 Back
BackMicrobial Metabolism: Foundations, Pathways, and Regulation
Study Guide - Smart Notes

Microbial Metabolism
Introduction to Metabolism
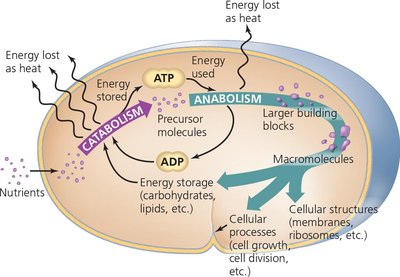
Metabolism encompasses all controlled biochemical reactions occurring within a microbe, with the ultimate function of enabling reproduction. These reactions are organized into metabolic pathways that are tightly regulated to ensure cellular efficiency and survival.
Metabolism: The sum of all chemical reactions in a cell.
Eight guiding principles include nutrient acquisition, energy extraction and storage (mainly as ATP), catabolism to form precursors, anabolism to build macromolecules, and regulated growth and reproduction.
Catabolism and Anabolism
Metabolic reactions are divided into two major classes: catabolic and anabolic pathways.
Catabolic pathways: Break down larger molecules into smaller products, releasing energy (exergonic).
Anabolic pathways: Synthesize large molecules from smaller products, requiring energy input (endergonic).
Oxidation-Reduction (Redox) Reactions
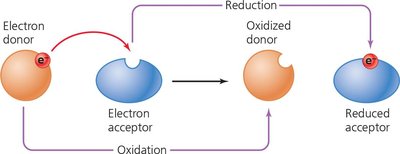
Redox reactions involve the transfer of electrons from an electron donor to an electron acceptor, always occurring simultaneously. Cells use electron carriers such as NAD+, NADP+, and FAD to shuttle electrons during metabolism.
Oxidation: Loss of electrons.
Reduction: Gain of electrons.
Electron carriers are essential for energy transfer in metabolic pathways.
ATP Production and Energy Storage
Cells release energy from nutrients and store it in high-energy phosphate bonds of ATP. ATP is generated by phosphorylation of ADP in three ways:
Substrate-level phosphorylation
Oxidative phosphorylation
Photophosphorylation
Anabolic pathways utilize ATP by breaking its phosphate bonds to drive biosynthetic reactions.
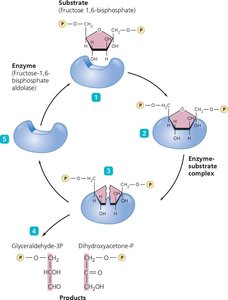
The Roles of Enzymes in Metabolism
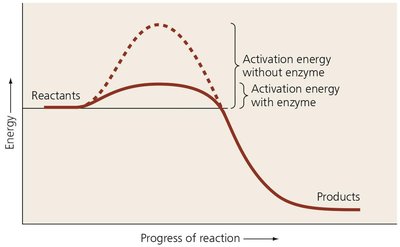
Enzymes are organic catalysts that increase the likelihood of chemical reactions by lowering activation energy. They are classified by the type of reaction they catalyze:
Hydrolases: Catalyze hydrolysis (catabolic)
Isomerases: Rearrangement of atoms (neither catabolic nor anabolic)
Ligases/Polymerases: Join molecules (anabolic)
Lyases: Split molecules without water (catabolic)
Oxidoreductases: Transfer electrons or hydrogen atoms
Transferases: Move functional groups between molecules
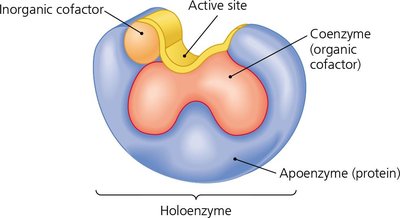
Enzymes may require cofactors (inorganic ions or organic coenzymes) for activity. The combination of an apoenzyme and its cofactor forms a holoenzyme.
Enzyme Function and Regulation
Enzymes lower the activation energy required for reactions.
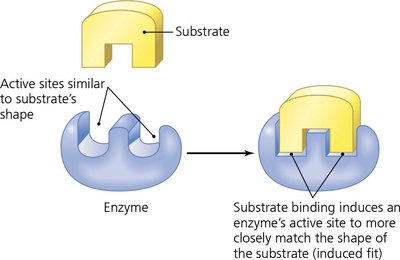
Substrate binding induces a conformational change (induced fit model).
