 Back
BackMicrobial Metabolism: Key Concepts and Pathways
Study Guide - Smart Notes

Microbial Metabolism
Basic Chemical Reactions Underlying Metabolism
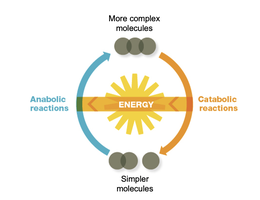
Microbial metabolism encompasses all chemical reactions occurring within a living organism. These reactions are essential for energy production, growth, and maintenance. Metabolism is divided into two main types: catabolism and anabolism.
Catabolism: The breakdown of macromolecules and smaller molecules to release energy. These reactions are exergonic (energy-releasing).
Anabolism: The synthesis of macromolecules from subunits, requiring energy input. These reactions are endergonic (energy-requiring).
Reactants: The starting components of a reaction, also known as substrates when enzymes are involved.
Products: The ending components or end products of a pathway.
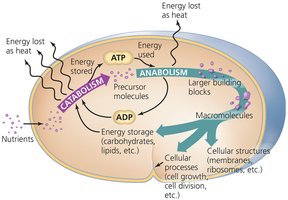
Energy Production and ATP
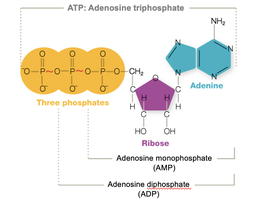
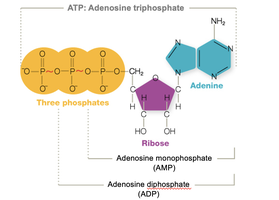
Energy is produced by breaking bonds in nutrients and reforming high-energy bonds in ATP (adenosine triphosphate). ATP acts as the cell's energy currency, storing energy for cellular work. Energy production involves coupled oxidation and reduction reactions.
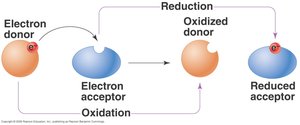
Oxidation: Loss of electrons (LEO: Lose Electrons = Oxidation).
Reduction: Gain of electrons (GER: Gain Electrons = Reduction).
NADH: The reduced form of NAD+, carries electrons and is referred to as reducing power. NADH can transfer electrons to generate a proton motive force.
ATP: Not much ATP is stored; it is produced as needed. When the high-energy bond breaks, work is done.
The Roles of Enzymes in Metabolism
Enzymes are essential for metabolic pathways, coupling anabolic and catabolic reactions. The metabolic pathways present in an organism are dictated by the enzymes it possesses.
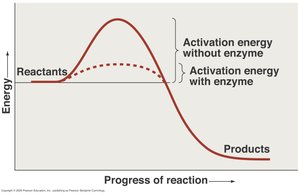
Enzymes: Biological catalysts that speed up reactions by lowering activation energy without being consumed or permanently changed.
Properties: Most enzymes are proteins (ribozymes are RNA exceptions), are specific for substrates, and have optimal pH and temperature.
Turnover Number: The number of substrate molecules converted to products per second (can be very high, e.g., 500,000/sec).
Naming: Enzymes often end in -ase or -sin and are named for the reaction they catalyze (e.g., glucose-6-phosphate phosphatase, trypsin, lysozyme).
Enzyme Structure and Function
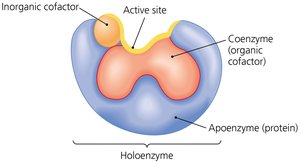
Enzymes consist of a protein part (apoenzyme) and may require cofactors (inorganic ions or organic coenzymes) to be functional. The whole, active enzyme is called a holoenzyme.
Apoenzyme: The protein component of an enzyme.
Cofactor: An ion or organic molecule required for enzyme function. Organic cofactors are called coenzymes.
Coenzymes: Often act as electron carriers or donors (e.g., NAD+, NADP+, FMN, FAD, CoA).
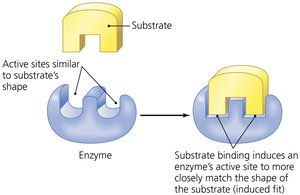
Active Site: Substrates must fit into a specific pocket or shape on the enzyme.
