 Back
BackMicrobial Metabolism: Pathways, Enzymes, and Energy Production
Study Guide - Smart Notes

Microbial Metabolism
Introduction to Microbial Metabolism
Microbial metabolism encompasses all the chemical reactions that occur within a microorganism, enabling it to grow, reproduce, maintain its structures, and respond to environments. These reactions are broadly categorized into catabolic (energy-releasing) and anabolic (energy-consuming) processes.
Energy and Work in Microbes
Types of Cellular Work
Chemical work: Synthesis of complex molecules from simpler ones.
Transport work: Movement of substances across cellular membranes.
Mechanical work: Physical movement, such as motility.

Energy Sources for Microbes
ATP: The universal energy currency of the cell.
Light: Utilized by phototrophs for energy.
Organic chemicals: Used by chemoorganotrophs.
Inorganic chemicals: Used by chemolithotrophs.


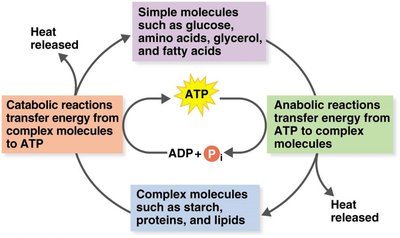
Catabolic and Anabolic Reactions
Overview of Catabolism and Anabolism
Catabolism: The breakdown of complex molecules into simpler ones, releasing energy and providing building blocks for anabolism.
Anabolism: The synthesis of complex molecules from simpler ones, requiring energy input.
Metabolic pathways are sequences of enzymatically catalyzed reactions, determined by the cell's enzymes, which are encoded by genes.
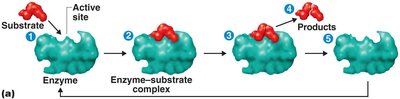
Enzymes and Their Role in Metabolism
Enzyme Function and Mechanism
Enzymes are biological catalysts that speed up chemical reactions by lowering the activation energy required. They are highly specific for their substrates and are not consumed in the reaction.
Activation energy: The minimum energy required to initiate a chemical reaction.
Enzyme-substrate complex: Temporary association between enzyme and substrate during catalysis.

Enzyme Classification
Oxidoreductase: Catalyzes oxidation-reduction reactions.
Transferase: Transfers functional groups between molecules.
Hydrolase: Catalyzes hydrolysis reactions.
Lyase: Removes groups of atoms without hydrolysis.
Isomerase: Rearranges atoms within a molecule.
Ligase: Joins two molecules, usually with ATP hydrolysis.
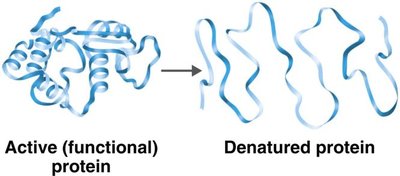
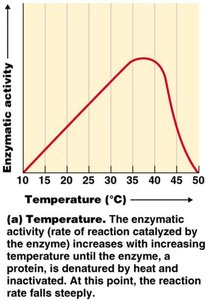
Factors Influencing Enzyme Activity
Temperature: Each enzyme has an optimal temperature; high temperatures can denature enzymes.
pH: Each enzyme has an optimal pH; extreme pH can denature enzymes.
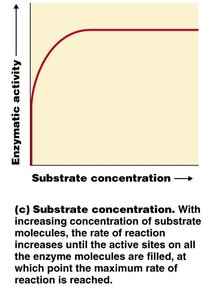
Substrate concentration: Increasing substrate increases reaction rate until saturation.
Inhibitors: Chemicals that reduce enzyme activity (competitive and noncompetitive).
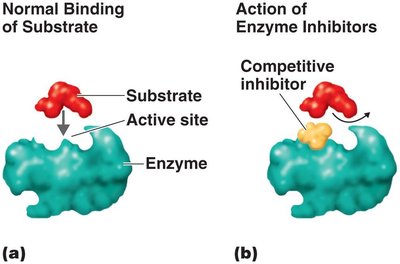
Enzyme Inhibition
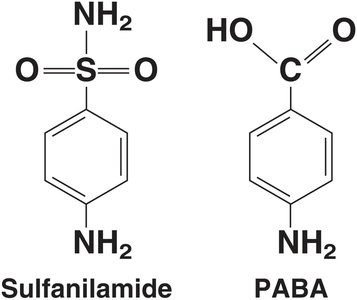
Competitive inhibition: Inhibitor competes with substrate for the active site.
Noncompetitive inhibition: Inhibitor binds elsewhere, changing enzyme shape and function.
Oxidation and Reduction in Metabolism
Redox Reactions
Oxidation: Loss of electrons.
Reduction: Gain of electrons.
Redox reaction: Coupled oxidation and reduction reactions.