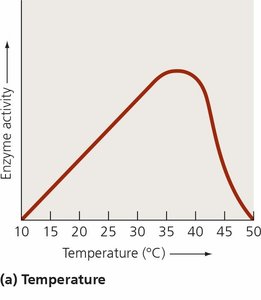
Factors Affecting Enzyme Activity
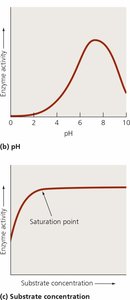
Temperature
pH
Enzyme and substrate concentrations
Presence of inhibitors
Enzyme Regulation
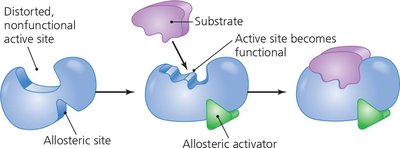
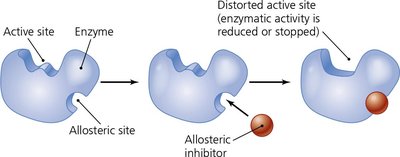
Allosteric activation: Cofactor binding at a site other than the active site activates the enzyme.
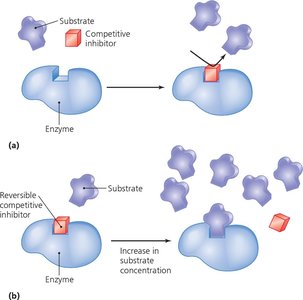
Inhibition: Competitive inhibitors block the active site; noncompetitive inhibitors bind allosteric sites, altering enzyme activity.
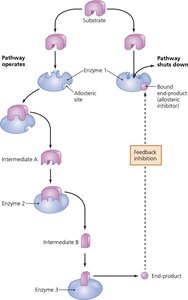
Feedback inhibition: End-product of a pathway inhibits an earlier enzyme, regulating pathway activity.
Carbohydrate Catabolism
Overview
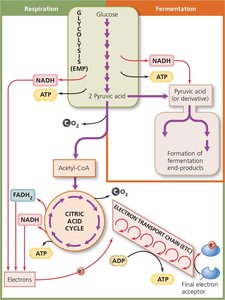
Carbohydrate catabolism is the primary means by which cells obtain energy. Glucose is the most common substrate, catabolized by cellular respiration or fermentation.
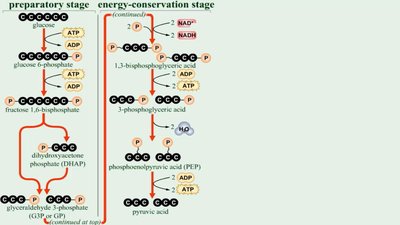
Glycolysis
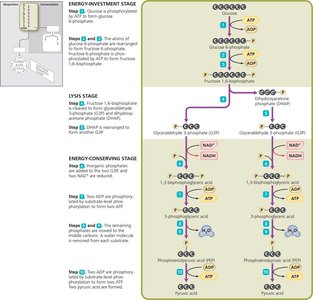
Glycolysis occurs in the cytoplasm and splits glucose into two pyruvic acid molecules, yielding a net gain of 2 ATP and 2 NADH. It consists of three stages:
Energy-investment stage
Lysis stage
Energy-conserving stage
Cellular Respiration
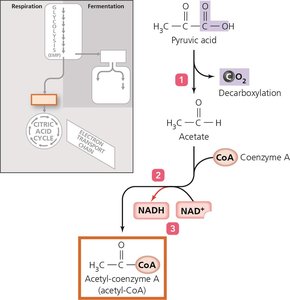
Cellular respiration completely oxidizes pyruvic acid to produce ATP via three stages:
Synthesis of acetyl-CoA
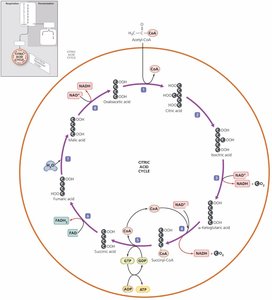