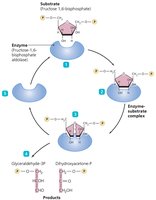
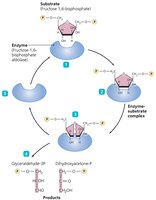
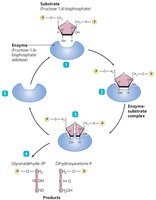
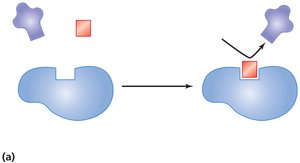
Mechanism of Enzyme Action
Enzyme action involves several steps:
Substrate binds to the enzyme's active site.
Temporary bonds (usually hydrogen bonds) form, creating an enzyme-substrate complex.
Substrate changes: either combination of substrates to form one product or breakdown of a substrate into two products.
Products are released as they no longer fit the active site.
The enzyme is free to react with new substrate molecules.
Factors Affecting Enzyme Activity
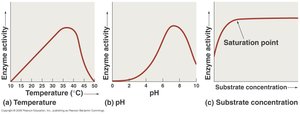
Enzyme activity is influenced by several factors:
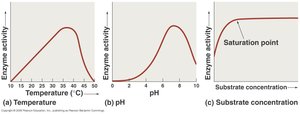
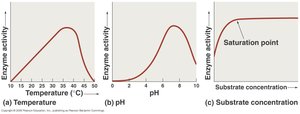
Temperature: Determines the enzyme's 3D structure and functionality. Enzyme activity follows a bell-shaped curve with temperature.
pH: Affects the 3D structure and activity, also follows a bell-shaped curve.
Substrate Concentration: Activity increases with substrate concentration until the enzyme becomes saturated.
Enzyme Inhibition
Enzyme activity can be regulated or inhibited by various mechanisms:
Inhibitor: Slows down or stops enzyme activity. Used to limit microbial growth by targeting microbial enzymes.
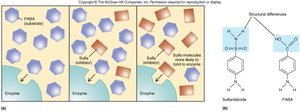
Competitive Inhibitor: Competes with substrate for the active site.
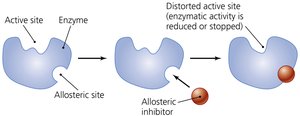
Noncompetitive (Allosteric) Inhibitor: Binds elsewhere on the enzyme, changing its conformation and reducing activity.
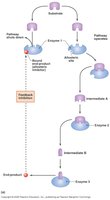
Feed-back Inhibition: The end-product of a pathway inhibits an earlier step, preventing unnecessary product formation.
Carbohydrate Catabolism
Overview of Carbohydrate Catabolism
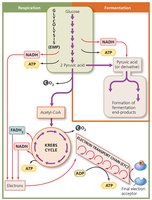
Carbohydrate catabolism is the breakdown of sugars to produce ATP. Glucose is the primary fuel for most organisms, serving as both an energy source and a carbon source for macromolecule synthesis.
Aerobic Cellular Respiration: Uses oxygen as the final electron acceptor.
Anaerobic Cellular Respiration: Uses an inorganic molecule other than oxygen as the final electron acceptor.
Fermentation: Uses an organic molecule (such as pyruvate) as the final electron acceptor.
Summary of Glucose Catabolism Pathways
Cellular respiration consists of three main stages: glycolysis, Krebs cycle, and the electron transport chain. These pathways generate ATP and a proton motive force in prokaryotic cells.
Key Terms and Concepts
Proton Motive Force: Generated by the electron transport chain in the plasma membrane of prokaryotic cells.
ATP: The energy currency for all life forms.
NADH: Electron carrier, reduced form of NAD+.
Enzyme: Biological catalyst, lowers activation energy.
Summary Table: Types of Metabolic Reactions
Type | Description | Energy |
|---|---|---|
Catabolism | Breakdown of molecules | Releases energy (exergonic) |
Anabolism | Synthesis of molecules | Requires energy (endergonic) |
Summary Table: Types of Enzyme Inhibition
Type | Mechanism | Example |
|---|---|---|
Competitive | Competes for active site | Sulfa drugs inhibit folate synthesis |
Noncompetitive (Allosteric) | Binds to allosteric site, changes enzyme shape | Allosteric inhibitors |
Feedback | End-product inhibits earlier step | Metabolic pathway regulation |
Key Equations
ATP hydrolysis:
Oxidation-Reduction:
Additional info: Academic context was added to clarify enzyme mechanisms, metabolic pathways, and regulatory processes.
