 Back
BackMicrobial Metabolism: Foundations, Pathways, and Regulation
Study Guide - Smart Notes

Basic Chemical Reactions Underlying Metabolism
Overview of Metabolism
Metabolism encompasses all controlled biochemical reactions occurring within a microbe, with the ultimate function of enabling cellular reproduction. These reactions are organized into metabolic pathways that are tightly regulated to ensure cellular efficiency and survival.
Metabolism: The sum of all chemical reactions in a cell.
Key functions include nutrient acquisition, energy extraction, macromolecule synthesis, growth, and reproduction.
Energy is primarily stored and transferred via adenosine triphosphate (ATP).
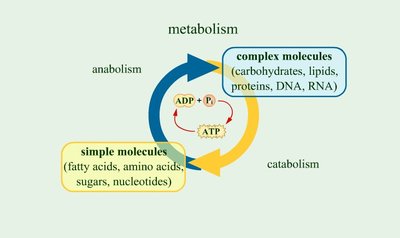
Catabolism and Anabolism
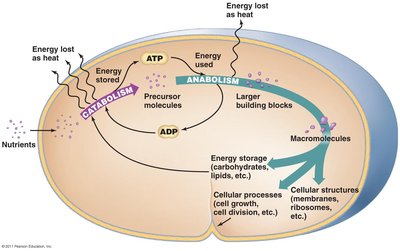
Metabolic reactions are divided into two major classes: catabolic and anabolic pathways. These processes are interconnected and essential for cellular function.
Catabolism: The breakdown of larger molecules into smaller ones, releasing energy (exergonic).
Anabolism: The synthesis of complex molecules from simpler ones, requiring energy input (endergonic).
ATP generated by catabolism is used to drive anabolic reactions.
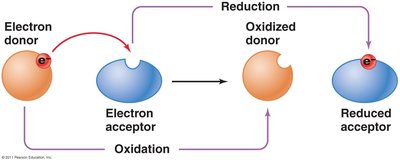
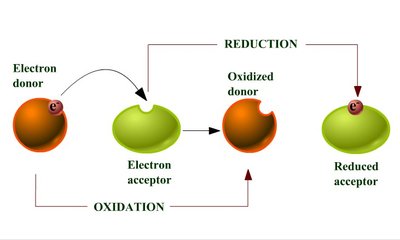
Oxidation and Reduction Reactions
Redox Reactions in Metabolism
Oxidation-reduction (redox) reactions are fundamental to energy transfer in cells. These reactions involve the transfer of electrons from an electron donor to an electron acceptor, and always occur in pairs.
Oxidation: Loss of electrons by a molecule, atom, or ion.
Reduction: Gain of electrons by a molecule, atom, or ion.
Cells use electron carriers such as NAD+, NADP+, and FAD to shuttle electrons during metabolic reactions.
ATP Production and Energy Storage
Mechanisms of ATP Generation
Cells produce ATP by adding a phosphate group to ADP, a process known as phosphorylation. There are three main mechanisms:
Substrate-level phosphorylation: Direct transfer of phosphate between two substrates.
Oxidative phosphorylation: Energy from redox reactions of an electron transport chain is used to add phosphate to ADP.
Photophosphorylation: Light energy is used to phosphorylate ADP in photosynthetic organisms.
The Roles of Enzymes in Metabolism
Enzyme Structure and Function
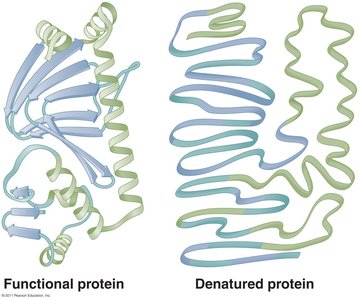
Enzymes are biological catalysts that increase the rate of chemical reactions without being consumed. They are highly specific for their substrates and are essential for metabolic efficiency.
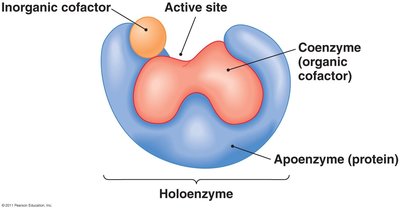
Apoenzyme: The protein portion of an enzyme, inactive without its cofactor.
Cofactor: Non-protein component (inorganic ion or organic coenzyme) required for enzyme activity.
Holoenzyme: The complete, active enzyme with its cofactor.
Some enzymes are RNA molecules called ribozymes.
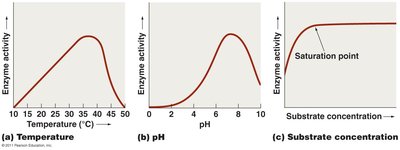
Enzyme Activity and Regulation
Enzyme activity is influenced by several factors, including temperature, pH, substrate concentration, and the presence of inhibitors.