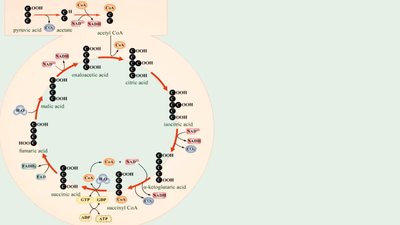
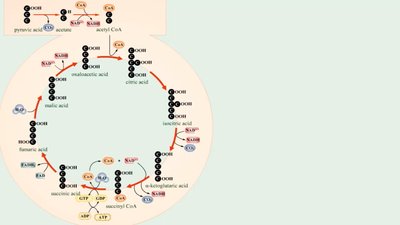
Krebs cycle (citric acid cycle)
Electron transport chain (ETC)
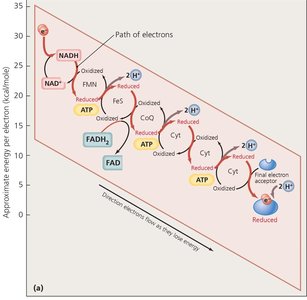
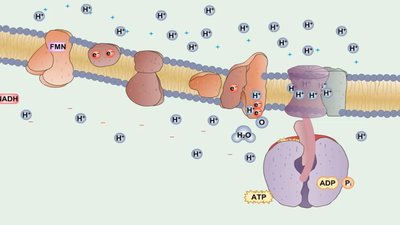
Electron Transport Chain and Chemiosmosis
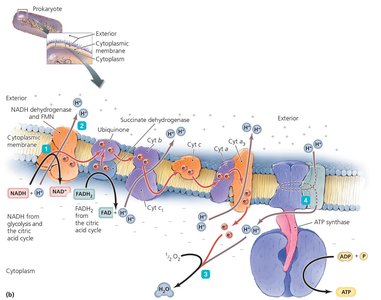
The ETC is a series of carrier molecules that transfer electrons to a final electron acceptor, generating a proton gradient used by ATP synthase to produce ATP (oxidative phosphorylation).
Aerobic respiration: Oxygen is the final electron acceptor.
Anaerobic respiration: Other molecules (e.g., nitrate, sulfate) serve as final acceptors.

Summary Table: Prokaryotic Aerobic Respiration
Pathway | ATP Produced | ATP Used | NADH Produced | FADH2 Produced |
|---|---|---|---|---|
Glycolysis | 4 | 2 | 2 | 0 |
Synthesis of acetyl-CoA & Citric Acid Cycle | 2 | 0 | 8 | 2 |
Electron Transport Chain | 34 | 0 | 0 | 0 |
Total | 40 | 2 | ||
Net Total | 38 |
Metabolic Diversity: Alternative Pathways
Entner-Doudoroff pathway: Found in some prokaryotes, yields 1 ATP, 1 NADH, and 1 NADPH per glucose.
Pentose phosphate pathway: Produces precursor metabolites and NADPH for biosynthesis.
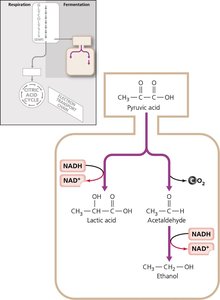
Fermentation
Fermentation provides an alternative to respiration when cells cannot fully oxidize glucose. It regenerates NAD+ by transferring electrons to organic molecules, producing various end-products (e.g., lactic acid, ethanol).
