 Back
BackMicrobial Metabolism: Structured Study Notes for College Microbiology
Study Guide - Smart Notes

Microbial Metabolism
Introduction to Energy
Energy is fundamental to all biological processes, enabling organisms to perform work and drive chemical reactions. In biology, work refers to the transfer of energy that results in a change in the state of matter.
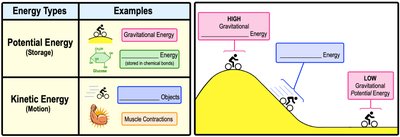
Potential Energy: Stored energy available to do work, such as energy in chemical bonds or gravitational energy.
Kinetic Energy: Energy of motion, such as moving objects or muscle contractions.
Example: A glucose molecule contains potential energy, while water rushing over a waterfall demonstrates kinetic energy.
Thermodynamics in Biological Systems
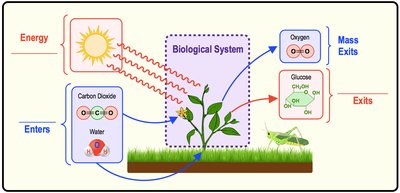
Thermodynamics is the study of energy transfers between bodies of matter. Biological systems are defined as the specific portion of matter being studied, while surroundings are everything outside the system. Biological systems exchange both energy and mass with their surroundings.
System: The part of the universe being studied (e.g., a cell).
Surroundings: Everything outside the system.
Importance: Principles of thermodynamics govern chemical processes and energy exchange in all living organisms.
Laws of Thermodynamics
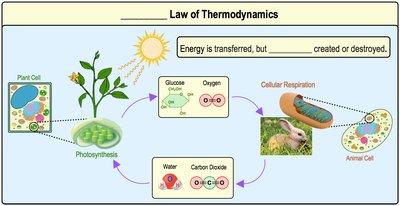
First Law of Thermodynamics
The first law states that energy can be transferred and transformed, but it cannot be created or destroyed. This is also known as the Principle of Conservation of Energy. The total amount of energy in the universe remains constant.
Example: Energy from sunlight is transferred to plants via photosynthesis and then to animals via cellular respiration.
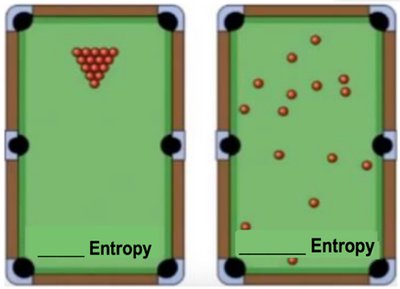
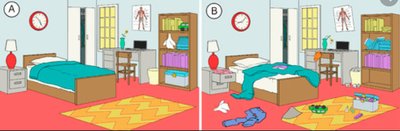
Entropy
Entropy is a measure of disorder or randomness. The greater the disorder, the higher the entropy. Reactions tend to move the universe toward higher entropy, but energy input can decrease entropy locally.
Example: A neatly arranged room (low entropy) versus a messy room (high entropy).
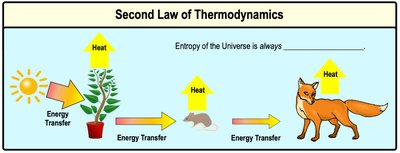
Second Law of Thermodynamics
The second law states that energy conversions are never 100% efficient; some energy is always lost as heat, increasing universal entropy.
Heat: A form of kinetic energy transferred between objects with different temperatures.
Example: Energy transfer up the food chain results in heat loss at each step.
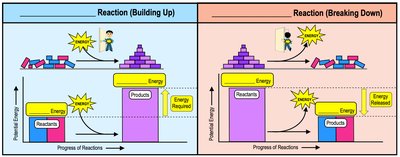
Chemical Reactions
Chemical reactions involve the making and breaking of chemical bonds, leading to changes in matter. Reactants are the starting materials, and products are the resulting materials.
Endergonic Reactions: Require an input of energy (energy enters the reaction).
Exergonic Reactions: Release energy (energy exits the reaction).
Example: Building up molecules (endergonic) vs. breaking down molecules (exergonic).
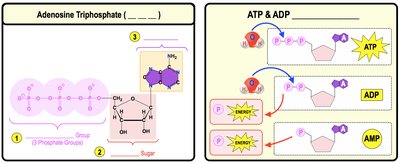
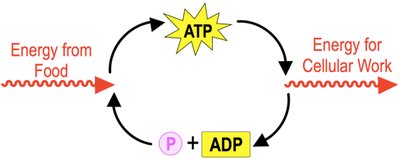
Adenosine Triphosphate (ATP)
ATP is the primary energy molecule used to power cellular activities. It consists of three phosphate groups, a ribose sugar, and an adenine nitrogenous base. ATP hydrolysis releases energy by breaking bonds between phosphate groups, forming ADP and inorganic phosphate (Pi).
ATP Hydrolysis:
Example: Energy from food is stored in ATP and used for cellular work.
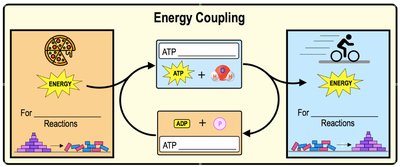
Energy Coupling and Phosphorylation
Energy coupling occurs when energy released by an exergonic reaction is used to drive an endergonic reaction. ATP hydrolysis is often coupled to endergonic reactions to provide the necessary energy input.