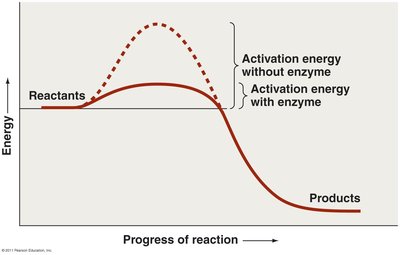
Enzymes lower the activation energy required for reactions.
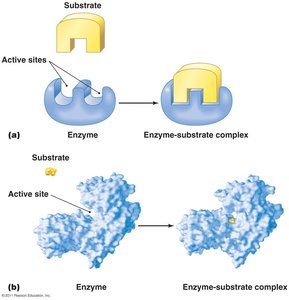
Enzyme-substrate specificity is often described by the "lock-and-key" or "induced fit" models.
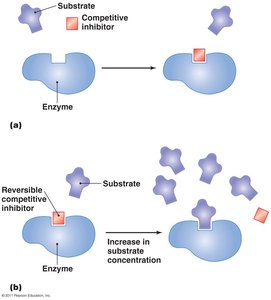
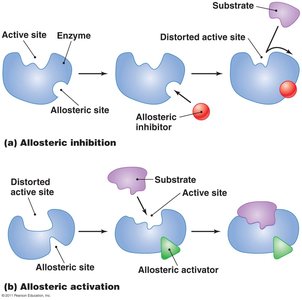
Inhibitors can be competitive (bind to the active site) or noncompetitive (bind to an allosteric site).
Carbohydrate Catabolism
Glycolysis
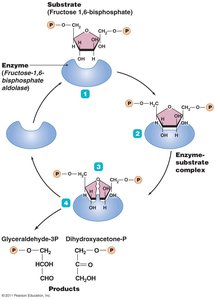
Glycolysis is the central pathway for glucose catabolism, occurring in the cytoplasm of most cells. It splits one six-carbon glucose into two three-carbon pyruvic acid molecules, generating ATP and NADH.
Net gain: 2 ATP, 2 NADH, and 2 pyruvic acid per glucose.
Divided into three stages: energy-investment, lysis, and energy-conserving.
Cellular Respiration
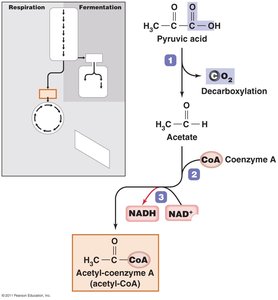
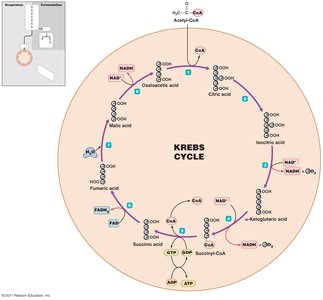
Cellular respiration completely oxidizes pyruvic acid to CO2 and H2O, producing large amounts of ATP through three stages: synthesis of acetyl-CoA, Krebs cycle, and electron transport chain (ETC).
Synthesis of acetyl-CoA: Pyruvic acid is converted to acetyl-CoA, producing NADH and CO2.
Krebs cycle: Acetyl-CoA is oxidized, generating ATP, NADH, FADH2, and CO2.
Electron transport chain: Electrons from NADH and FADH2 are transferred through a series of carriers, creating a proton gradient used to synthesize ATP (chemiosmosis).
Fermentation
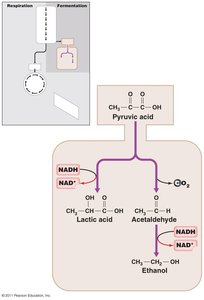
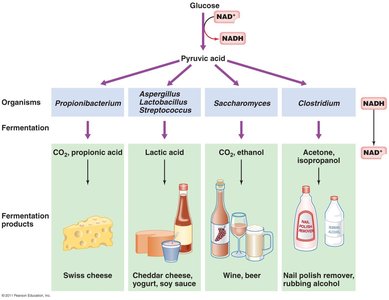
Fermentation is an alternative pathway for energy production when oxygen or other external electron acceptors are unavailable. It regenerates NAD+ for glycolysis by transferring electrons to organic molecules, producing various end products.
Common products: lactic acid, ethanol, propionic acid, acetone, etc.
Fermentation yields less ATP than respiration.
Other Catabolic Pathways
Cells can also catabolize lipids and proteins for energy when carbohydrates are scarce.
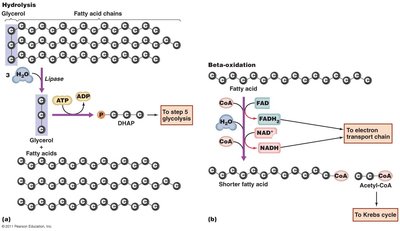
Lipid catabolism: Fats are hydrolyzed to glycerol and fatty acids; fatty acids undergo beta-oxidation to form acetyl-CoA.