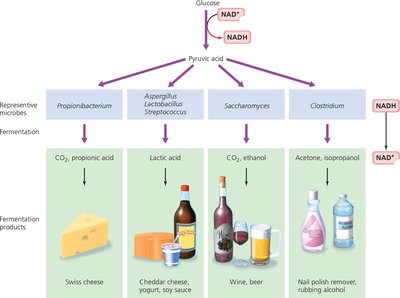
Comparison Table: Aerobic Respiration, Anaerobic Respiration, and Fermentation
Aerobic Respiration | Anaerobic Respiration | Fermentation | |
|---|---|---|---|
Oxygen Required | Yes | No | No |
Type of Phosphorylation | Substrate-level & oxidative | Substrate-level & oxidative | Substrate-level |
Final Electron Acceptor | Oxygen | NO3-, SO42-, CO32-, etc. | Cellular organic molecules |
ATP Yield (per glucose) | 38 (prokaryotes) | 4–36 | 2 |
Other Catabolic Pathways
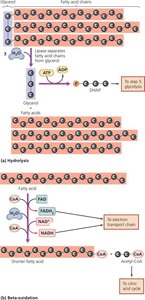
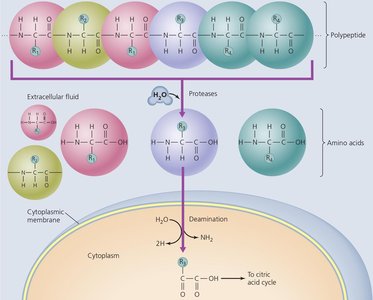
Lipid and Protein Catabolism
Lipids and proteins can be catabolized to provide energy and precursor metabolites for biosynthesis. Lipids are broken down by lipases and beta-oxidation; proteins are degraded by proteases and deaminated before entering central metabolic pathways.
Photosynthesis
Overview and Structures
Photosynthesis is the process by which organisms synthesize organic molecules from CO2 and H2O using light energy. Chlorophylls and photosystems are key components, with light-dependent and light-independent (Calvin-Benson cycle) reactions.
Light-Dependent and Light-Independent Reactions
Light-dependent reactions: Use light energy to generate ATP and NADPH via photophosphorylation (cyclic and noncyclic).
Light-independent reactions: Use ATP and NADPH to fix carbon dioxide into glucose (Calvin-Benson cycle).
Comparison Table: Types of Phosphorylation
Source of Phosphate | Source of Energy | Location in Eukaryotes | Location in Prokaryotes | |
|---|---|---|---|---|
Substrate-Level | Organic molecule | High-energy phosphate bond | Cytosol, mitochondrial matrix | Cytosol |
Oxidative | Inorganic phosphate | Proton motive force | Inner mitochondrial membrane | Cytoplasmic membrane |
Photophosphorylation | Inorganic phosphate | Proton motive force | Thylakoid of chloroplast | Thylakoid of cytoplasmic membrane |
Other Anabolic Pathways
Anabolism and Precursor Metabolites
Anabolic reactions synthesize macromolecules using energy (ATP) and precursor metabolites derived from catabolic pathways. Many pathways are amphibolic (reversible).
Table: 12 Precursor Metabolites and Their Uses
Metabolite | Pathway | Macromolecule Synthesized | Functional Use |
|---|---|---|---|
Glucose 6-Phosphate | Glycolysis | Lipopolysaccharide | Outer membrane |
Fructose 6-Phosphate | Glycolysis | Peptidoglycan | Cell wall |
Glyceraldehyde 3-Phosphate | Glycolysis | Glycerol (lipids) | Energy storage |
Phosphoglyceric Acid | Glycolysis | Amino acids | Enzymes |
Phosphoenolpyruvic Acid | Glycolysis | Amino acids | Enzymes |
Pyruvic Acid | Glycolysis | Amino acids | Enzymes |
Ribose 5-Phosphate | Pentose phosphate | DNA, RNA | Genome, enzymes |
Erythrose 4-Phosphate | Pentose phosphate | Amino acids | Enzymes |
Acetyl-CoA | Citric acid cycle | Fatty acids | Membranes |
α-Ketoglutaric Acid | Citric acid cycle | Amino acids | Enzymes |
Succinyl-CoA | Citric acid cycle | Heme | Electron carrier |
Oxaloacetate | Citric acid cycle | Amino acids | Enzymes |
Integration and Regulation of Metabolic Function
Cells regulate metabolism by controlling enzyme synthesis and activity, choosing energy sources, and using feedback inhibition. Eukaryotes compartmentalize pathways in organelles, and amphibolic pathways are regulated by coenzyme specificity.
Control of gene expression: Regulates enzyme production.
Control of metabolic expression: Regulates enzyme activity post-production.
Summary: Microbial metabolism is a complex, highly regulated network of catabolic and anabolic pathways that enable microbes to grow, reproduce, and adapt to their environments. Understanding these processes is fundamental to microbiology and biotechnology.
