 Back
BackMicrobial Nutrition and Metabolism: Laboratory Media, Nutrient Acquisition, and Catabolic Diversity
Study Guide - Smart Notes

Microbial Nutrition and Laboratory Culture Media
Macronutrients and Micronutrients
Microorganisms require a variety of nutrients for growth and metabolism. These nutrients are classified based on their abundance and necessity for cellular processes.
Macronutrients: Elements required in large amounts, including carbon (C), hydrogen (H), nitrogen (N), oxygen (O), phosphorus (P), sulfur (S), potassium (K), magnesium (Mg), and calcium (Ca). These are the most abundant elements in cells and are essential for building cellular structures and molecules.
Micronutrients (Trace Elements): Elements needed in much smaller quantities, such as iron (Fe), manganese (Mn), cobalt (Co), zinc (Zn), copper (Cu), nickel (Ni), selenium (Se), and molybdenum (Mo). Despite their low abundance, these elements are crucial for enzyme function and overall viability.
Acquisition of Essential Nutrients: Iron and Siderophores
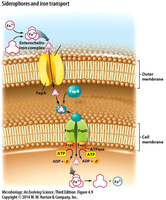
Iron is a vital micronutrient for many cellular processes but is often found in the environment as insoluble Fe3+, making it difficult for bacteria to acquire. To overcome this, bacteria synthesize and secrete siderophores, which are specialized molecules that bind (chelate) Fe3+ and facilitate its transport into the cell.
Siderophores: High-affinity iron-chelating compounds secreted by bacteria to scavenge iron from the environment.
Transport Mechanism: In Gram-negative bacteria, siderophore-Fe3+ complexes are transported across the outer membrane via specific proteins (e.g., FepA), then through the periplasm and into the cytoplasm using ABC transporters, where Fe3+ is reduced to Fe2+.
Example: Escherichia coli produces enterochelin, a potent siderophore.
Types of Laboratory Culture Media
Defined (Minimal) vs. Complex Media
Microbiologists use different types of culture media to grow bacteria in the laboratory, depending on the experimental needs and the nutritional requirements of the organisms.
Defined (Minimal) Media: All chemical components and their concentrations are precisely known. Used for experiments where control over nutrient composition is essential.
Complex Media: Contains extracts or digests of natural materials (e.g., yeast extract, peptone), so the exact composition is not fully known. Used for routine cultivation and for organisms with complex nutritional requirements.
Examples of Common Complex Media
Trypticase Soy Agar (TSA): A general-purpose medium supporting the growth of many bacteria.
Blood Agar: Enriched medium containing blood, used for growing fastidious organisms and for detecting hemolytic activity.
Chocolate Agar: Contains lysed blood cells, supporting the growth of even more fastidious bacteria.

Comparison of Defined and Complex Media
The choice of medium can reveal physiological differences between microorganisms. For example, E. coli can grow on minimal defined media, while Leuconostoc mesenteroides requires complex media due to its demanding nutritional requirements.
Medium Type | Defined Medium for E. coli | Defined Medium for L. mesenteroides | Complex Medium (for either) |
|---|---|---|---|
Major Components | Glucose, salts, NH4Cl, MgSO4, CaCl2, trace elements | Glucose, salts, amino acids, vitamins, nucleotides, trace elements | Peptone, yeast extract, NaCl |
Growth Support | E. coli | L. mesenteroides | Both |

Microbial Growth in the Laboratory and the Environment
Environmental Sampling and the Uncultured Majority
Many microbes observed in nature cannot be cultured in the laboratory using standard media. This phenomenon is known as the "Great Plate Count Anomaly." Environmental sampling and microscopy reveal a much greater diversity than what is recovered by culturing.
Reasons for Unculturability: Unknown nutritional requirements, dependence on other organisms, or sensitivity to laboratory conditions.
Approaches to Culturing the Unculturable: Mimicking natural environments, co-culturing with helper species, or using culture-free DNA sequencing methods to study microbial diversity.

Microbial Metabolism: Catabolism and Anabolism
Overview of Metabolism
Microbial metabolism encompasses all chemical reactions that occur within a cell. These reactions are divided into two main categories:
Catabolism: The breakdown of molecules to release energy, which is used to generate ATP.
Anabolism: The synthesis of cellular components, using energy stored in ATP.
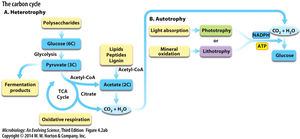
Classification by Carbon and Energy Sources
Microorganisms are classified based on their sources of carbon and energy:
Heterotrophs: Obtain carbon from organic compounds (e.g., sugars, lipids, proteins).
Autotrophs: Use carbon dioxide (CO2) as their carbon source (carbon fixation).
Phototrophs: Derive energy from light.
Organotrophs: Obtain energy from organic compounds.
Lithotrophs: Obtain energy from inorganic compounds (e.g., H2, Fe2+, H2S).
These terms can be combined to describe metabolic strategies, such as photoautotrophy (light energy, CO2 carbon) or organoheterotrophy (organic energy and carbon).
Energy Generation: Oxidation–Reduction Reactions
Redox Reactions in Metabolism
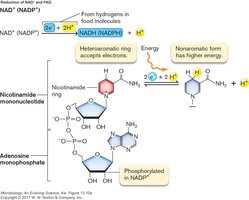
Energy generation in cells depends on oxidation–reduction (redox) reactions, where electrons are transferred from electron donors to electron acceptors. The further electrons "fall" from donor to acceptor, the more energy is released.
Oxidation: Loss of electrons (and usually H+).
Reduction: Gain of electrons (and usually H+).
Electron Tower: Visual representation of common redox couples, with good electron donors at the top and good acceptors at the bottom.

Electron Carriers
Electron carriers such as NAD+/NADH and FAD/FADH2 play a central role in shuttling electrons during metabolic reactions. These molecules accept electrons during catabolism and donate them to the electron transport chain or other acceptors.
ATP: The Energy Currency of the Cell
ATP Synthesis Mechanisms
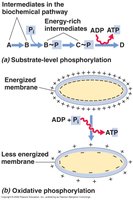
Cells synthesize ATP (adenosine triphosphate) via two main mechanisms:
Substrate-Level Phosphorylation: Direct transfer of a phosphate group to ADP from a phosphorylated intermediate during catabolism (e.g., glycolysis, fermentation).
Oxidative Phosphorylation: ATP synthesis driven by the transfer of electrons through an electron transport chain, generating a proton motive force used by ATP synthase (e.g., aerobic and anaerobic respiration).
Diversity of Catabolic Pathways and Carbon Sources
Central Catabolic Pathways
Microbes can utilize a wide variety of carbon sources, which are funneled into central metabolic pathways such as glycolysis, the tricarboxylic acid (TCA) cycle, and fermentation. These pathways enable the breakdown of complex molecules and the generation of energy and biosynthetic precursors.
Polysaccharides: Broken down into monosaccharides and funneled into glycolysis.
Lipids: Hydrolyzed into glycerol and fatty acids, which enter glycolysis and the TCA cycle, respectively.
Peptides: Degraded into amino acids, which can be deaminated and used in central metabolism.
Aromatic Compounds: Some bacteria can degrade complex aromatic molecules, including environmental pollutants.
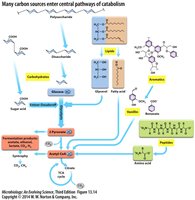
Example: Bacteria capable of degrading polychlorinated aromatics play a role in bioremediation of environmental contaminants.
Additional info: The diversity of microbial metabolism underpins their ecological roles and biotechnological applications, including waste treatment, fermentation, and environmental cleanup.