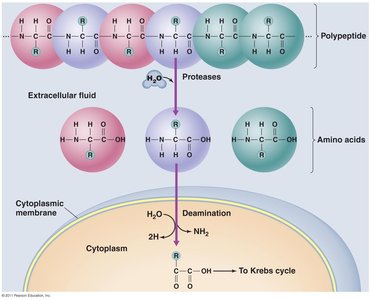
Protein catabolism: Proteins are broken down to amino acids, which are deaminated and converted to intermediates of the Krebs cycle.
Photosynthesis
Overview and Structures
Photosynthesis is the process by which organisms convert light energy into chemical energy, synthesizing carbohydrates from CO2 and H2O. This process is essential for autotrophic growth.
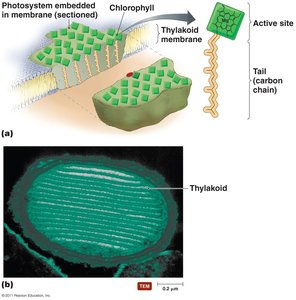
Chlorophylls: Pigments that capture light energy, located in thylakoid membranes.
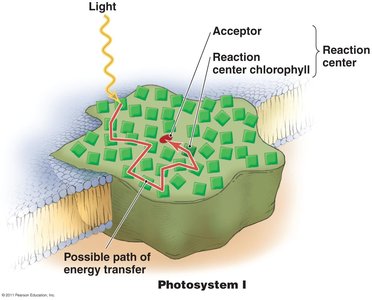
Photosystems: Complexes of chlorophyll and proteins that initiate light-dependent reactions.
In prokaryotes, thylakoids are invaginations of the cytoplasmic membrane; in eukaryotes, they are within chloroplasts.
Light-Dependent and Light-Independent Reactions
Photosynthesis consists of two main stages:
Light-dependent reactions: Use light energy to generate ATP and NADPH via photophosphorylation.
Light-independent reactions: Use ATP and NADPH to fix carbon dioxide into organic molecules (Calvin-Benson cycle).
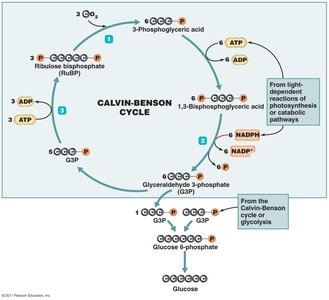
Other Anabolic Pathways
Amphibolic Pathways and Biosynthesis
Anabolic reactions synthesize complex molecules from simpler ones, often using intermediates from catabolic pathways. Many pathways are amphibolic, meaning they can function in both directions depending on cellular needs.
Gluconeogenesis: Synthesis of glucose from non-carbohydrate precursors.
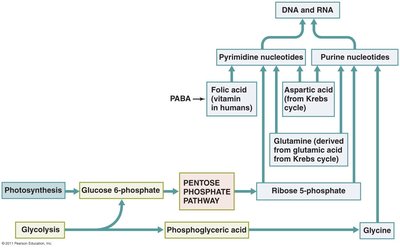
Biosynthesis of fats, amino acids, and nucleotides utilizes intermediates from glycolysis, Krebs cycle, and the pentose phosphate pathway.
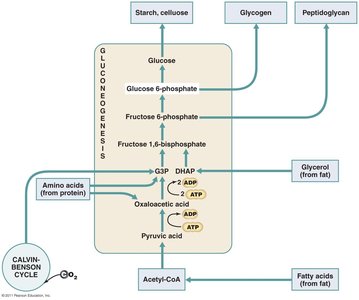
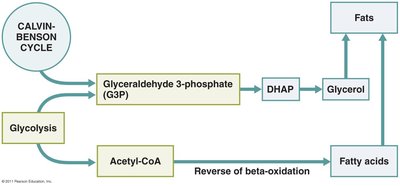
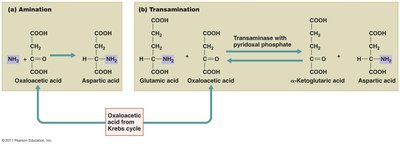
Integration and Regulation of Metabolic Function
Metabolic Regulation
Cells regulate metabolism at multiple levels to optimize resource use and respond to environmental changes.
Synthesis or degradation of transport proteins and enzymes as needed.
Preference for the most energy-efficient substrates.
Feedback inhibition and allosteric regulation of enzymes.
Compartmentalization of metabolic pathways in eukaryotes.
Regulation at the genetic level (gene expression) and at the protein level (enzyme activity).