Electron Carrier Molecules
NAD+
NADP+
FAD
Coenzyme Q
Cytochromes
ATP Formation Mechanisms
Three Main Mechanisms
Substrate-level phosphorylation: Direct transfer of phosphate to ADP from a phosphorylated substrate.
Oxidative phosphorylation: ATP generated from the energy released by electrons as they are transferred to oxygen (or another final electron acceptor) via the electron transport chain.
Photophosphorylation: ATP formed using light energy, primarily in photosynthetic organisms.


Catabolic Pathways
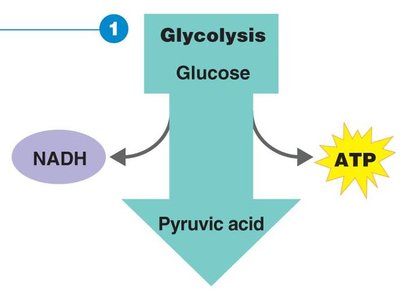
Glycolysis
Glycolysis is the oxidation of glucose to pyruvic acid, producing ATP and NADH. It is the first step in both aerobic and anaerobic respiration.
Occurs in the cytoplasm.
Does not require oxygen.
Net gain: 2 ATP and 2 NADH per glucose molecule.

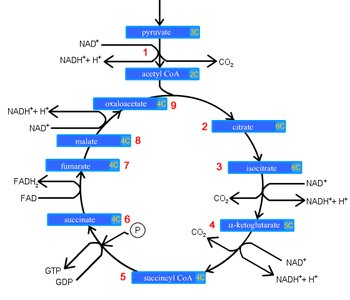
Krebs Cycle (Citric Acid Cycle)
The Krebs cycle completes the oxidation of organic molecules, generating NADH, FADH2, and ATP, and releasing CO2.
Occurs in the cytoplasm (prokaryotes) or mitochondria (eukaryotes).
Produces electron carriers for the electron transport chain.
Electron Transport Chain (ETC) and Chemiosmosis
The ETC is a series of carrier molecules that transfer electrons from NADH and FADH2 to a final electron acceptor, releasing energy used to generate ATP by chemiosmosis.
In aerobic respiration, the final electron acceptor is O2.
In anaerobic respiration, the final electron acceptor is an inorganic molecule other than O2 (e.g., NO3-, SO42-).


Summary Table: Electron Acceptors and Products in Anaerobic Respiration
Electron Acceptor | Products |
|---|---|
NO3- | NO2-, N2 + H2O |
SO42- | H2S + H2O |
CO32- | CH4 + H2O |
Fermentation
Fermentation is an anaerobic process that releases energy from the oxidation of organic molecules. It does not require oxygen, the Krebs cycle, or the ETC, and uses an organic molecule as the final electron acceptor.
Produces less ATP than respiration.
Common products: lactic acid, ethanol, CO2.
Protein Catabolism
Proteins are broken down into amino acids, which can be deaminated and enter central metabolic pathways such as glycolysis or the Krebs cycle.
Biochemical Tests in Microbiology
Biochemical tests are used to identify bacteria based on their metabolic characteristics, such as the ability to ferment specific sugars or produce certain enzymes (e.g., urease).
Additional info: The above notes integrate and expand upon the provided slides, ensuring a comprehensive overview of microbial metabolism suitable for college-level microbiology students.
