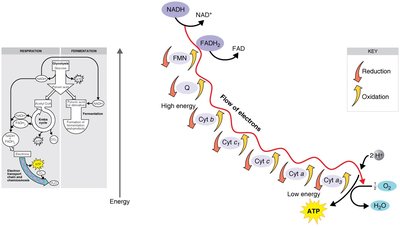
Electron Transport Chain and Chemiosmosis
Electrons from NADH and FADH2 are transferred through a series of carriers, creating a proton gradient that drives ATP synthesis via ATP synthase.
Final electron acceptor: O2 (aerobic) or other inorganic molecules (anaerobic)
ATP yield: Most ATP is generated in this step
Fermentation Pathways
Fermentation allows energy production in the absence of oxygen. Pyruvate is converted to organic end products such as alcohol or lactic acid.
Alcoholic Fermentation: Occurs in yeast; produces ethanol and CO2.
Lactic Acid Fermentation: Occurs in animal cells and some bacteria; produces lactic acid.
Summary
Microbial metabolism is essential for energy production, growth, and survival. Understanding the roles of enzymes, ATP, and metabolic pathways provides a foundation for studying microbial physiology and biotechnology.