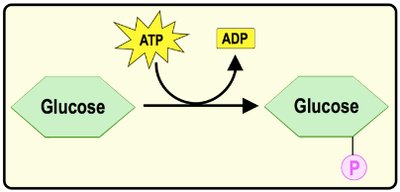
Phosphorylation: Transfer of a phosphate group from ATP to another molecule, activating it or changing its function.
Example: ATP phosphorylates glucose to initiate glycolysis.
Enzymes and Catalysis
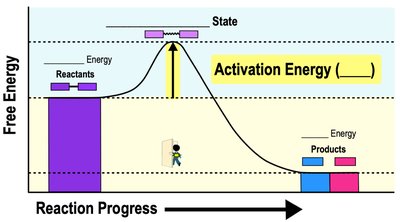
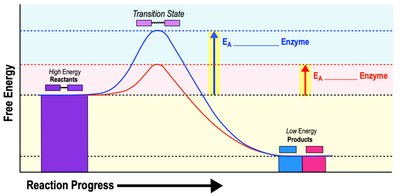
Enzymes are biological catalysts that speed up chemical reactions without being consumed. Substrates are the reactants in enzyme-catalyzed reactions. Enzymes lower the activation energy required for reactions, increasing reaction rates.
Functions: Building molecules, copying DNA, digesting food.
Activation Energy (EA): Minimum energy required to start a reaction.
Transition State: Temporary high-energy state during a reaction.
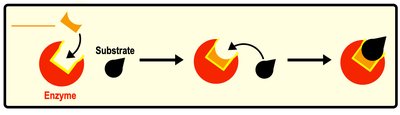
Enzyme Binding and Cofactors
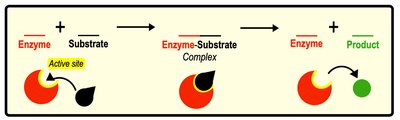
Substrates bind to the enzyme's active site, forming an enzyme-substrate complex. After catalysis, products are released, and the enzyme remains unchanged. Some enzymes require cofactors (non-protein substances) or coenzymes (organic cofactors) for activity.
Active Site: Region of enzyme where substrate binds.
Cofactors: Metal ions or organic molecules aiding catalysis.
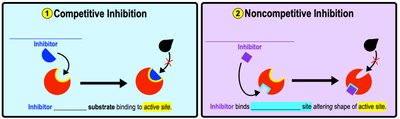
Enzyme Inhibition
Enzyme inhibitors interfere with enzyme activity. Competitive inhibitors compete for the active site, while noncompetitive inhibitors bind to an allosteric site, changing the enzyme's shape and reducing activity.
Competitive Inhibition: Inhibitor binds to active site.
Noncompetitive Inhibition: Inhibitor binds elsewhere, altering active site.
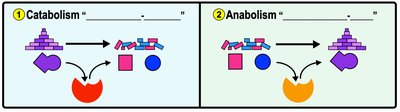
Metabolism: Catabolic and Anabolic Pathways
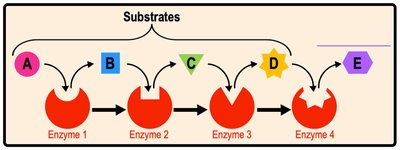
Metabolism encompasses all chemical reactions in an organism. Metabolic pathways are series of reactions altering substrates stepwise.
Catabolic Pathways: Break down molecules, releasing energy (e.g., glycolysis).
Anabolic Pathways: Build larger molecules, consuming energy (e.g., protein synthesis).
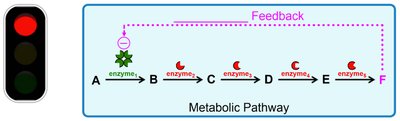
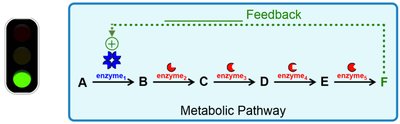
Feedback Regulation
Metabolic pathways are regulated by feedback mechanisms. Negative feedback inhibits earlier steps, while positive feedback stimulates them.
Negative Feedback: Final product inhibits pathway (e.g., end-product inhibition).
Positive Feedback: Final product stimulates pathway.
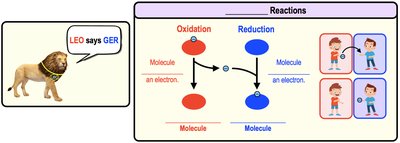
Redox Reactions
Oxidation-reduction (redox) reactions transfer electrons between molecules. Oxidation is the loss of electrons, while reduction is the gain of electrons. These reactions are coupled and essential for energy transfer in cells.
LEO the Lion says GER: Lose Electrons = Oxidation; Gain Electrons = Reduction.
Example: Glucose is oxidized, NAD+ is reduced to NADH.

Electron Carriers: NADH, FADH2, NADPH
Electron carriers such as NADH, FADH2, and NADPH transport electrons during metabolic processes. NADH and FADH2 are used in cellular respiration, while NADPH is used in biosynthetic (anabolic) reactions.
NADH/FADH2: Shuttle electrons to the electron transport chain.
NADPH: Used in photosynthesis and biosynthesis.
Summary Table: Catabolic vs. Anabolic Pathways
Pathway | Function | Energy | Example |
|---|---|---|---|
Catabolic | Breaks down molecules | Releases | Glycolysis |
Anabolic | Builds molecules | Consumes | Protein synthesis |
Additional info: These notes cover foundational concepts in microbial metabolism, including energy, thermodynamics, chemical reactions, ATP, enzymes, metabolic pathways, feedback regulation, and redox reactions. The included images directly reinforce the explanations and are selected for their clear relevance to the adjacent content.
